23Days
5Hours
57Minutes
If you're not using Knowt you're already behind.
Personalized schedules, exams, and more.
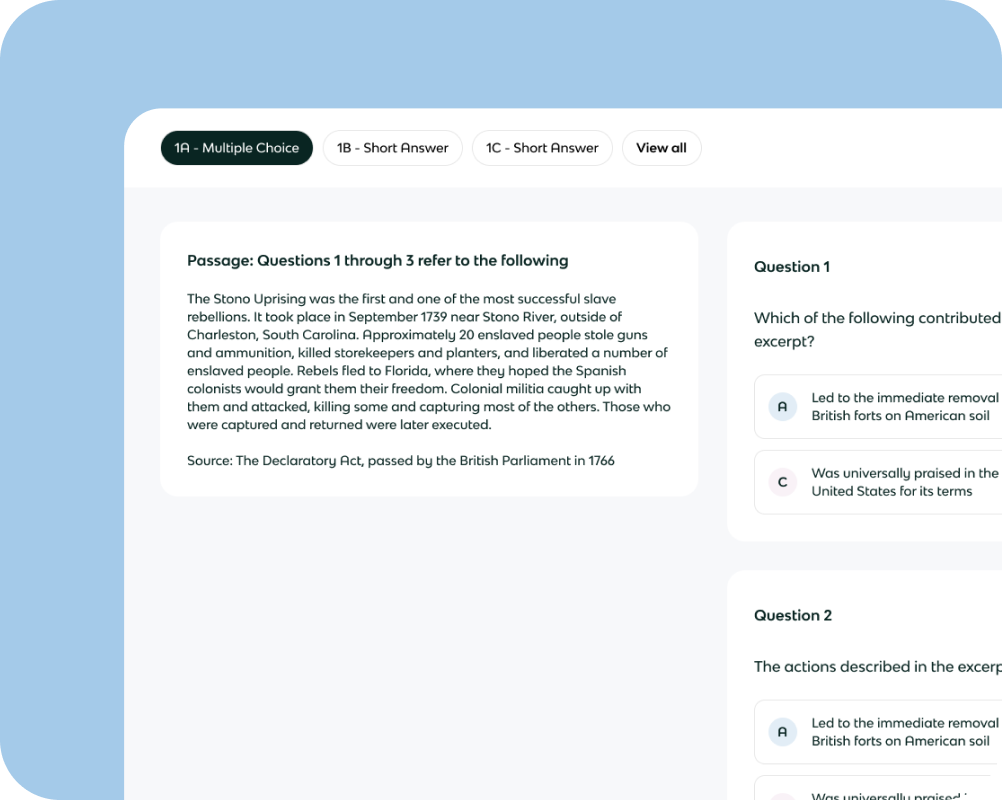
Unlimited mock exams using the same exact format as the actual AP exam
Get a custom study plan based on your exam date and the days you like to study
Study guides & practice for each unit in any AP Exam
Cram Sheets for last minute review
Start browsing our top notes & flashcards across a library of 5 million resources.
We're on the hot seat.