Mastering Thermodynamics: Energy Conservation, Entropy, and Cycles
The First Law of Thermodynamics
Concept Definition
The First Law of Thermodynamics is essentially the law of Conservation of Energy applied to a thermal system. It states that energy cannot be created or destroyed, only transferred or transformed. In the context of a gas enclosed in a cylinder (the standard AP Physics 2 model), the change in the system's internal energy is equal to the sum of energy added by heating and energy added by doing work on the system.
The Formula and Sign Conventions
The mathematical statement is:
Where:
- $\Delta U$ (Internal Energy): The change in the total kinetic and potential energy of the gas molecules. For an ideal gas, $U$ depends only on Temperature ($T$). If $T$ goes up, $U$ goes up.
- $Q$ (Heat): Energy transferred due to a temperature difference.
- $Q > 0$: Heat is added to the system (Net heat in).
- $Q < 0$: Heat travels out of the system (Net heat out).
- $W$ (Work): Energy transferred by mechanical means (compression or expansion).
CRITICAL AP SIGN CONVENTION:
The College Board typically uses the convention where $W$ is work done ON the system.
- Compression: The gas is squeezed ($V$ decreases). Work is done on the gas. $W$ is positive (+).
- Expansion: The gas expands ($V$ increases). Work is done by the gas. $W$ is negative (-).
Work and PV Diagrams
Work done on a gas at constant pressure is calculated as:
On a Pressure-Volume (PV) Diagram, the absolute value of the work done ($|W|$) is equal to the area under the curve. If the process forms a closed loop (a cycle), the area inside the loop represents the net work done per cycle.
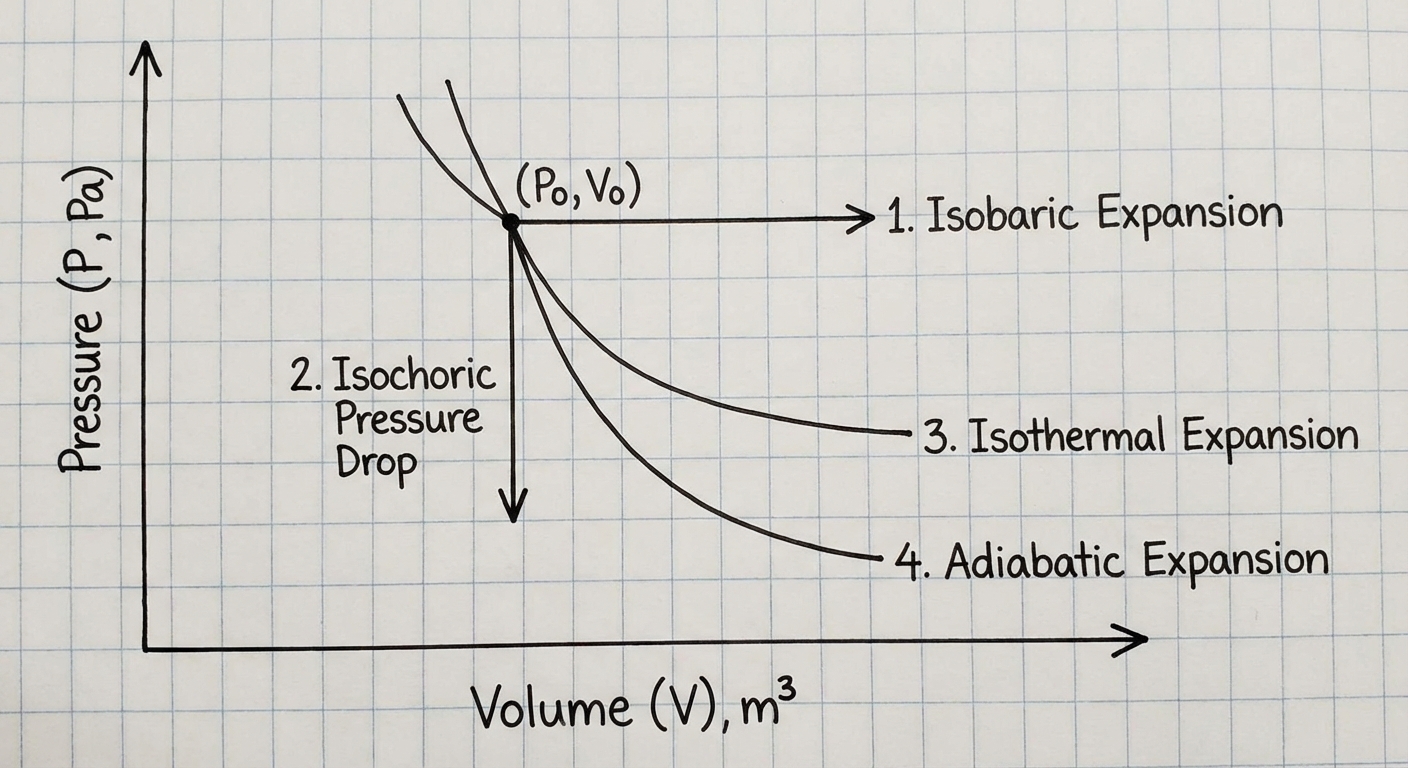
The Four Major Thermodynamic Processes
You must know how the First Law applies to these specific scenarios:
| Process | Definition | Property Held Constant | First Law Implication | Visual on PV Graph |
|---|---|---|---|---|
| Isobaric | Constant Pressure | $\Delta P = 0$ | $\Delta U = Q + W$ (Full equation used) | Horizontal line |
| Isochoric (Isovolumetric) | Constant Volume | $\Delta V = 0$ ($W = 0$) | $\Delta U = Q$ | Vertical line |
| Isothermal | Constant Temperature | $\Delta T = 0$ ($\Delta U = 0$) | $Q = -W$ | Curved line (Hyperbola) |
| Adiabatic | No Heat Transfer | $Q = 0$ | $\Delta U = W$ | Steep curve (steeper than isothermal) |
Heat Engines and Efficiency
A Heat Engine is a device that converts thermal energy into mechanical work. It operates in a cycle, meaning it returns to its initial state ($\Delta U_{cycle} = 0$).
Energy Flow in Heat Engines
- Heat ($QH$) is absorbed from a high-temperature reservoir ($TH$).
- Some of that energy is converted into useful Work ($W$).
- The remaining waste heat ($QC$) is expelled to a low-temperature reservoir ($TC$).
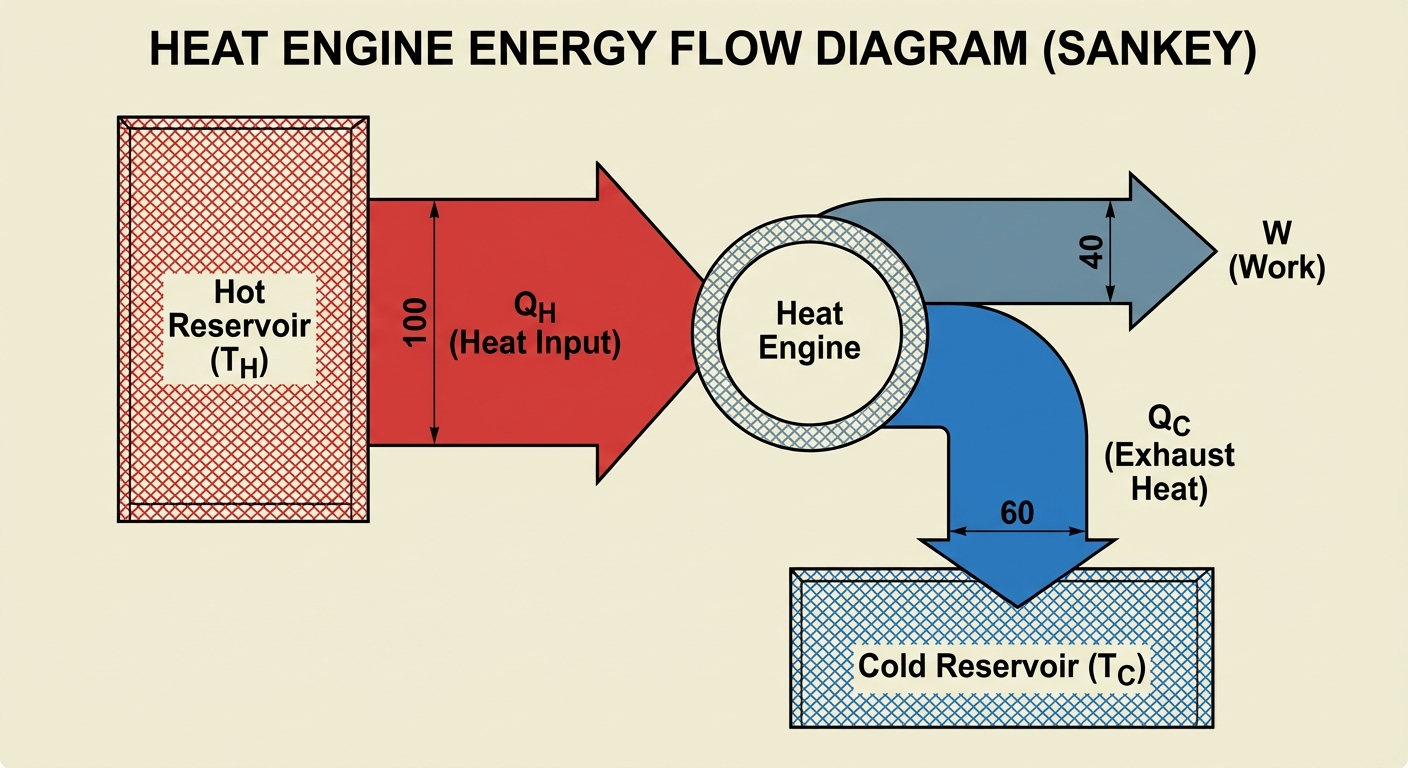
Thermal Efficiency ($e$)
Efficiency measures how much of the input heat equals useful work. It is a ratio (often expressed as a percentage).
Remember: No real engine can be 100% efficient ($e < 1$). $Q_C$ can never be zero.
The Second Law of Thermodynamics and Entropy
While the First Law says energy is conserved, the Second Law dictates the direction of thermodynamic processes. It explains why heat naturally flows from hot to cold, but never the reverse, without external work.
Entropy ($S$)
Entropy is a quantitative measure of disorder or randomness in a system. In statistical mechanics, it relates to the number of microstates (arrangements of molecules) accessible to a system.
- Solids have low entropy (highly ordered).
- Gases have high entropy (highly disordered).
Determining Entropy Change
For a reversible process occurring at a constant temperature $T$ (in Kelvin):
- If heat is added ($Q > 0$), entropy increases.
- If heat is removed ($Q < 0$), entropy decreases.
Statement of the Second Law
There are several equivalent ways to state the Second Law:
- Entropy Statement: The total entropy of the universe (System + Surroundings) always increases for irreversible natural processes and remains constant for reversible processes. It never decreases.
- Heat Flow Statement: Heat never flows spontaneously from a cold object to a hot object.
- Efficiency Statement (Carnot): No heat engine operating between two given heat reservoirs can be more efficient than a Carnot engine (an ideal, reversible engine).
The Carnot Efficiency (Maximum Efficiency)
The theoretical maximum efficiency for any heat engine depends only on the temperatures of the reservoirs:
Warning: You MUST use the Kelvin scale for this calculation.
Common Mistakes & Pitfalls
Celsius vs. Kelvin Errors:
- The Mistake: Using Celsius in the Ideal Gas Law ($PV=nRT$) or the Carnot Efficiency formula ($e = 1 - TC/TH$).
- The Fix: Always convert to Kelvin ($K = ^{\circ}C + 273$) immediately. $\Delta T$ is the same in C and K, but plain $T$ is not.
Sign Convention Confusion:
- The Mistake: Getting the sign of $W$ wrong.
- The Fix: If the gas Expands, energy leaves the system as work ($W$ is negative). If the gas is Compressed, energy enters the system ($W$ is positive).
Comparing $\Delta S{sys}$ vs. $\Delta S{univ}$:
- The Mistake: Thinking the entropy of a system cannot decrease. It can (e.g., water freezing into ice).
- The Fix: The entropy of a specific system can decrease, but the entropy of the surroundings must increase by a larger amount so that the total $\Delta S_{universe} \ge 0$.
Misunderstanding Cycle Area:
- The Mistake: Thinking the area under a curve is heat ($Q$).
- The Fix: The area under a PV curve is the magnitude of Work ($W$). Heat ($Q$) must be solved for using the First Law $\Delta U = Q + W$.