Unit 7: Understanding the Quantum Atom and Nuclear Physics
In AP Physics 2, we transition from the deterministic world of classical mechanics to the probabilistic and discrete world of Modern Physics. This section focuses on the behavior of matter on the smallest scales: electron energy states within the atom and the powerful interactive forces within the nucleus.
Atomic Energy Levels and Emission Spectra
Classical physics predicted that electrons orbiting a nucleus should emit radiation continuously and crash into the nucleus. They don't. Modern physics explains this using quantized energy levels.
The Bohr Model and Energy States
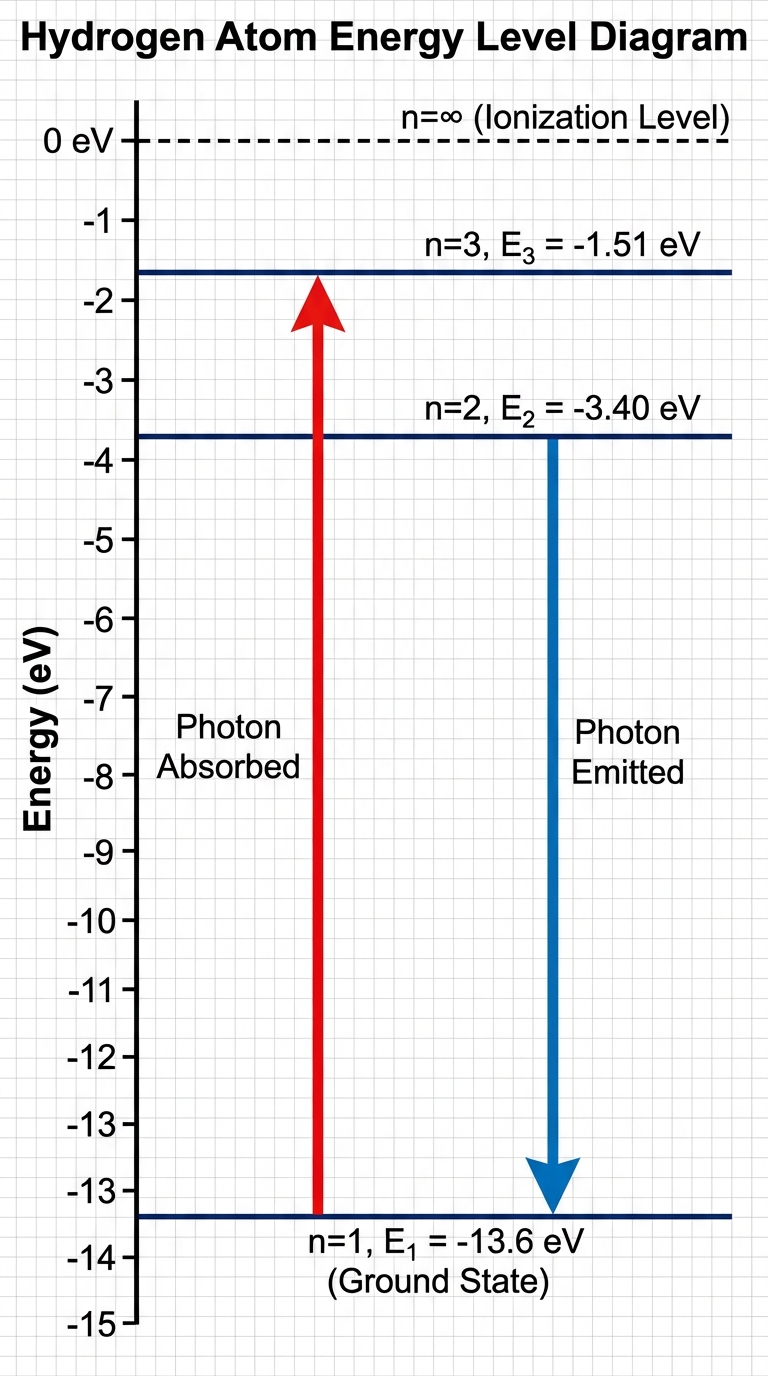
Atoms exist in discrete energy states. An electron cannot have an arbitrary amount of energy; it must occupy a specific energy level ($n$).
- Ground State: The lowest possible energy level ($n=1$). This is the most stable state.
- Excited States: Any energy level higher than the ground state ($n=2, 3, 4…$).
- Ionization Energy: The energy required to remove an electron completely from the atom (transition to $n=\infty$, where $E=0$).
Emission and Absorption of Photons
Electrons transition between energy levels by interacting with photons (particles of light). Since energy levels are discrete, the photons absorbed or emitted must have a specific energy equal exactly to the difference between the two levels.
1. Absorption: An electron absorbs a photon and jumps from a lower level to a higher level. The photon's energy must match the gap.
2. Emission: An electron falls from a higher level to a lower level, releasing a photon. The photon's energy equals the energy lost by the electron.
Key Formulas
The energy of a photon relates to its frequency ($f$) and wavelength ($\lambda$):
Where:
- $h$ is Planck's constant ($6.63 \times 10^{-34} \text{ J}\cdot\text{s}$ or $4.14 \times 10^{-15} \text{ eV}\cdot\text{s}$)
- $c$ is the speed of light ($3.0 \times 10^8 \text{ m/s}$)
The change in energy during a transition is:
Emission Spectra and Wave-Particle Duality
Because each element has a unique configuration of energy levels, each element emits a unique set of photon wavelengths. This acts as a "fingerprint" for the element.
- Continuous Spectrum: An uninterrupted band of colors (like a rainbow), usually from a hot, dense solid or gas.
- Emission Spectrum (Bright Line): Individual bright lines on a dark background. Produced by a hot, low-density gas.
- Absorption Spectrum (Dark Line): A continuous spectrum with dark lines missing. Produced when cool gas sits between a full-spectrum source and the observer.
Nuclear Physics and Radioactive Decay
While atomic physics deals with electrons, nuclear physics looks at the nucleons (protons and neutrons) inside the nucleus.
Nuclear Notation and Forces
Elements are denoted as $_Z^A\text{X}$, where:
- $A$ (Mass Number): Total number of protons + neutrons.
- $Z$ (Atomic Number): Number of protons (defines the element).
- $N$ (Neutron Number): $A - Z$.
The Strong Nuclear Force:
Protons are positively charged and repel each other via the electrostatic force. The nucleus stays together because of the Strong Nuclear Force.
- It is attractive and extremely strong.
- It is short-range (it only acts over distances of a few femtometers, roughly the size of a nucleus).
- It overcomes the electrostatic repulsion to bind nucleons together.
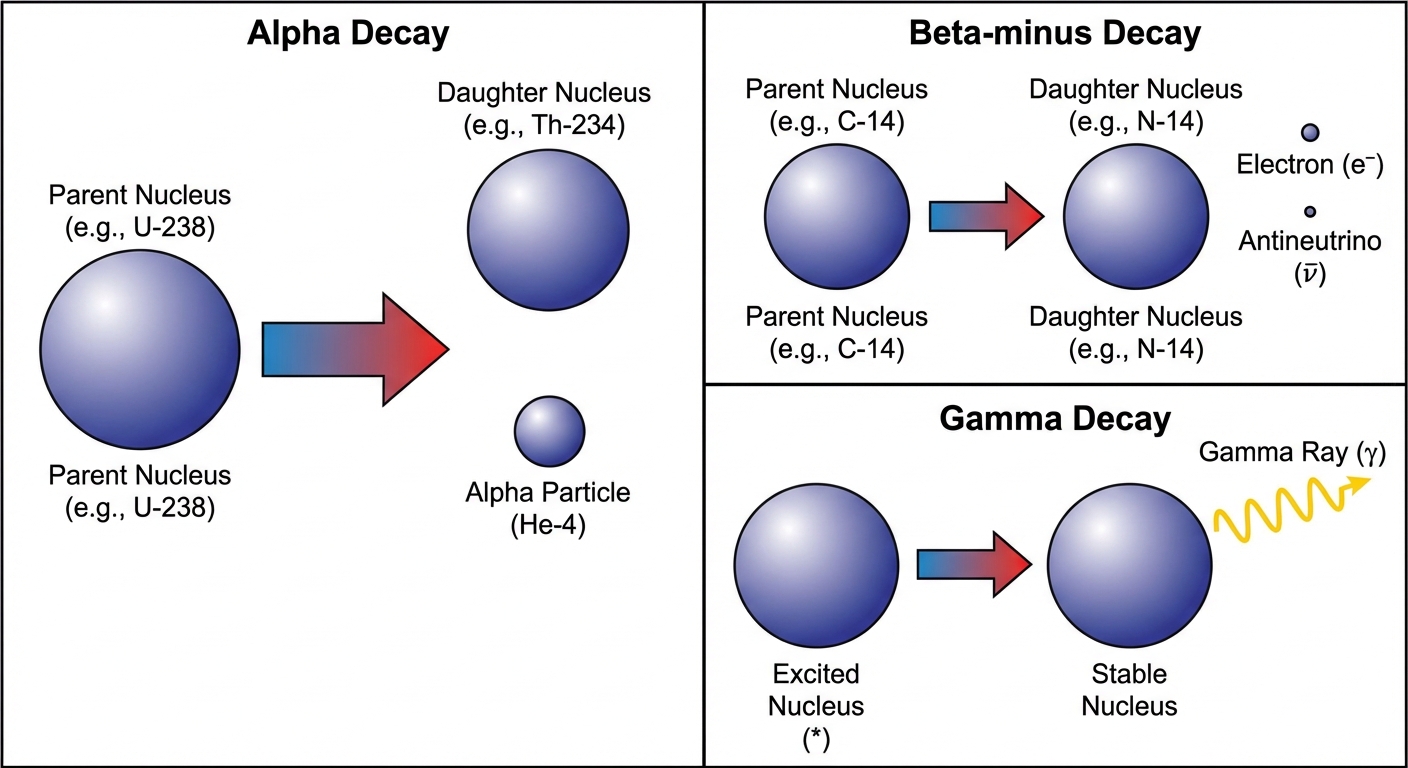
Radioactive Decay Modes
Unstable nuclei (parent nuclei) decay into more stable nuclei (daughter nuclei) by emitting particles or energy. In all nuclear equations, Charge (Z) and Mass Number (A) must be conserved.
| Decay Type | Particle Emitted | Symbol | Change to Parent ($A$) | Change to Parent ($Z$) | Description |
|---|---|---|---|---|---|
| Alpha Decay | Helium Nucleus | $\alpha$ or $_2^4\text{He}$ | Decreases by 4 | Decreases by 2 | Heavy, slow, low penetration. Occurs in heavy nuclei. |
| Beta-Minus | Electron | $\beta^-$ or $_{-1}^0\text{e}$ | Unchanged | Increases by 1 | A neutron acts as a proton + electron. Introduction of antineutrino ($\bar{\nu}$). |
| Beta-Plus | Positron | $\beta^+$ or $_{+1}^0\text{e}$ | Unchanged | Decreases by 1 | A proton acts as a neutron + positron. Introduction of neutrino ($\nu$). |
| Gamma Decay | High-energy Photon | $\gamma$ or $_0^0\gamma$ | Unchanged | Unchanged | Nucleus relaxes from excited state. High penetration. |
Worked Example: Beta-Minus Decay
Write the decay equation for Carbon-14 undergoing beta-minus decay.
Check conservation:
- Mass # top: $14 = 0 + 14$ (Correct)
- Atomic # bottom: $6 = -1 + 7$ (Correct)
Mass-Energy Equivalence
In classical physics, mass and energy are conserved separately. In modern physics, they are interchangeable quantities conserved as a single system.
The Universal Formula
This equation implies that mass is a concentrated form of energy. Even a tiny amount of mass corresponds to a massive amount of energy because $c^2$ is a huge number.
Mass Defect and Binding Energy
If you sum the individual masses of protons and neutrons in a nucleus, the total is greater than the actual mass of the bonded nucleus.
- Mass Defect ($ \Delta m $): The difference between the mass of the constituent nucleons and the actual nucleus mass.
- Binding Energy ($BE$): This "missing mass" has been converted into the energy required to hold the nucleus together (Strong Force potential energy).
Note on Units:
In nuclear physics, we often use atomic mass units ($u$) and electron-volts ($eV$).
- $1 u \approx 931.5 \text{ MeV}/c^2$
- To find Binding Energy in MeV when mass defect is in $u$, simply multiply by 931.5.
Binding Energy Per Nucleon
To compare stability across different elements, we look at the Binding Energy per nucleon ($BE/A$).
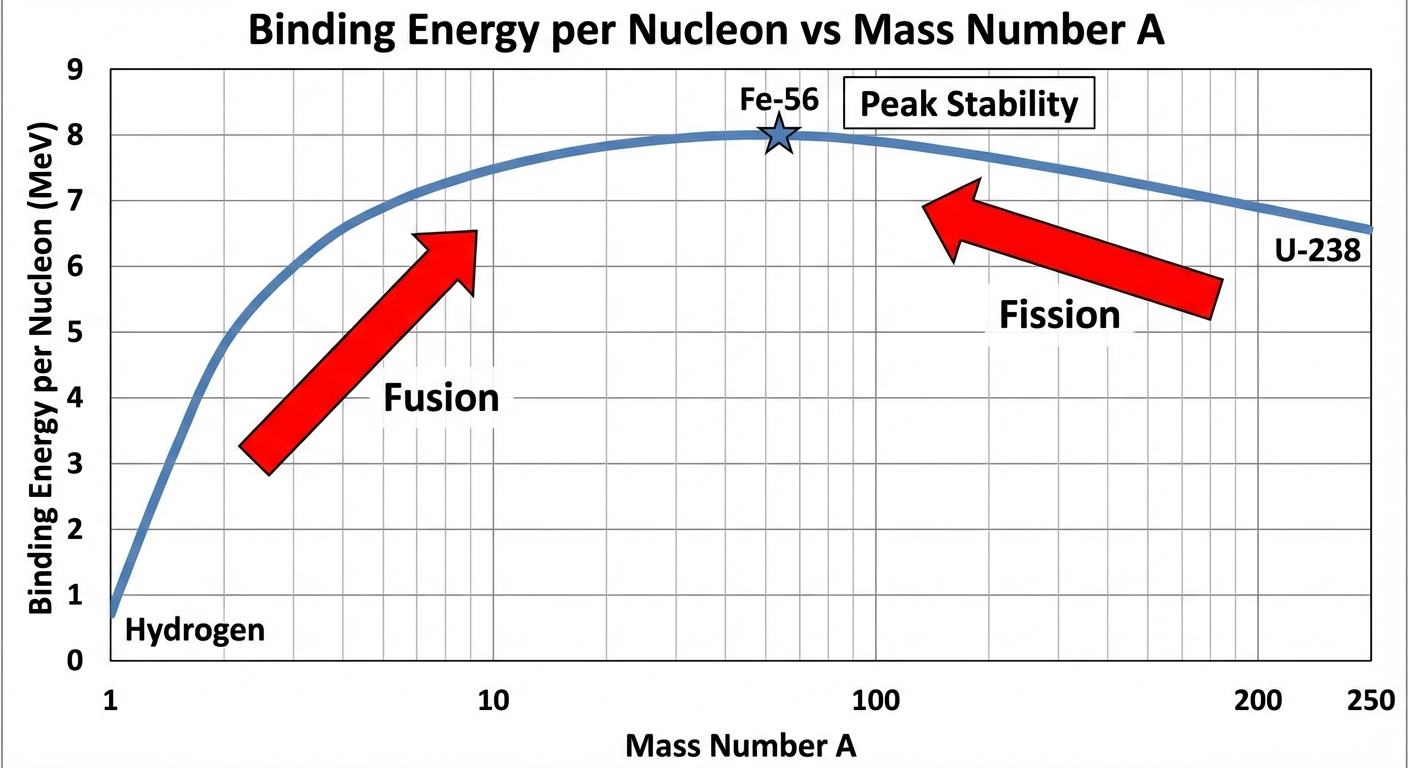
- Iron-56 ($^{56}\text{Fe}$) has the highest binding energy per nucleon and is the most stable element.
- Fission: Heavy nuclei (like Uranium) split into lighter ones, moving up the curve toward Iron, releasing energy.
- Fusion: Light nuclei (like Hydrogen) combine into heavier ones, moving up the curve toward Iron, releasing energy.
Common Mistakes & Pitfalls
- Confusing Nucleon Number ($A$) with Neutron Number ($N$): Remember $A = Protons + Neutrons$. $N$ is just neutrons. The top number in notation is $A$.
- Beta Decay Signs: Students often mix up $\beta^-$ (electron emission, $Z$ goes up) and $\beta^+$ (positron emission, $Z$ goes down).
- Mnemonic: Beta-Minus creates a More positive nucleus ($+1$ proton).
- Joules vs. eV: Always replace $c$ with $3\times 10^8$ only if working in kg/Joules. If you have mass in $u$, do not plug in $3\times 10^8$. Use the conversion factor $1u = 931.5 \text{ MeV}/c^2$.
- Negative Energy signs: In energy level problems, energy values are often negative (bound states). When calculating $\Delta E$, $\Delta E = E{final} - E{initial}$. Watch your double negatives! $E_{photon}$ is always positive.