Unit 7 Study Guide: Quantum Theory and Models
Here is a comprehensive review of the Quantum Physics section of AP Physics 2. This guide connects the breakdown of classical physics to the modern models of light and matter.
Photons and the Photoelectric Effect
For centuries, physicists debated whether light was a wave or a particle. While Young's Double Slit experiment (Unit 6) strongly supported the wave model, the Photoelectric Effect—observed in the late 19th and early 20th centuries—provided irrefutable evidence that light also behaves like a particle.
The Particle Model of Light: Photons
In the quantum model, light is not a continuous wave of energy but rather a stream of discrete energy packets called photons. The energy of a single photon is directly proportional to the frequency of the light.
Where:
- $E$ = Energy of the photon (Joules or Electron-Volts)
- $h$ = Planck's Constant ($6.63 \times 10^{-34} \text{ J}\cdot\text{s}$ or $4.14 \times 10^{-15} \text{ eV}\cdot\text{s}$)
- $f$ = Frequency of the light (Hz)
- $\lambda$ = Wavelength (m)
- $c$ = Speed of light ($3.0 \times 10^8 \text{ m/s}$)
Note on Units: In Quantum Physics, energy is often very small. We frequently use the electron-volt (eV) rather than Joules.
The Photoelectric Effect Experiment
The Photoelectric Effect occurs when electromagnetic radiation (light) strikes a metal surface and ejects electrons from that surface. These ejected electrons are often called photoelectrons.
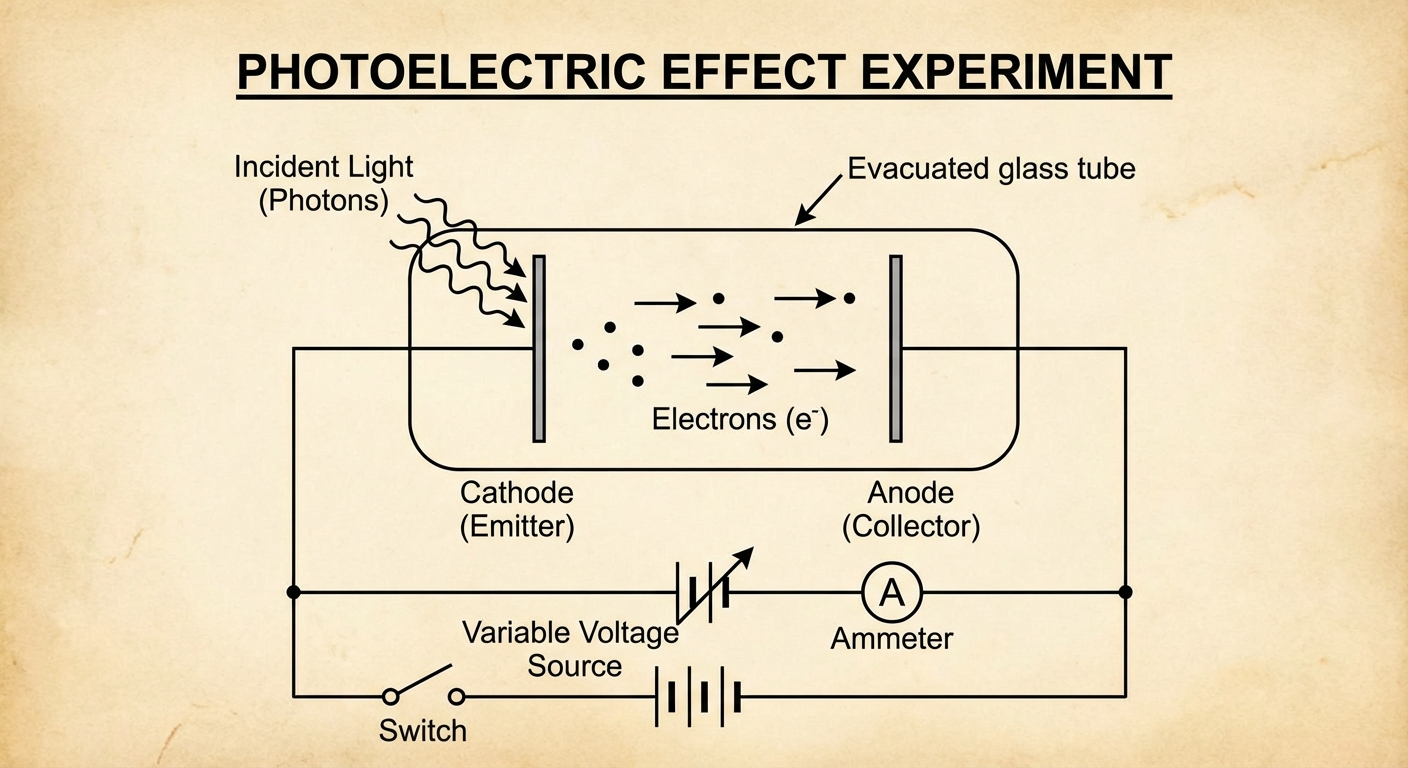
When light strikes the metal plate (cathode) in a vacuum tube:
- If the light has enough energy, electrons are ejected.
- These electrons travel to a collector plate (anode), creating a current.
- A retarding voltage (stopping potential) can be applied to stop these electrons, allowing us to measure their kinetic energy.
Einstein's Equation and the Work Function
Classical wave theory predicted that brighter light (higher intensity) would deliver energy faster, eventually ejecting electrons regardless of frequency. This was proven wrong. Experimental evidence showed that if the frequency is too low, no electrons are ejected, no matter how bright the light is.
Einstein explained this by conserving energy between a single photon and a single electron:
Where:
- $K{max}$ = Maximum Kinetic Energy of the ejected electron ($K{max} = \frac{1}{2}mv^2$)
- $hf$ = Energy of the incoming photon
- $\phi$ (Phi) = Work Function (The minimum energy required to unbind an electron from the specific metal)
Graphical Analysis
This is a very common AP Physics 2 exam topic. If you plot $K_{max}$ vs. Frequency ($f$):
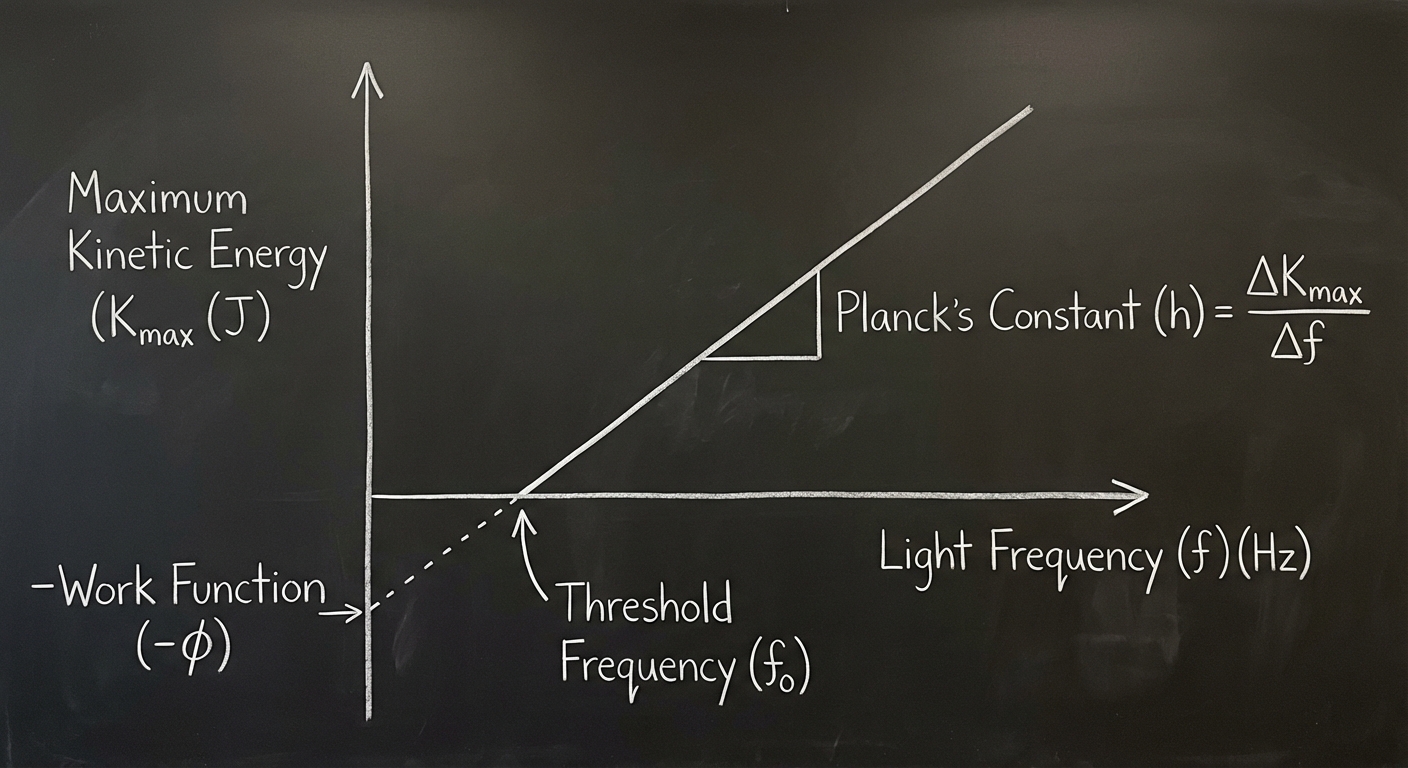
- Slope: Planck’s constant ($h$). The slope is universal for all metals.
- X-Intercept: The Threshold Frequency ($f_0$). Frequencies below this cannot eject electrons.
- Y-Intercept: The negative Work Function ($-\phi$).
Stopping Potential ($V_s$)
To measure the kinetic energy of the fastest electrons, we apply a reverse voltage until the current drops to zero. The work done by the electric field equals the kinetic energy of the electrons.
Thus, combined with Einstein's equation:
Comparison: Intensity vs. Frequency
It is vital to distinguish between the effects of changing the light's intensity (brightness) versus its frequency (color).
| Change Made | Effect on Current (Rate of Ejection) | Effect on $K_{max}$ (Speed of Electrons) |
|---|---|---|
| Increase Intensity (Brighter Light) | Increases (More photons hit the metal $\rightarrow$ more electrons ejected) | No Change (Each photon still has same energy) |
| Increase Frequency (Higher Energy Color) | No Change (Assuming constant photon count) | Increases (Excess energy becomes KE) |
Wave-Particle Duality
Understanding light requires a dual model. Light exhibits properties of both waves and particles, depending on the experiment performed.
Wave Nature Evidence:
- Interference: Young's Double Slit experiment creates fringes of constructive and destructive interference.
- Diffraction: Light bends around corners and spreads through openings.
- Polarization: Light waves can be filtered based on orientation.
Particle Nature Evidence:
- Photoelectric Effect: Energy is transferred in discrete packets; instantaneous ejection of electrons.
- Compton Scattering: Photons collide with electrons and scatter like billiard balls, conserving momentum and energy.
Conceptual Summary: Light travels as a wave (probability wave) but interacts with matter as a particle.
de Broglie Wavelength
If light (which we thought was a wave) can act like a particle, can matter (which we think of as particles) act like a wave? Louis de Broglie proposed that all matter has a wavelength associated with its momentum.
The de Broglie Hypothesis
The wavelength of a particle is inversely proportional to its momentum:
Where:
- $\lambda$ = de Broglie wavelength (m)
- $h$ = Planck's Constant
- $p$ = Momentum ($kg \cdot m/s$)
- $m$ = Mass ($kg$)
- $v$ = Velocity ($m/s$)
Macroscopic vs. Microscopic
Why don't you diffract when you walk through a doorway?
- Macroscopic Objects (Baseballs, Humans): $m$ is large, so $p$ is large. Since $h$ is tiny ($10^{-34}$), the wavelength $\lambda$ becomes infinitesimally small (smaller than an atomic nucleus). Wave effects are unobservable.
- Microscopic Objects (Electrons, Protons): $m$ is tiny. The wavelength $\lambda$ can be comparable to atomic spacing ($10^{-10}$ m). Therefore, electrons can diffract and interfere.
Real-World Application: Electron Microscope
Because electrons can differ in speed (and therefore wavelength) based on voltage, we can accelerate electrons to have much smaller wavelengths than visible light. This helps electron microscopes achieve much higher resolution than optical microscopes, allowing us to image viruses and crystal lattices.
Worked Example: Electron Wavelength
Problem: An electron ($m_e = 9.11 \times 10^{-31}$ kg) moves with a velocity of $2.0 \times 10^6$ m/s. Calculate its de Broglie wavelength.
Solution:
- Identify variables: $m = 9.11 \times 10^{-31}$ kg, $v = 2.0 \times 10^6$ m/s.
- Calculate momentum:
- Apply de Broglie equation:
This wavelength is in the X-ray range, meaning this electron would diffract if it passed through a crystal lattice.
Common Mistakes & Pitfalls
Confusing Intensity and Frequency:
- Mistake: Thinking that increasing the brightness (intensity) of light increases the energy of the electrons.
- Correction: Brightness only increases the number of photons (and thus the current). Only Frequency determines the energy per electron.
Mixing Units (eV and Joules):
- Mistake: Using $h = 6.63 \times 10^{-34}$ (J$\cdot$s) but plugging in energy in eV, or vice versa.
- Correction: Always checking your units. If the problem gives energy in eV, either convert to Joules ($1 \text{ eV} = 1.6 \times 10^{-19} \text{ J}$) or use the eV version of Planck's constant ($4.14 \times 10^{-15} \text{ eV}\cdot\text{s}$).
Work Function Logic:
- Mistake: Thinking the work function depends on the light source.
- Correction: The work function ($\phi$) is a property of the metal, not the light. Changing the light bulb does not change the amount of energy required to escape the metal surface.
$K_{max}$ vs. Actual K:
- Mistake: Assuming all ejected electrons have $K_{max}$.
- Correction: $K{max}$ represents the electrons that come from the very surface and lose the least energy escaping. Electrons deeper in the metal lose more energy on the way out and have $K < K{max}$.