Mastering Thermodynamics: Energy, Gases, and Heat
Thermodynamic Systems
Thermodynamics is the study of energy transfers involving thermal energy and heat. To analyze these transfers effectively, we must first define the "universe" of our problem.
Defining the System and Surroundings
In physics, we arbitrarily define a region of space or a collection of matter to study.
- System: The specific object or collection of objects being analyzed (e.g., the gas inside a piston).
- Surroundings: Everything outside the system with which the system can exchange energy or mass.
- Boundary: The conceptual or physical wall separating the system from the surroundings.
Types of Systems
Classifying the system dictates which conservation laws apply.
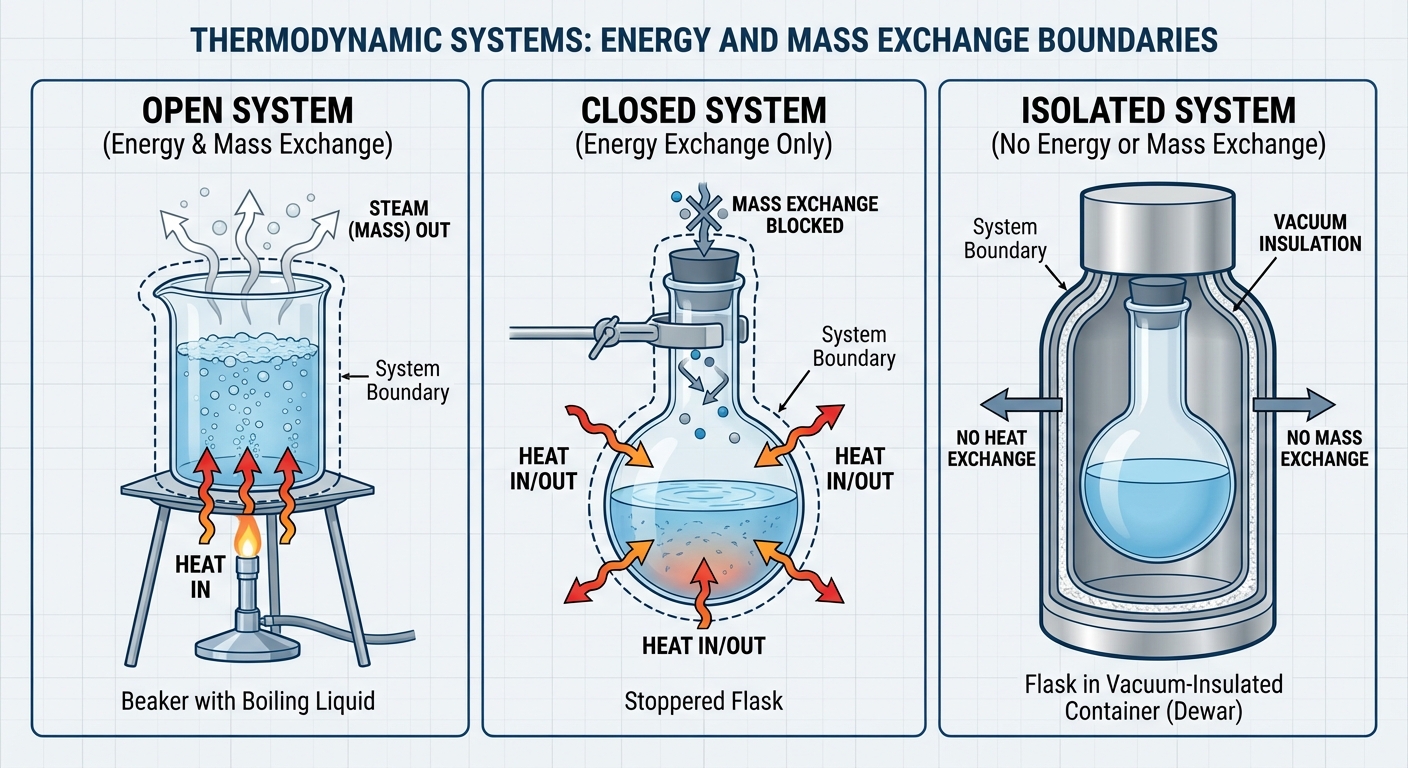
| System Type | Can Exchange Energy? | Can Exchange Matter? | Example |
|---|---|---|---|
| Open System | Yes | Yes | A boiling pot of water without a lid (steam escapes). |
| Closed System | Yes | No | A sealed balloon (heat enters/exits, air stays inside). |
| Isolated System | No | No | A perfectly insulated thermos (idealized concept). |
Key Concept: In AP Physics 2, we most frequently deal with Closed Systems containing an ideal gas, where mass ($n$) remains constant, but heat ($Q$) and work ($W$) can cross the boundary.
Pressure, Thermal Equilibrium, and Ideal Gas Law
Macroscopic Properties
Before looking at molecules, we look at the observable properties of a gas: Pressure ($P$), Volume ($V$), Amount of substance ($n$), and Temperature ($T$).
- Pressure ($P$): The force exerted perpendicular to a surface per unit area.
In a gas, this is caused by the collisions of molecules against the container walls. - Temperature ($T$): A scalar quantity that determines the direction of heat flow. Heat naturally flows from high $T$ to low $T$.
The Zeroth Law of Thermodynamics
This law provides the definition of thermal equilibrium. It states: If Object A is in thermal equilibrium with Object B, and Object B is in thermal equilibrium with Object C, then Object A and Object C are in thermal equilibrium with each other.
- Thermal Equilibrium: A state where two objects in contact no longer exchange net heat energy. This occurs when they reach the same temperature.
The Ideal Gas Law
The state of an ideal gas is determined by the relationship between its macroscopic variables. There are two standard forms of the Ideal Gas Law you need to know:
1. The Moles Form (Chemistry standard):
- $P$: Pressure (Pa)
- $V$: Volume ($m^3$)
- $n$: Number of moles
- $R$: Universal Gas Constant ($8.31 \, J/(mol\cdot K)$)
- $T$: Temperature (Must be in Kelvin)
2. The Molecules Form (Physics standard):
- $N$: Number of individual molecules or atoms
- $k_B$: Boltzmann’s Constant ($1.38 \times 10^{-23} \, J/K$)
Note: $R = NA \cdot kB$, where $N_A$ is Avogadro's number.
Kinetic Theory and Thermodynamic Models
Measurements like Pressure and Temperature are just macroscopic averages of chaos happening at the microscopic level. Kinetic Theory bridges the gap between the micro and macro worlds.
Assumptions of Ideal Gases
An ideal gas is a theoretical model that real gases approximate well at high temperatures and low pressures.
- Particles are point masses (volume of particles is negligible).
- Particles move in random, straight-line motion.
- Collisions with walls and other particles are perfectly elastic (kinetic energy is conserved).
- There are no intermolecular forces (attraction/repulsion) between particles.
Temperature and Kinetic Energy
The most critical concept in this unit is the definition of temperature (Kelvin):
Temperature is directly proportional to the average translational kinetic energy ($K_{avg}$) of the molecules.
- $v_{rms}$: Root-mean-square speed (a statistical average speed).
- $m$: Mass of a single molecule.
Crucial Relationships:
- If you double the absolute temperature ($T$), the average kinetic energy doubles.
- Since $K \propto v^2$, if you quadruple the temperature, the speed ($v_{rms}$) only doubles.
Maxwell-Boltzmann Distribution
Gas molecules do not all move at the same speed. They follow a probability distribution.
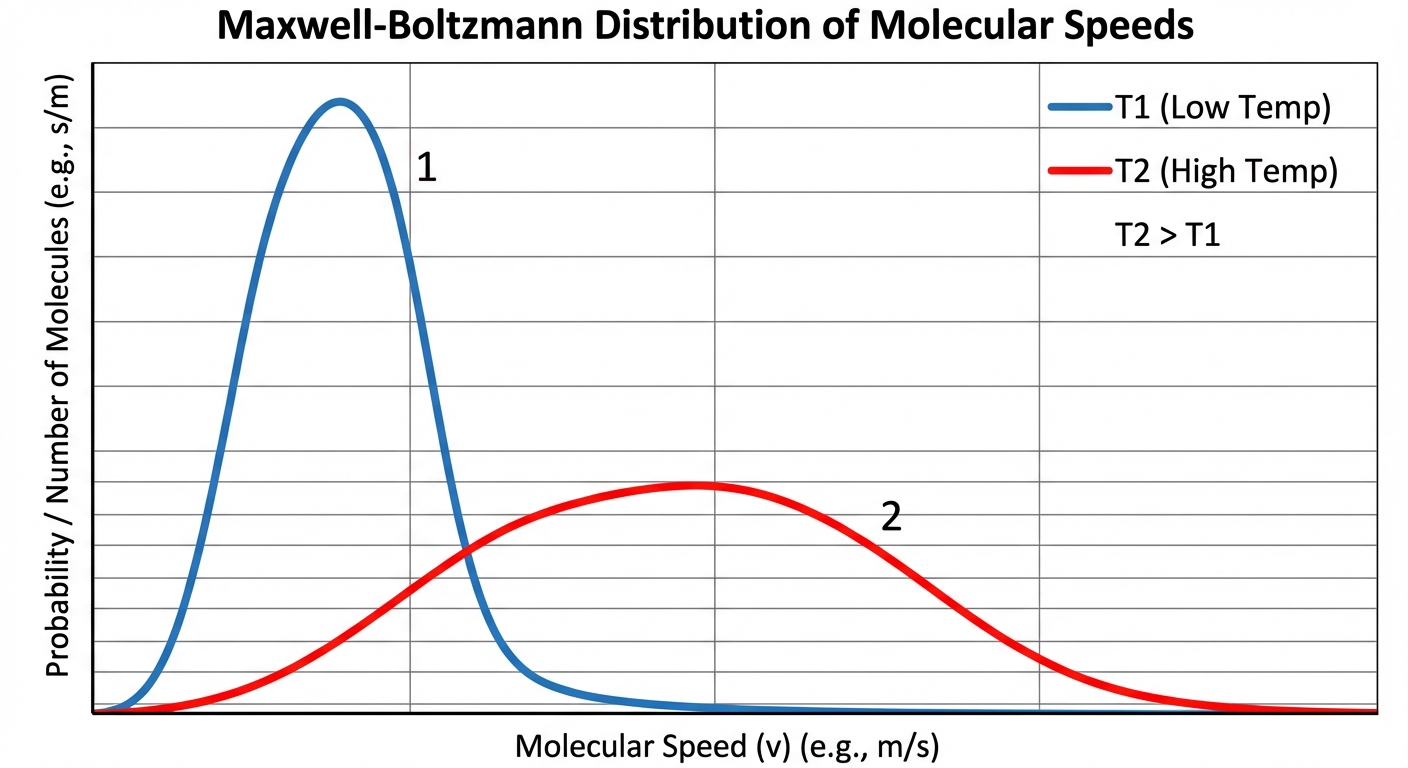
Reading the Graph:
- X-axis: Particle Speed.
- Y-axis: Number of particles (or probability).
- Peak: The most probable speed.
- Effect of Temperature: As $T$ increases, the curve flattens and shifts to the right (higher average speed, wider range of speeds).
- Effect of Mass: Heavier gases (at the same $T$) move slower. Their curve is taller and shifted to the left.
Internal Energy ($U$)
For a monoatomic ideal gas, internal energy is the sum of all kinetic energies of the particles.
Heat and Temperature
It is vital to distinguish between describing the state of an object versus the transfer of energy.
- Internal Energy ($U$): The total energy stored inside the system (State function).
- Temperature ($T$): The average kinetic energy per molecule (State function).
- Heat ($Q$): The transfer of thermal energy between systems due to a temperature difference (Process function).
Heat Transfer Mechanisms
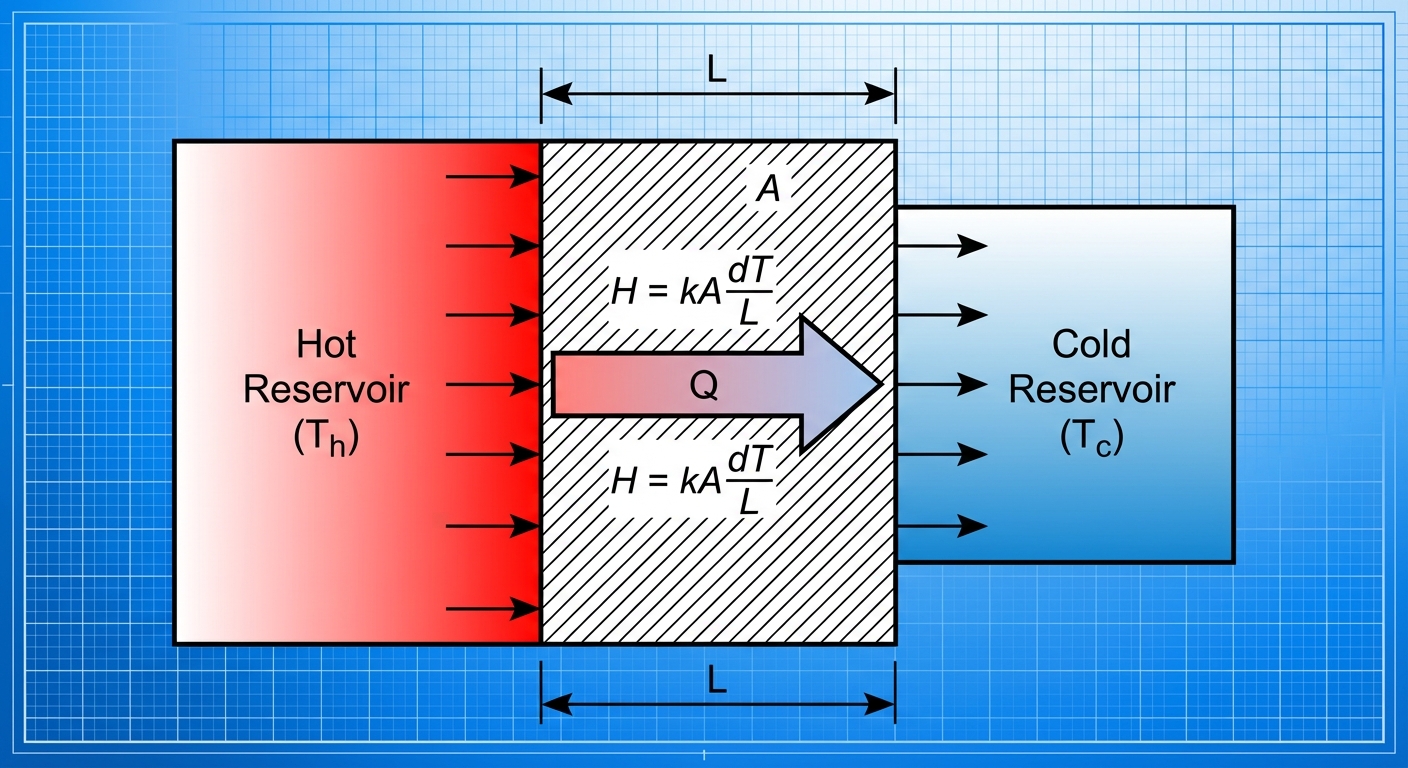
1. Conduction
Transfer of heat through stationary matter by physical contact (collisions of atoms).
- Thermal Conductivity Equation:
- $\frac{Q}{\Delta t}$: Rate of heat transfer (Watts or J/s).
- $k$: Thermal conductivity of the material (property of the substance).
- $A$: Cross-sectional area.
- $L$: Length or thickness of the barrier.
- $\Delta T$: Temperature difference across the barrier.
2. Convection
Transfer of heat by the macroscopic movement of a fluid (gas or liquid).
- Mechanism: Warmer fluid expands, becomes less dense, and rises; cooler fluid sinks.
3. Radiation
Transfer of energy via electromagnetic waves. No medium is required.
- Dark/rough objects absorb and emit radiation better than light/shiny objects.
Specific Heat and Latent Heat
Changing Temperature:
When adding heat results in a temperature rise:
- $c$: Specific heat capacity (Energy required to raise 1kg of substance by 1K).
Changing Phase:
When adding heat results in a phase change (Melting/Boiling), temperature does not change.
- $L$: Latent heat (of fusion or vaporization).
Common Mistakes & Pitfalls
- Celsius vs. Kelvin: ALWAYS convert to Kelvin involved in the Ideal Gas Law ($PV=nRT$) or Thermodynamic calculations.
- Correction: $T{Kelvin} = T{Celsius} + 273$.
- Heat vs. Temperature: Students often say "This object has a lot of heat." Objects cannot "have" heat; they have Internal Energy. Heat is the transfer flow.
- Mole vs. Molecule: Confusing $n$ (moles) with $N$ (number of particles). $N$ is a huge number ($10^{23}$ range); $n$ is usually small ($<10$).
- Pressure is Force: Pressure is not Force. It is Force on an Area. If you compress a gas into a smaller volume, Pressure increases because the frequency of collisions increases, not necessarily because individual particles hit harder (unless T also rises).
- Interpretation of $v_{rms}$: Heavy molecules and light molecules at the same temperature have the same average Kinetic Energy, but the light molecules move much faster.