Unit 4: Quantitative Analysis and Titration
Stoichiometry Fundamentals
Stoichiometry is the study of the quantitative relationships between the amounts of reactants used and amounts of products formed by a chemical reaction. It rests on the Law of Conservation of Mass—matter is neither created nor destroyed, meaning atoms must be balanced on both sides of the equation.
The Mole Ratio
The core of all stoichiometric calculations is the mole ratio. This ratio is derived from the coefficients of a balanced chemical equation.
In this generic equation, the mole ratio of $A$ to $C$ is $a:c$. To convert between substances, you must always pass through the "mole bridge."
General Flow of Calculation:
- Mass/Volume/Particles of Known $\rightarrow$ Convert to Moles
- Moles of Known $\times$ (Mole Ratio) $\rightarrow$ Moles of Unknown
- Moles of Unknown $\rightarrow$ Convert to Mass/Volume/Particles
Limiting and Excess Reactants
In the real world, reactants are rarely present in exact stoichiometric proportions. One reactant will be consumed first, stopping the reaction.
- Limiting Reactant (Reagent): The substance that is totally consumed when the chemical reaction is complete. It determines the maximum amount of product that can be formed.
- Excess Reactant: The reactant that remains after the limiting reactant is used up.
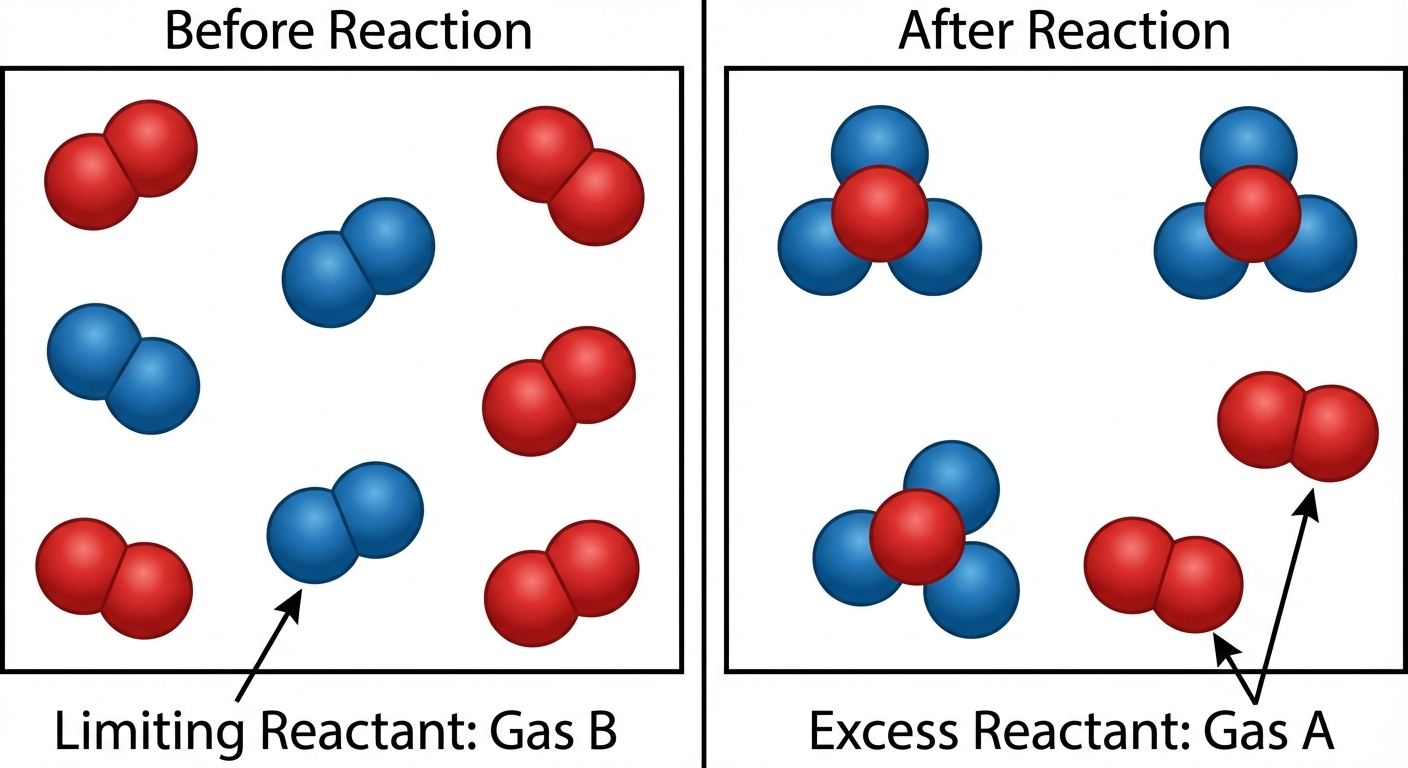
Strategy to Find the Limiting Reactant
Do not guess based on starting mass. A lower mass does not necessarily mean it is the limiting reactant because molar masses differ.
- Calculate the moles of product that each reactant could theoretically produce.
- The reactant that produces the least amount of product is the limiting reactant.
- The amount of product calculated from the limiting reactant is the Theoretical Yield.
Reaction Yields
- Theoretical Yield: The maximum amount of product that can be generated from a given amount of reactant (calculated via stoichiometry).
- Actual Yield: The amount of product actually produced when the experiment is performed in the lab.
- Percent Yield: An efficiency metric for the reaction.
Worked Example: Stoichiometry & Limiting Reactant
Problem: Consider the reaction between aluminum and chlorine gas to form aluminum chloride:
If 5.00 g of Al reacts with 15.00 g of $\text{Cl}2$, determine the theoretical yield of $\text{AlCl}3$ in grams.
Solution:
Convert Mass to Moles:
- Moles Al: $5.00 \text{ g} / 26.98 \text{ g/mol} = 0.185 \text{ mol Al}$
- Moles $\text{Cl}2$: $15.00 \text{ g} / 70.90 \text{ g/mol} = 0.211 \text{ mol Cl}2$
Test for Limiting Reactant (Calculate Product for Both):
- From Al: $0.185 \text{ mol Al} \times \frac{2 \text{ mol AlCl}3}{2 \text{ mol Al}} = 0.185 \text{ mol AlCl}3$
- From $\text{Cl}2$: $0.211 \text{ mol Cl}2 \times \frac{2 \text{ mol AlCl}3}{3 \text{ mol Cl}2} = 0.141 \text{ mol AlCl}_3$
Identify Limiting Reactant:
- $0.141 < 0.185$, therefore $\text{Cl}_2$ is the limiting reactant.
Calculate Theoretical Mass:
- $0.141 \text{ mol AlCl}3 \times 133.34 \text{ g/mol} = 18.80 \text{ g AlCl}3$
Introduction to Titration
Titration is a quantitative analytical method used to determine the concentration of an identified analyte (a substance to be analyzed) by reacting it with a solution of known concentration (the standard solution or titrant).
Experimental Setup and Terminology
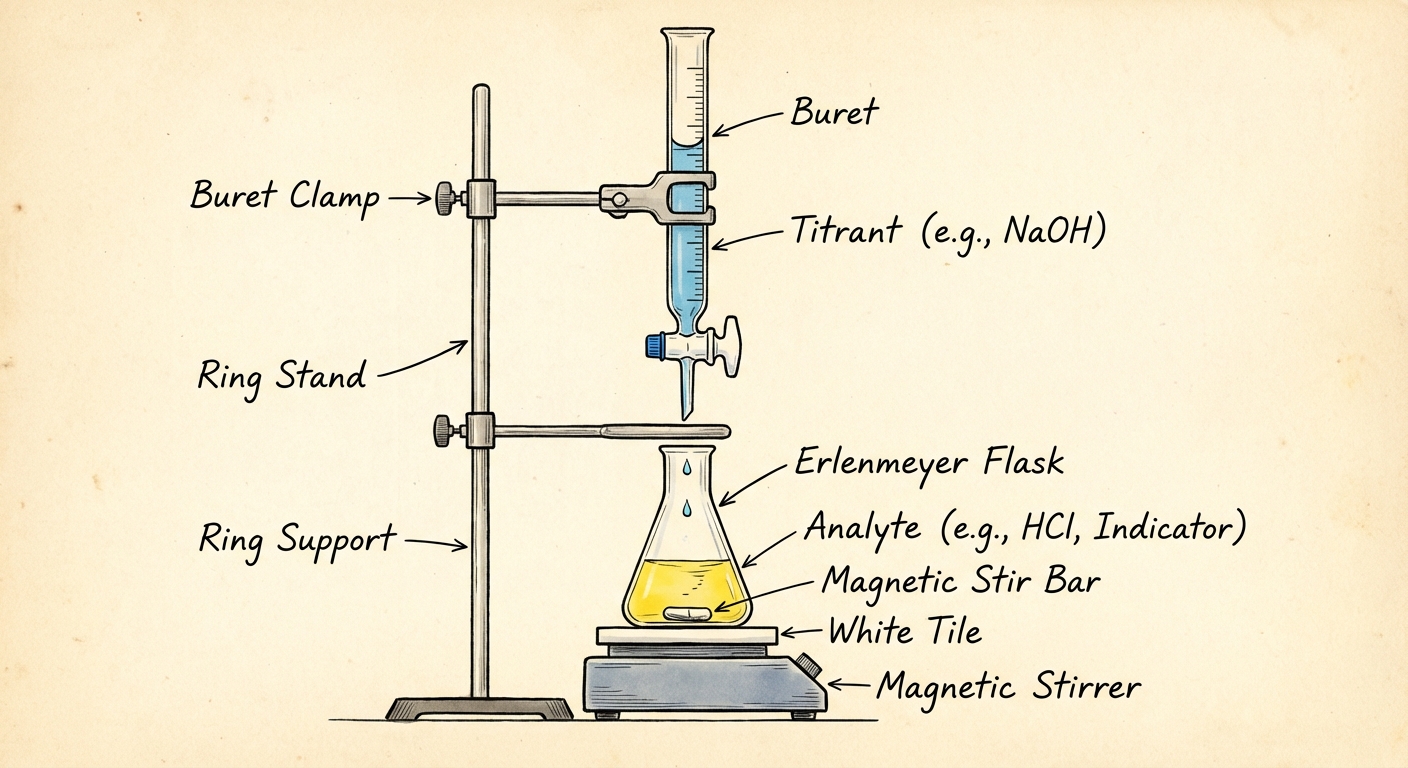
| Term | Definition |
|---|---|
| Titrant | The solution of known concentration (usually in the buret). |
| Analyte | The solution of unknown concentration (usually in the flask). |
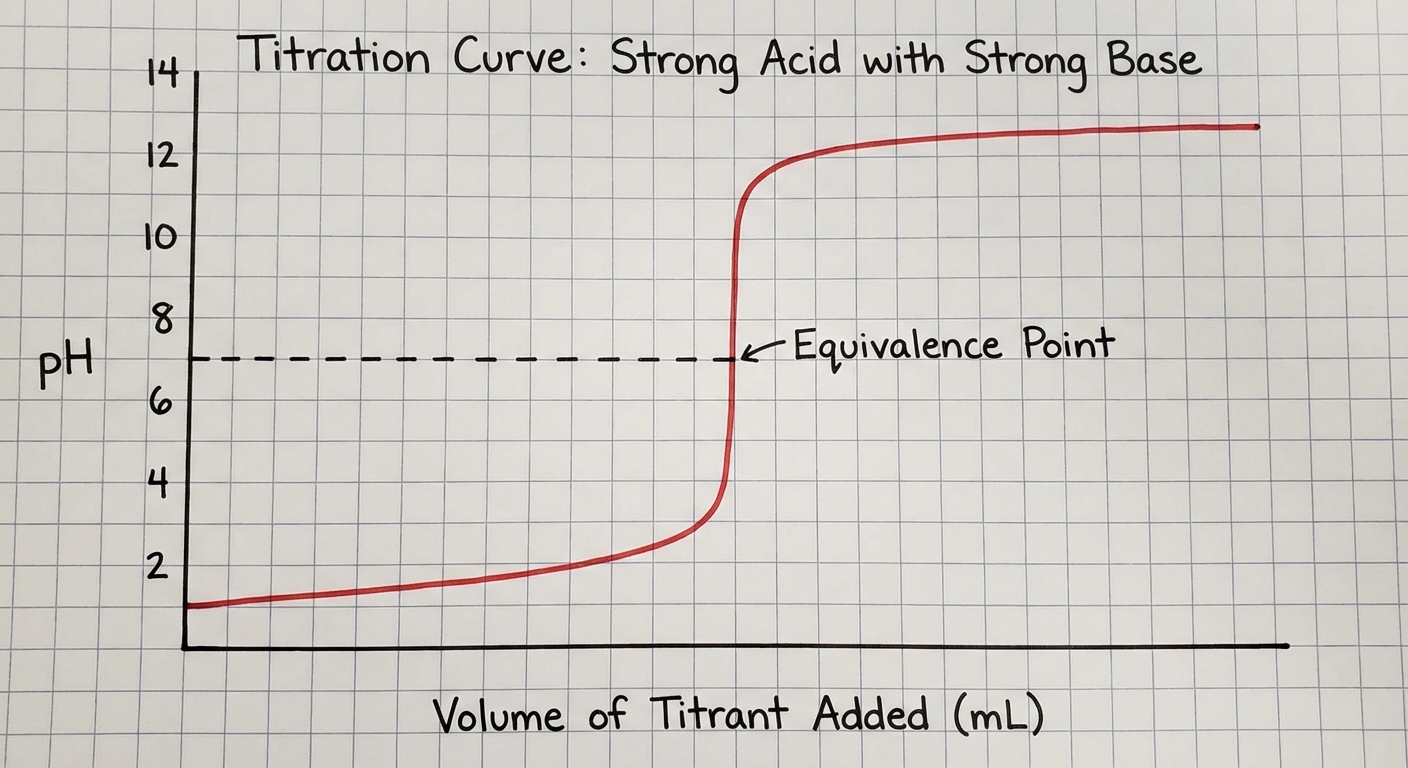
| Equivalence Point | The point where the number of moles of titrant added is stoichiometrically equal to the number of moles of analyte originally present. |
| Endpoint | The point where the indicator changes color. Ideally, the endpoint should occur extremely close to the equivalence point. |
| Standardization | The process of determining the exact concentration of a solution by titrating it against a primary standard. |
The Mathematics of Titration
Calculations for titrations are essentially stoichiometry problems involving solution volume and molarity.
Key Formula:
Where $M$ is Molarity (mol/L), $n$ is moles, and $V$ is volume (L).
The Danger of $M1V1 = M2V2$
Students frequently memorize the dilution formula ($M1V1 = M2V2$) or the acid-base shortcut ($MaVa = MbVb$). These formulas are only valid if the mole ratio is 1:1.
For a reaction like $2\text{HCl} + \text{Ba(OH)}2 \rightarrow \text{BaCl}2 + 2\text{H}_2\text{O}$, the ratio is not 1:1. It is safer to use dimensional analysis or the modified formula:
(Where $na$ and $nb$ are the coefficients from the balanced equation).
Types of Titrations in Unit 4
While Acid-Base is the most common, titrations can apply to any reaction where species react in fixed ratios.
- Acid-Base Titrations: Net ionic equation often involves $H^+(aq) + OH^-(aq) \rightarrow H_2O(l)$.
- Redox Titrations: Uses an oxidizing agent and a reducing agent. The equivalence point is reached when electrons lost equal electrons gained (based on the balanced redox equation). An indicator (or the color of the reactant itself, like Permanganate) signals the end.
Example: Titration Calculation
Problem: A student titrates 25.0 mL of an unknown $\text{H}2\text{SO}4$ solution with 0.100 M $\text{NaOH}$. The equivalence point is reached when 34.5 mL of $\text{NaOH}$ is added.
Calculate the molarity of the sulfuric acid.
Solution:
- Calculate moles of Titrant (NaOH):
- Use Mole Ratio to find moles of Analyte ($\text{H}2\text{SO}4$):
- Calculate Molarity:
Common Mistakes & Pitfalls
Confusing Equivalence Point vs. Endpoint:
- Correction: The equivalence point is a theoretical chemical state (moles match stoichiometry). The endpoint is a physical observation (color change). If an indicator is chosen poorly, these two points will not match.
Blindly applying $MaVa = MbVb$:
- Correction: Always write the balanced chemical equation first. If the coefficients are not 1:1, this formula will yield the wrong answer. Use dimensional analysis (moles $\rightarrow$ mole ratio $\rightarrow$ concentration).
Buret Cleaning Errors:
- Correction: A common lab question asks what happens if the buret is rinsed with water but not the titrant. This dilutes the titrant, meaning you will need more volume to reach the equivalence point, leading to an overestimation of the analyte's concentration.
Forgetting to convert Milliliters to Liters:
- Correction: Molarity is defined as moles per Liter. While volumes sometimes cancel out in ratio equations, it is best practice to convert to Liters immediately to avoid order-of-magnitude errors.