Comprehensive Guide to Thermodynamics and Fluids
Kinetic Theory and Ideal Gases
Thermodynamics in AP Physics 2 begins at the microscopic level. Understanding how individual particles behave allows us to predict the macroscopic properties of a system, such as pressure, temperature, and volume.
The Ideal Gas Model
An Ideal Gas is a theoretical gas composed of many randomly moving point particles that are not subject to interparticle interactions. The ideal gas law relates the macroscopic state variables of a system.
Where:
- $P$ = Pressure (Pa)
- $V$ = Volume ($m^3$)
- $n$ = Number of moles
- $R$ = Universal gas constant ($8.31 \, J/(mol\cdot K)$)
- $T$ = Temperature (Must be in Kelvin)
- $N$ = Number of molecules
- $k_B$ = Boltzmann’s constant ($1.38 \times 10^{-23} \, J/K$)
Kinetic Molecular Theory (KMT)
KMT explains macroscopic properties based on the motion of molecules. The core assumptions are:
- Collisions between molecules and walls are perfectly elastic (kinetic energy is conserved).
- Molecules move in random directions.
- Intermolecular forces are negligible.
Temperature is a measure of the average translational kinetic energy per molecule. It is not a measure of heat.
From this, we see that if you double the absolute temperature (Kelvin), you double the average kinetic energy. However, the root-mean-square velocity ($v_{rms}$) only increases by a factor of $\sqrt{2}$.
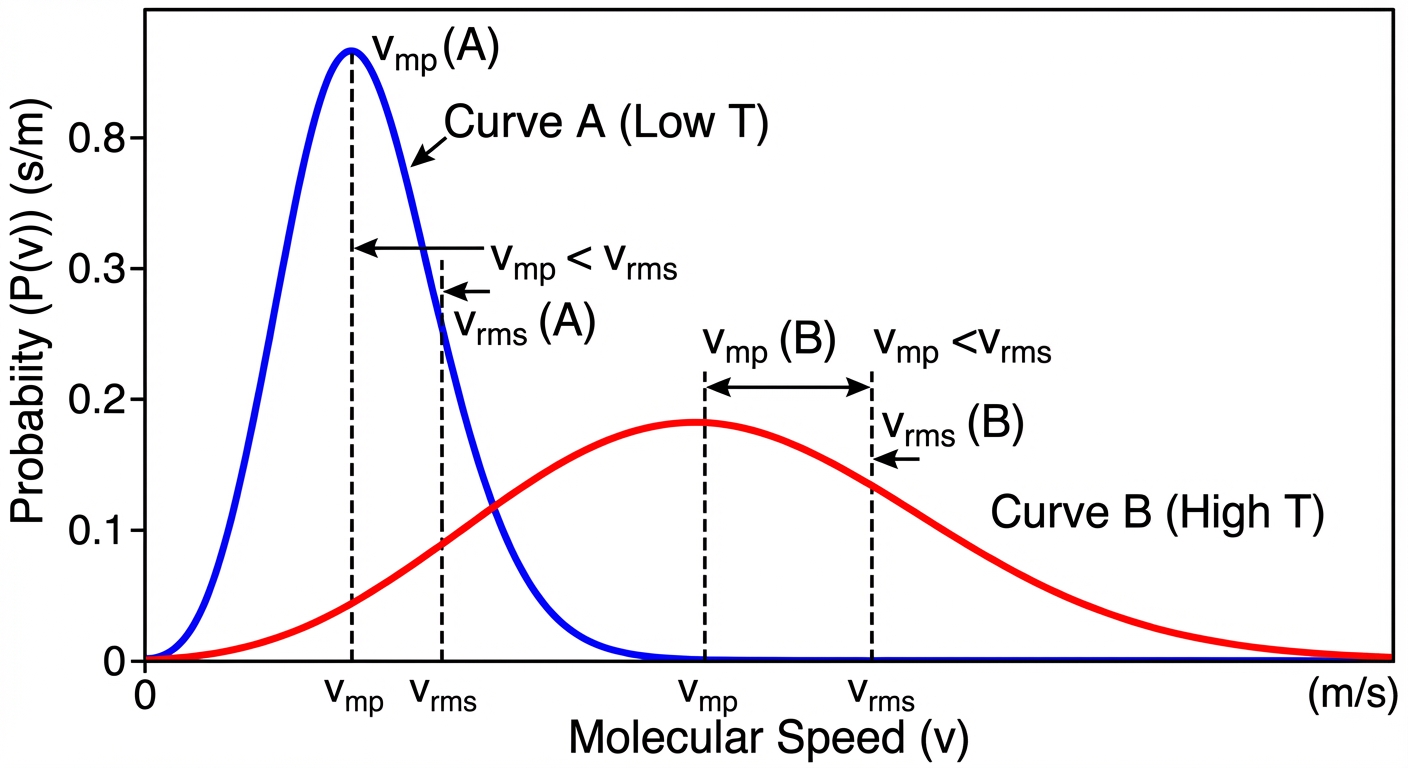
Key Takeaways from the Maxwell-Boltzmann Distribution:
- The peak represents the most probable speed.
- As temperature increases, the curve flattens and shifts to the right (higher speeds).
- Heavier molecules move slower than lighter molecules at the same temperature.
Heat Transfer and Thermal Equilibrium
Zeroth Law of Thermodynamics
If System A is in thermal equilibrium with System B, and System B is in equilibrium with System C, then System A and C are in equilibrium. Thermal Equilibrium means no net heat flows between objects because they are at the same temperature.
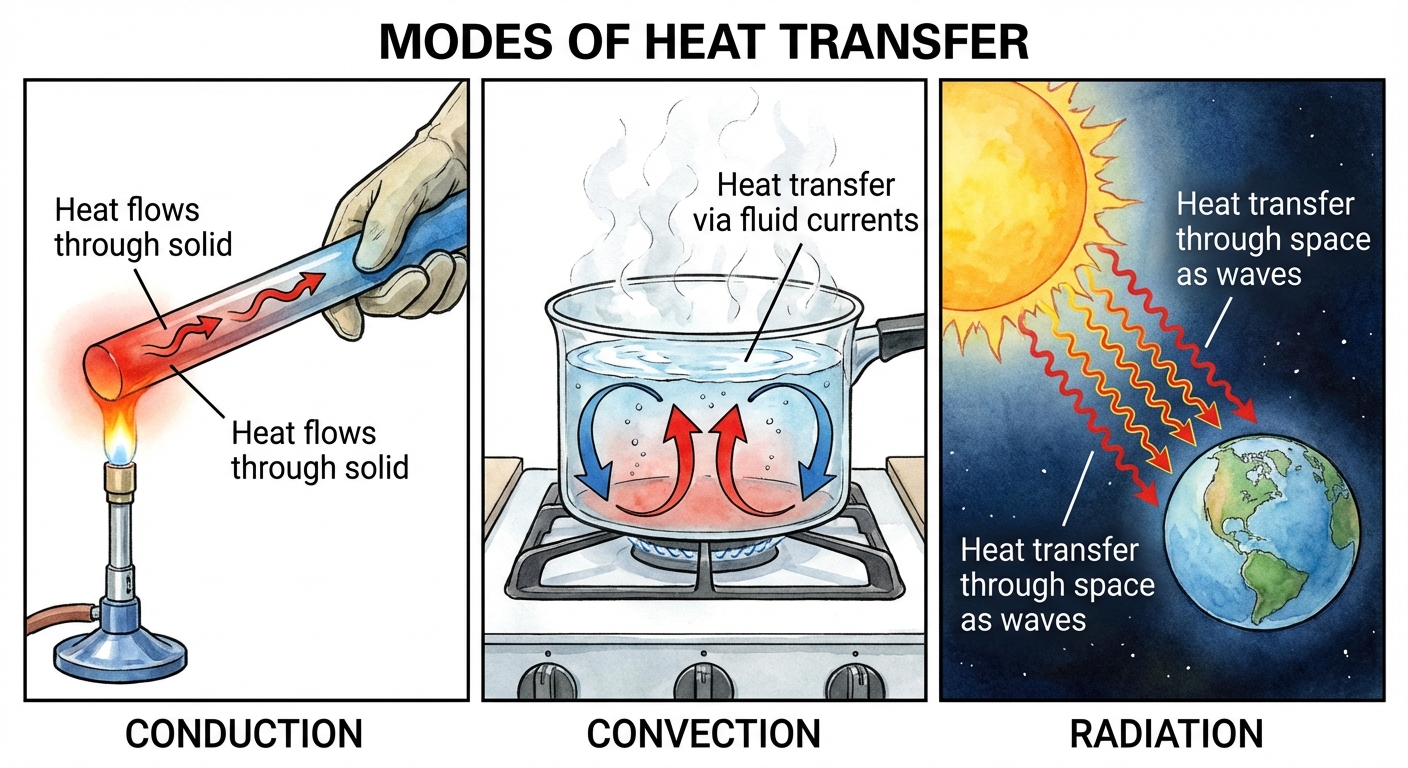
Mechanisms of Heat Transfer
Heat ($Q$) is the transfer of energy due to a temperature difference. It flows spontaneously from hot to cold.
- Conduction: Transfer through direct contact via molecular collisions. Solids are best at this.
- Convection: Transfer via the bulk movement of fluid (liquid or gas). Hot fluid rises (less dense), cold fluid sinks.
- Radiation: Transfer via electromagnetic waves. No medium is required.
Thermal Conductivity Equation
The rate of heat transfer through a material via conduction is given by:
Where:
- $k$ = Thermal conductivity of the material ($W/m\cdot K$)
- $A$ = Cross-sectional area ($m^2$)
- $\Delta T$ = Temperature difference ($K$ or $^\circ C$)
- $L$ = Thickness or length of the material ($m$)
Note: To insulate a house, you want a material with a low $k$ value, a large thickness $L$, or a small surface area $A$.
The First Law of Thermodynamics
The First Law is essentially the Law of Conservation of Energy applied to thermodynamic systems.
The Equation
Where:
- $\Delta U$ = Change in Internal Energy (Joules)
- $Q$ = Heat added to the system (Joules)
- $W$ = Work done on the system (Joules)
Internal Energy ($U$)
For an ideal gas, internal energy depends only on temperature. If $T$ is constant, $\Delta U = 0$.
Sign Conventions (CRITICAL)
This is the most common area for student errors. In AP Physics 2, we define the system as the gas.
| Variable | Positive (+) | Negative (-) |
|---|---|---|
| Q (Heat) | Heat added to the system | Heat removed from the system |
| W (Work) | Work done ON the gas (Compression) | Work done BY the gas (Expansion) |
| $\Delta U$ | Internal energy increases ($T$ goes up) | Internal energy decreases ($T$ goes down) |
Work Done on a Gas
Work is related to volume change. If volume doesn't change, no work is done.
- Compression: $\Delta V$ is negative $\rightarrow$ $W$ is positive (Energy added).
- Expansion: $\Delta V$ is positive $\rightarrow$ $W$ is negative (Energy leaves).
Thermodynamic Processes and PV Diagrams
A PV Diagram graphs Pressure vs. Volume. The state of a gas is a point on this graph. A process is a path from one point to another.
The Golden Rule of PV Diagrams: The absolute value of the area under the curve equals the magnitude of the work done ($|W| = Area$).
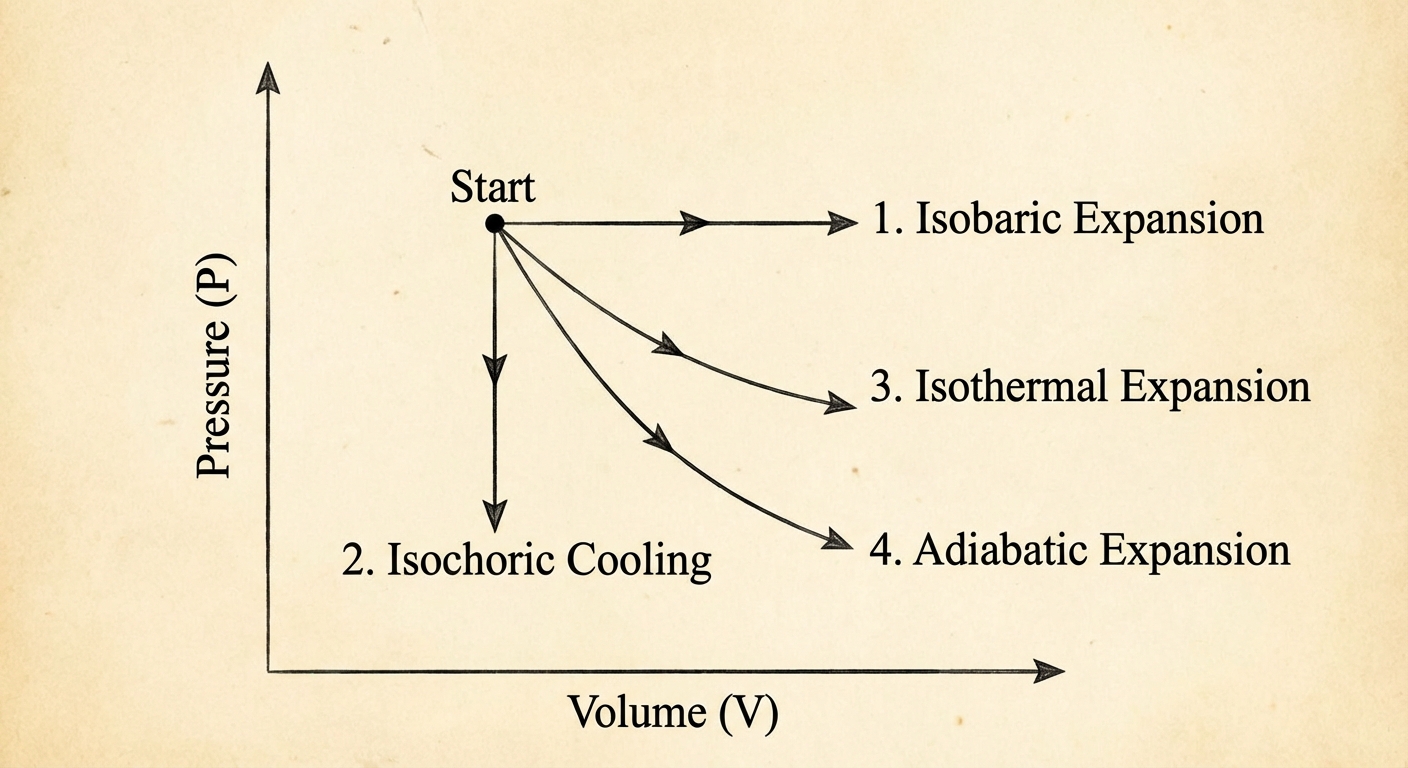
1. Isobaric Process (Constant Pressure)
- Graph: Horizontal line.
- Work: $W = -P(Vf - Vi)$.
- First Law: $\Delta U = Q + P(- \Delta V)$. Both $Q$ and $W$ occur.
- Example: Heating a gas in a cylinder with a movable piston free to expand.
2. Isochoric / Isovolumetric Process (Constant Volume)
- Graph: Vertical line.
- Work: No area under curve $\rightarrow$ $W = 0$.
- First Law: $\Delta U = Q$. All heat added goes directly into increasing internal energy (temperature).
- Example: Heating a gas in a rigid, sealed container.
3. Isothermal Process (Constant Temperature)
- Graph: A curve following the isotherm ($P \propto 1/V$).
- Internal Energy: Since $\Delta T = 0$, $\Delta U = 0$.
- First Law: $Q = -W$ (or $Q = P\Delta V$ roughly, but requires calculus for exact value; usually area is given). Heat added is converted entirely into work done by the gas.
- Example: Very slow expansion where the gas stays in equilibrium with a heat reservoir.
4. Adiabatic Process (No Heat Transfer)
- Graph: A steep curve (steeper than isothermal).
- Heat: $Q = 0$ (Insulated system or very fast process).
- First Law: $\Delta U = W$.
- Example: Rapid compression of a piston (diesel engine) or spraying an aerosol can (can gets cold).
The Second Law and Entropy
While the First Law says energy is conserved, the Second Law dictates the direction of natural processes.
Entropy ($S$)
Entropy is a measure of the disorder or randomness of a system. More formally, it relates to the number of microstates (arrangements) accessible to a system.
Statement: The total entropy of an isolated system can never decrease over time. $\Delta S_{universe} > 0$ for irreversible processes.
Heat Engines
A heat engine takes heat from a hot reservoir ($QH$), converts some to work ($W$), and exhausts the rest as waste heat to a cold reservoir ($QC$).
Energy Diagram: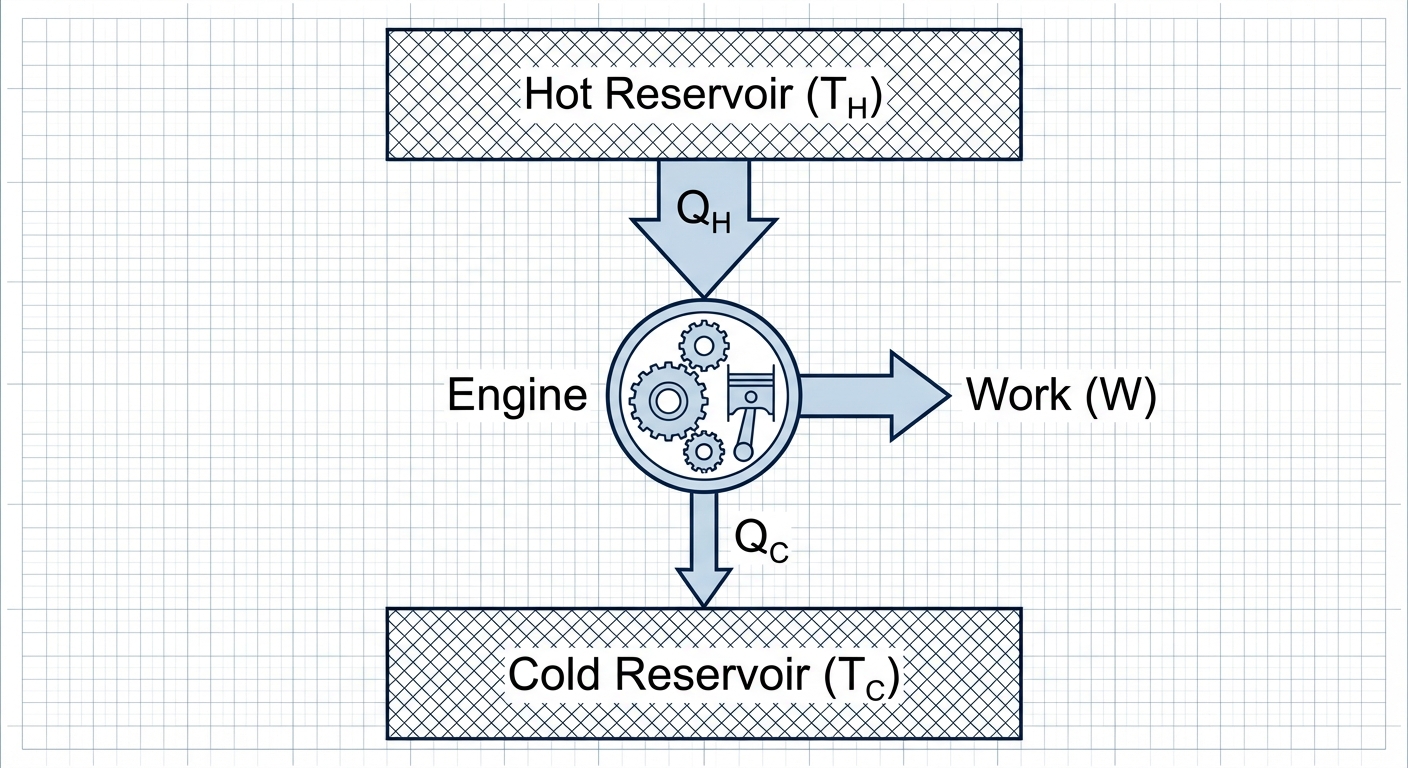
Conservation of Energy for Engines:
Thermal Efficiency ($e$):
Efficiency is "what you get" divided by "what you paid for."
Carnot Cycle (Ideal Engine)
The Carnot engine is a theoretical engine with the maximum possible efficiency. It operates on two isothermal and two adiabatic processes. Its efficiency depends only on temperatures.
Note: Temperatures MUST be in Kelvin.
Common Mistakes & Pitfalls
Celsius vs. Kelvin:
- Mistake: Using Celsius in $PV=nRT$ or Carnot efficiency equations.
- Correction: Always convert to Kelvin ($K = ^\circ C + 273$) immediately.
Sign Errors on Work:
- Mistake: Thinking expansion is positive work.
- Correction: In Physics 2, expansion means the gas pushes out, losing energy. $W$ is negative. Compression means you push in, adding energy. $W$ is positive.
Heat vs. Temperature:
- Mistake: Treating them as the same thing.
- Correction: Temperature is a state function (snapshot of KE). Heat is a process (energy in transit).
Cycle Work:
- Mistake: Thinking $\Delta U$ for a full cycle is non-zero.
- Correction: If a graph returns to the starting point, $\Delta T = 0$, so net $\Delta U = 0$. Net Work is the area enclosed by the cycle.
Isothermal vs. Adiabatic Curves:
- Mistake: Confusing the two on a PV diagram.
- Correction: Adiabats are steeper drops than Isotherms because temperature drops during adiabatic expansion, lowering pressure faster.