Comprehensive Guide to Unit 7: Quantum, Atomic, and Nuclear Physics
1. The Particle Nature of Waves (Photons)
Classical physics viewed light purely as a wave and matter purely as particles. Modern physics begins with the realization that light has particle-like properties and matter has wave-like properties. This breakdown of the classical distinction is the foundation of Quantum Mechanics.
The Photon Model
Electromagnetic waves consist of discrete packets of energy called photons. Unlike a continuous wave where energy depends on amplitude (brightness), the energy of a single photon depends solely on its frequency.
Where:
- $E$ = Energy of the photon (Joules or eV)
- $h$ = Planck’s Constant ($6.63 \times 10^{-34} \text{ J}\cdot\text{s}$ or $4.14 \times 10^{-15} \text{ eV}\cdot\text{s}$)
- $f$ = Frequency (Hz)
- $\lambda$ = Wavelength (m)
- $c$ = Speed of light ($3.0 \times 10^8$ m/s)
Key Concept: A brighter light source emits more photons, but the individual energy of each photon remains the same if the color (frequency) is unchanged.
The Photoelectric Effect
This is the definitive experiment proving the particle nature of light. When light shines on a metal surface, electrons (photoelectrons) are ejected—but only if the light's frequency is high enough.
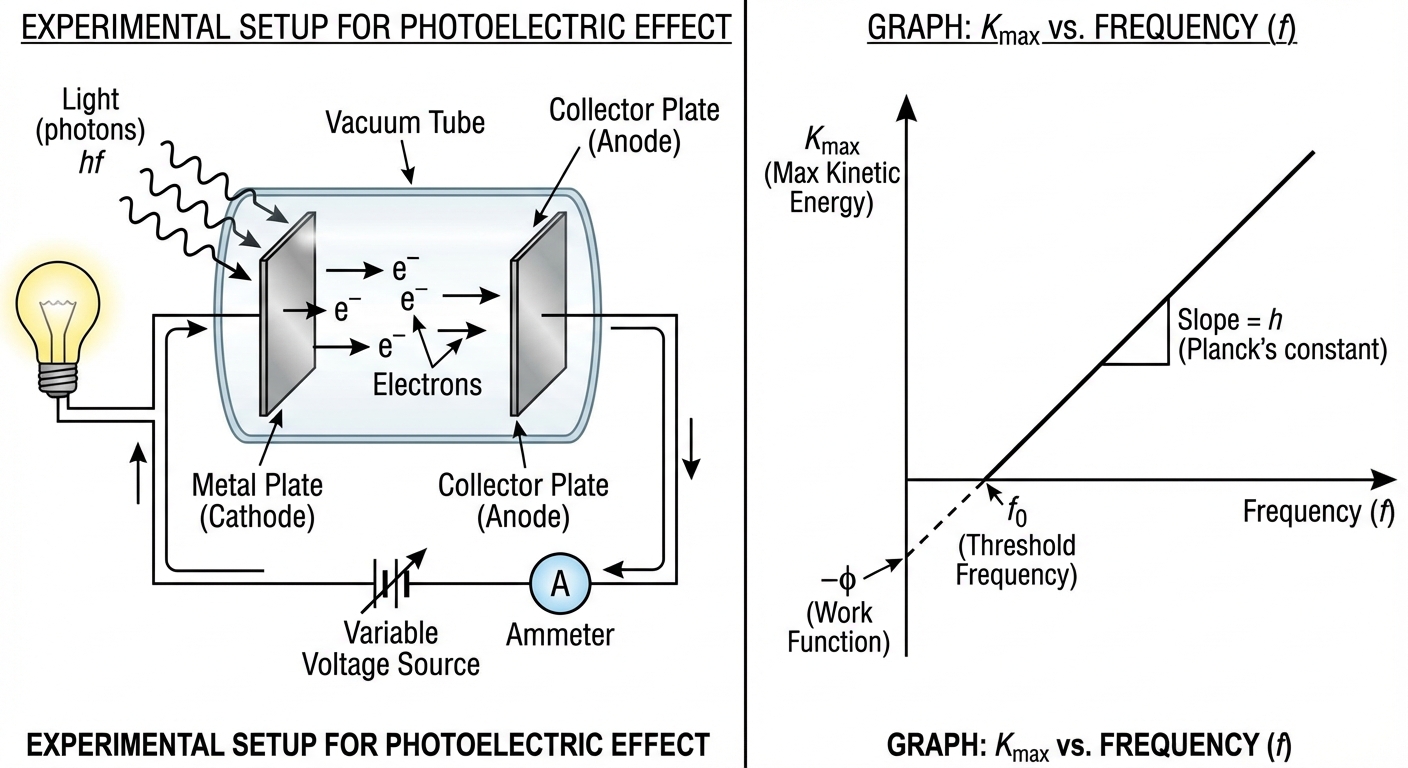
The Conservation of Energy Equation
The energy of the incoming photon is split into two parts: freeing the electron from the metal and giving it kinetic energy.
Where:
- $K_{max}$ = Maximum kinetic energy of the ejected electron
- $hf$ = Energy of the incident photon
- $\phi$ (or $W$) = Work Function: The minimum energy required to unbind an electron from the specific metal.
Rules of Photoelectric Emission
- Threshold Frequency ($f_0$): No electrons are ejected if $hf < \phi$. The frequency must exceed a minimum threshold.
- Intensity vs. Energy: Increasing the intensity (brightness) of the light increases the number of ejected electrons (current), but NOT their speed ($K_{max}$).
- Frequency vs. Energy: Increasing the frequency (color toward violet/UV) increases the speed ($K_{max}$) of the electrons.
Graphical Analysis
A graph of $K_{max}$ (y-axis) vs. Frequency $f$ (x-axis) is extremely common on the AP exam.
- Slope: Planck’s constant ($h$).
- X-intercept: Threshold frequency ($f_0$).
- Y-intercept: Negative Work function ($-\phi$).
Photon Momentum
Even though photons have no mass ($m=0$), they possess momentum ($p$). This is demonstrated by Compton Scattering (collisions between photons and electrons).
2. The Wave Nature of Matter
Just as light behaves like a particle, matter (electrons, protons, baseballs) behaves like a wave. This is Wave-Particle Duality.
De Broglie Wavelength
Louis de Broglie proposed that any particle with momentum has an associated wavelength.
Where:
- $m$ = mass of the particle (kg)
- $v$ = velocity limit (m/s)
Implications:
- Macroscopic objects (like a car) have massive momentum, so their wavelength is infinitesimally small and undetectable.
- Microscopic objects (electrons) have small momentum, resulting in wavelengths measurable through diffraction experiments.
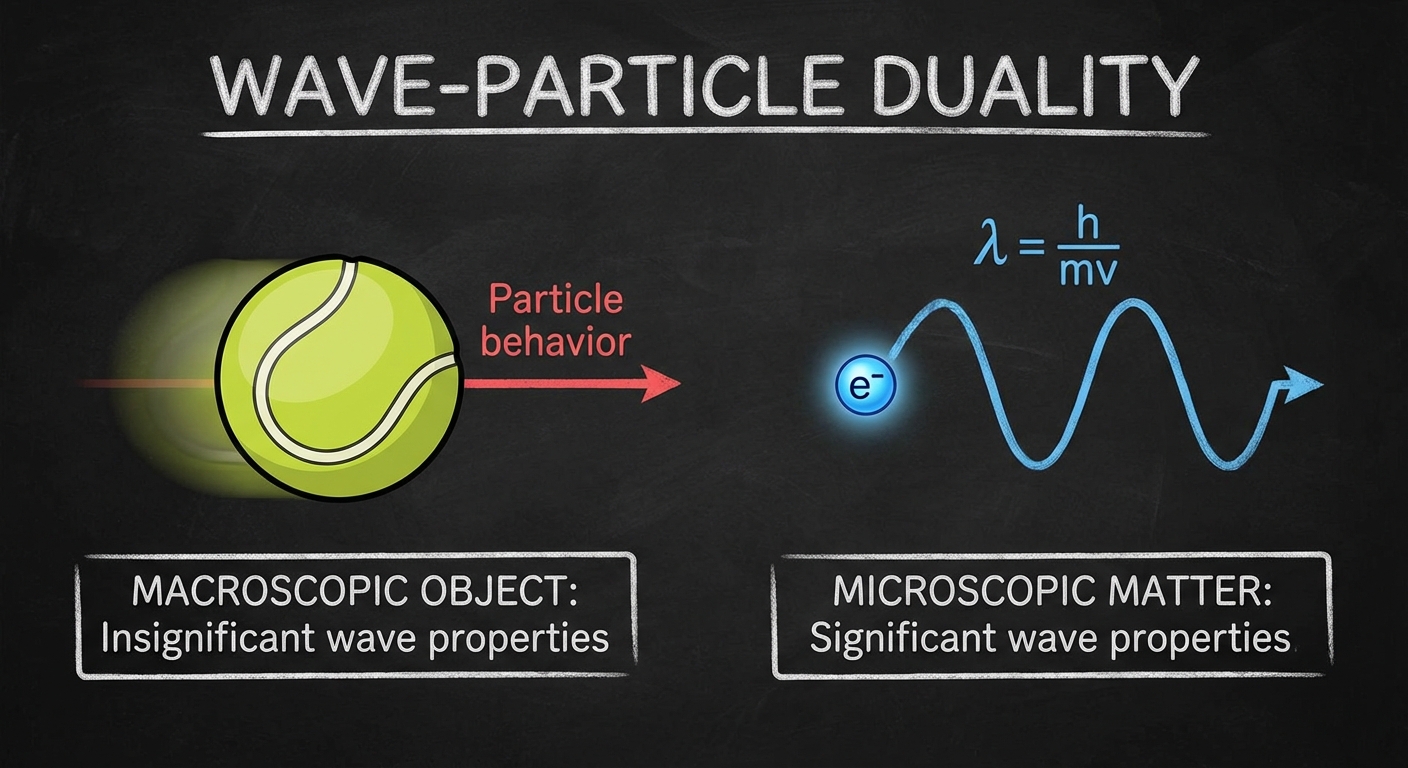
Davisson-Germer Experiment
This experiment fired electrons at a crystal lattice. The electrons produced an interference pattern (diffraction), typically associated with waves, proving that particles have wave properties.
3. Atomic Structure and Energy Levels
The Bohr Model
Niels Bohr combined classical mechanics with quantum theory to explain the hydrogen atom. He postulated that electrons exist in stable, discrete orbits (energy levels) without radiating energy.
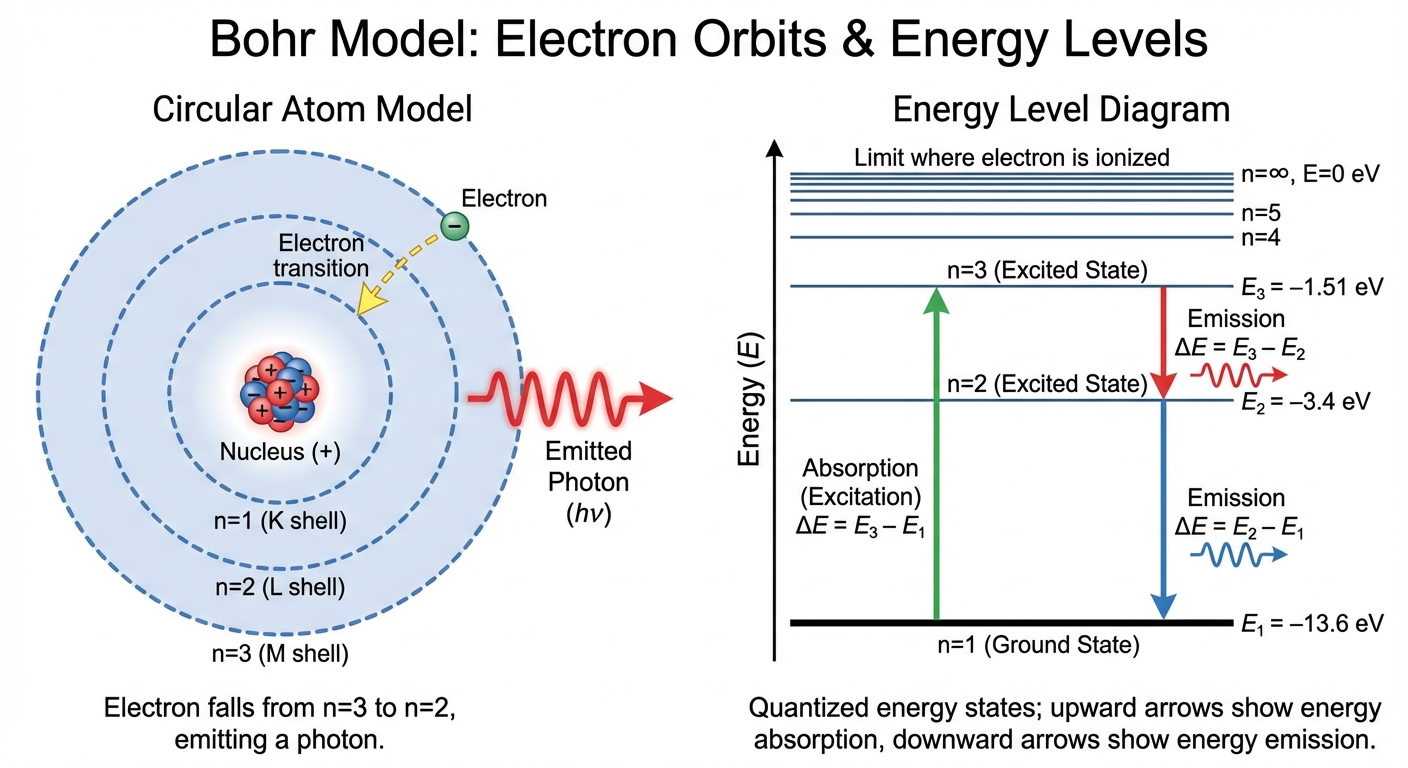
Emission and Absorption Spectra
Atoms have quantized energy levels. Electrons can "jump" between these levels by absorbing or emitting photons.
- Absorption: An electron in a lower energy ground state absorbs a photon and jumps to a higher energy excited state. The photon’s energy must exactly match the energy difference between levels.
- Emission: An electron falls from a higher level to a lower level, releasing a photon equal to the energy difference.
Calculating Transition Energy
- Ionization Energy: The energy required to remove an electron completely ($n = 1 \to n = \infty$). The energy at $n=\infty$ is 0 eV, so the ionization energy is the absolute value of the ground state energy.
4. Nuclear Physics
Nucleus Structure and Stability
The nucleus contains nucleons: protons (positive) and neutrons (neutral).
- Atomic Number ($Z$): Number of protons (defines the element).
- Mass Number ($A$): Total protons + neutrons.
- Neutron Number ($N$): $A - Z$.
Notation: $Z^AE$ (e.g., $^{238}{92}U$)
The Strong Nuclear Force
Protons repel each other via the electrostatic force (Coulomb's Law). The nucleus stays together because of the Strong Nuclear Force, which is attractive, very strong, but extremely short-range (acting only between adjacent nucleons).
Mass-Energy Equivalence
Mass and energy are interchangeable. The mass of a stable nucleus is always less than the sum of the masses of its individual protons and neutrons. This missing mass is called the Mass Defect.
In AP Physics 2, we often use the conversion factor for Atomic Mass Units (u):
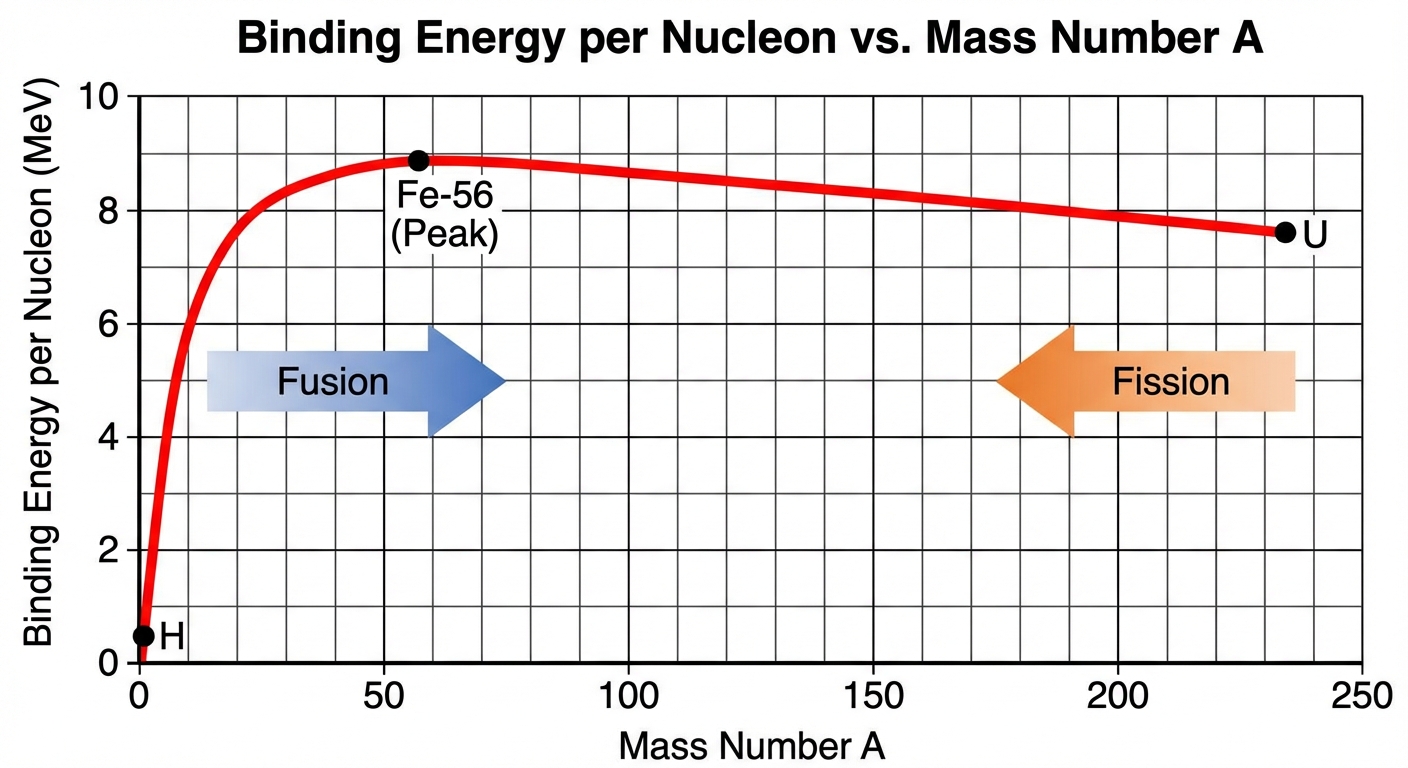
Binding Energy: The energy equivalent of the mass defect. It is the energy required to disassemble a nucleus into individual protons and neutrons. Higher binding energy per nucleon indicates a more stable nucleus (Iron-56 is the peak).
5. Radioactive Decay and Reactions
Unstable nuclei spontaneously decay to become more stable. Charge (Atomic Number $Z$) and Mass Number ($A$) must be conserved in all reactions.
Types of Decay
| Decay Type | Particle Emitted | Symbol | Change in $A$ | Change in $Z$ | Description |
|---|---|---|---|---|---|
| Alpha | Helium Nucleus | $2^4\alpha$ or $2^4He$ | $-4$ | $-2$ | Heavy nucleus ejects a standard helium nucleus. |
| Beta-Minus | Electron | ${-1}^0\beta$ or ${-1}^0e$ | $0$ | $+1$ | A neutron turns into a proton and an electron. |
| Beta-Plus | Positron | ${+1}^0\beta$ or ${+1}^0e$ | $0$ | $-1$ | A proton turns into a neutron and a positron. |
| Gamma | High-energy Photon | $_0^0\gamma$ | $0$ | $0$ | Nucleus transitions from excited to ground state. |
Half-Life
The time it takes for half of a radioactive sample to decay.
- After 1 half-life: 50% remains.
- After 2 half-lives: 25% remains.
- After 3 half-lives: 12.5% remains.
Formula for remaining amount $N$:
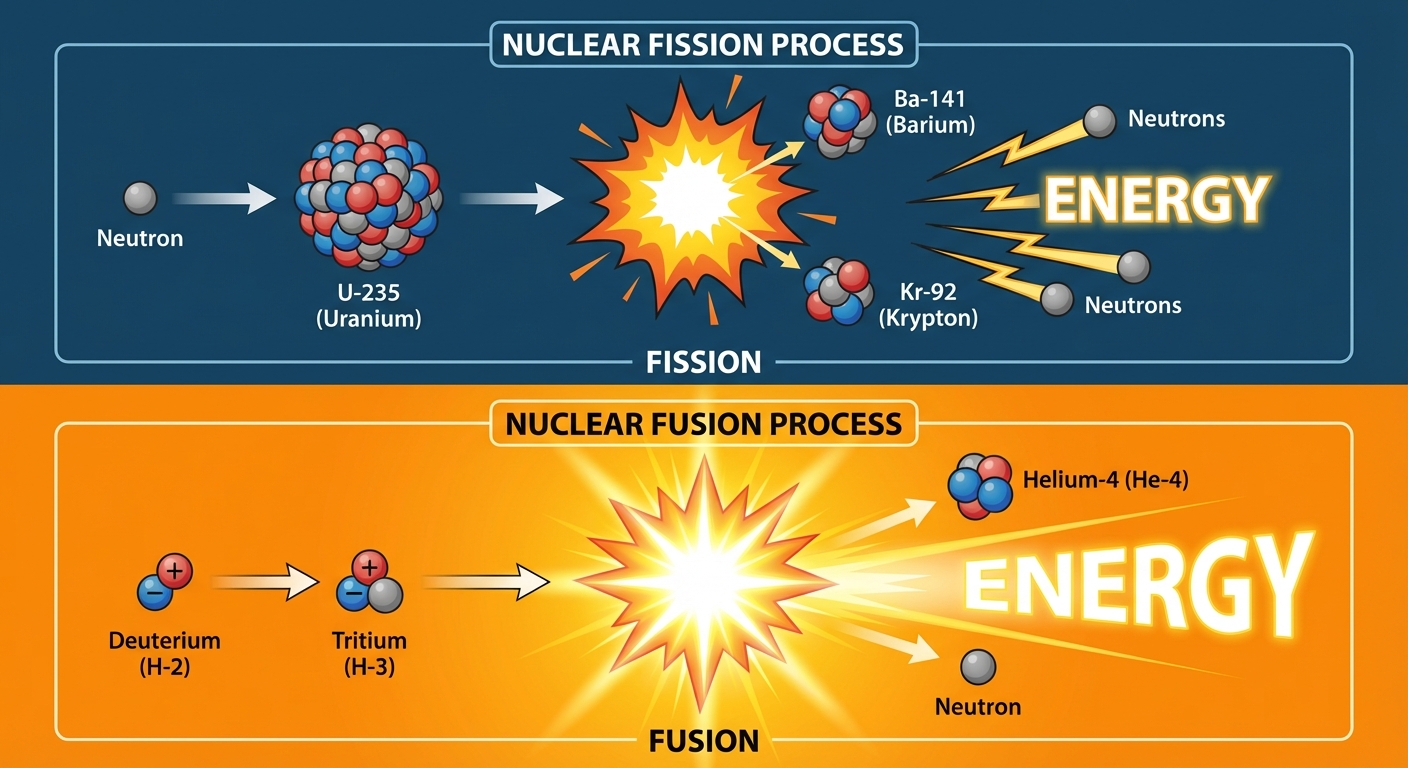
Fission vs. Fusion
- Fission: A heavy nucleus (like Uranium-235) splits into lighter nuclei. Occurs for elements heavier than Iron-56. Releases energy.
- Fusion: Light nuclei (like Hydrogen) combine to form a heavier nucleus. Occurs for elements lighter than Iron-56. Releases immense energy (powers the sun).
Common Mistakes & Pitfalls
- Confusing Intensity with Frequency: In the photoelectric effect, students often think brighter light (higher intensity) gives electrons more energy. Correction: Intensity increases the current (number of electrons); frequency increases the kinetic energy (speed per electron).
- Sign Errors in Energy Levels: Remember that bound energy levels are negative ($E_1 = -13.6$ eV). When calculating $\Delta E$, just take the absolute difference to find photon energy. If the answer is negative, it just indicates direction (emission).
- Mass Defect Calculation: When calculating $\Delta m$, do not just subtract mass numbers (integers like 238 - 234). You must use the precise atomic masses (e.g., 238.05078 u). The difference is tiny but essential.
- Beta Decay Conservation: In Beta-minus decay ($Z \to Z+1$), students incorrectly think a proton is added from outside. Correction: A neutron changes into a proton inside the nucleus.
- Stopping Potential: Often questions give "Stopping Potential" ($Vs$). This corresponds directly to kinetic energy: $K{max} = qV_s$. Don't forget to convert eV to Joules if using $h$ in J·s ($1 \text{ eV} = 1.6 \times 10^{-19} \text{ J}$).