Module 1.1: Foundations of Electrostatics
Foundations of Electrostatics
Electric Charge and Conservation
The Nature of Charge
Electric Charge is a fundamental intrinsic property of matter that governs how particles interact through electromagnetic forces. Matter is composed of atoms involving protons (positive charge), electrons (negative charge), and neutrons (neutral).
- Polarlity: Charge comes in two types: Positive ($+$) and Negative ($-$). Like charges repel; opposite charges attract.
- SI Unit: The unit of charge is the Coulomb (C).
- Fundamental Charge ($e$): The smallest isolated charge found in nature is the charge of an electron or proton.
Quantization of Charge
Because electric charge comes in discrete packets ($e$), the total charge ($Q$) of any object must be an integer multiple ($n$) of the elementary charge.
Note: While quarks have fractional charges (e.g., $e/3$), they are never found isolated in nature, so standard electrostatics treats $e$ as the fundamental unit.
Law of Conservation of Charge
In an isolated system, the total algebraic sum of electric charge remains constant. Charge cannot be created or destroyed, only transferred from one object to another.
Equation form: $\sum Q{\text{initial}} = \sum Q{\text{final}}$
Usually, the transfer of charge involves the movement of electrons. Protons are bound in the nucleus and do not move during standard electrostatic charging processes.
Conductors, Insulators, and Charging Methods
Material Classifications
- Conductors: Materials where charge carriers (valence electrons) are free to move throughout the atomic lattice (e.g., metals like copper, gold). When charged, the excess charge resides entirely on the surface due to mutual repulsion.
- Insulators (Dielectrics): Materials where electrons are tightly bound to atoms and cannot move freely (e.g., rubber, glass, plastic). Charges placed on an insulator stay where they are put.
- Semiconductors: Materials with intermediate conductivity that can be altered by temperature or doping (e.g., silicon).
Charging Mechanisms
1. Charging by Friction (Triboelectric Effect)
Transfer of electrons between two different insulators rubbed together based on electron affinity (e.g., rubbing a glass rod with silk).
2. Charging by Conduction (Contact)
Transfer of charge via direct physical contact. If a charged conductor touches a neutral conductor, electrons flow until the electric potential is balanced.
3. Charging by Induction
A process to charge a conductor without physical contact with the charging object. This requires a ground connection.
Step-by-Step Induction:
- Bring a charged object (e.g., negative rod) near a neutral sphere. Electrons on the sphere are repelled to the far side (polarization).
- Connect the sphere to ground (earth). The repelled electrons leave the sphere into the ground.
- Disconnect the ground first, then remove the charged rod.
- The sphere is left with a net positive charge.

Coulomb's Law
Coulomb's Law quantifies the electrostatic force between two stationary point charges. It allows us to calculate the magnitude and direction of the electric force.
The Scalar Formula
The magnitude of the electric force ($FE$) between two point charges $q1$ and $q_2$ separated by a distance $r$ is:
Constants
- Coulomb's Constant ($k$): $\approx 8.99 \times 10^9 \text{ N}\cdot\text{m}^2/\text{C}^2$
- Permittivity of Free Space ($\epsilon_0$): $\approx 8.85 \times 10^{-12} \text{ C}^2/(\text{N}\cdot\text{m}^2)$
Vector Properties
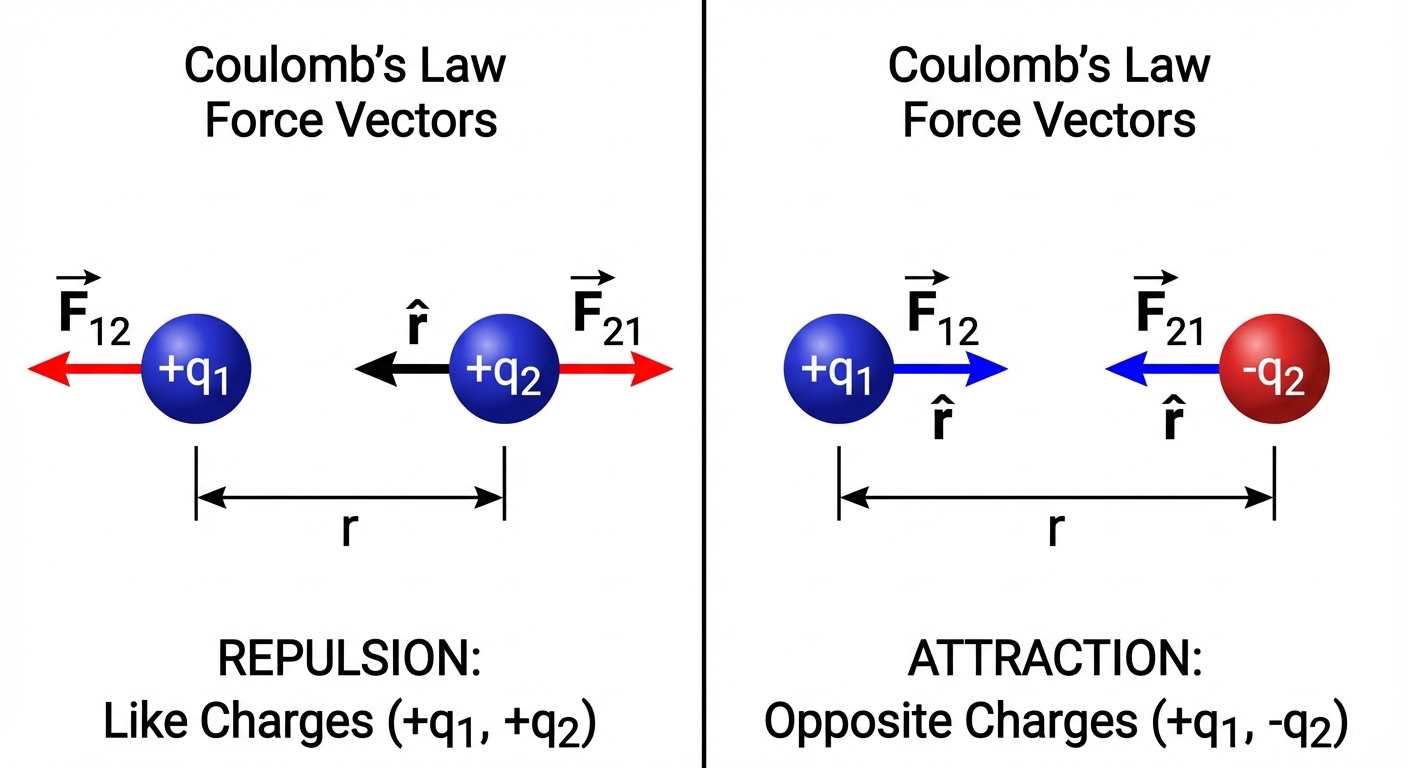
The Electrostatic Force is a vector. While the formula above gives magnitude, the direction is determined by the signs of the charges along the line connecting them.
- Attraction: Force vectors point inward toward each other.
- Repulsion: Force vectors point outward away from each other.
| Feature | Coulomb's Law ($F_E$) | Newton's Law of Gravity ($F_G$) |
|---|---|---|
| Source | Charge ($q$) | Mass ($m$) |
| Type | Attractive or Repulsive | Always Attractive |
| Magnitude | Very Strong ($k \approx 10^9$) | Very Weak ($G \approx 10^{-11}$) |
| Relation | Inverse-square ($1/r^2$) | Inverse-square ($1/r^2$) |
The Principle of Superposition
In systems with multiple point charges (System of $N$ charges), the net electric force exerted on a specific charge is the vector sum of the individual forces exerted by all other charges.
Strategy for Calculation
- Diagram: Draw the arrangement and visualize the force vectors on the target charge.
- Magnitudes: Calculate the magnitude of each individual force using Coulomb's Law ($k|q1q2|/r^2$).
- Components: Resolve each force vector into $x$ and $y$ components using trigonometry ($\,\cos\theta, \sin\theta\,$).
- Summation: Add the components: $F{net,x} = \sum Fx$ and $F{net,y} = \sum Fy$.
- Resultant: Combine components to find the final magnitude using Pythagorean theorem ($F = \sqrt{Fx^2 + Fy^2}$) and direction using $\tan^{-1}(Fy/Fx)$.
Worked Example: Three Charges on a Line
Problem: Consider three charges on the x-axis: $q1 = +2 \text{ \mu C}$ at $x=0$, $q2 = -3 \text{ \mu C}$ at $x=2\text{ m}$, and $q3 = +5 \text{ \mu C}$ at $x=4\text{ m}$. Find the net force on $q2$.
Solution:
Identify Forces:
- $F{12}$: Force on $q2$ by $q1$. Since signs are opposite ($+/-$), they attract. $q1$ pulls $q_2$ to the left ($-x$).
- $F{32}$: Force on $q2$ by $q3$. Signs are opposite ($-/+$), they attract. $q3$ pulls $q_2$ to the right ($+x$).
Calculate Magnitudes:
Vector Sum:
Answer: The net force is $2.03 \times 10^{-2}$ N directed to the right.
Continuous Charge Distributions
In AP Physics C: E&M, you must handle objects where charge is spread continuously over a length, area, or volume, rather than concentrated at points. While you typically calculate the Electric Field later, understanding the setup for Force is the foundation.
To find the force exerted by a continuous object on a point charge $Q$, we treat the object as a collection of infinitesimal point charges $dq$. The total force is the integral of all infinitesimal forces $d\vec{F}$.
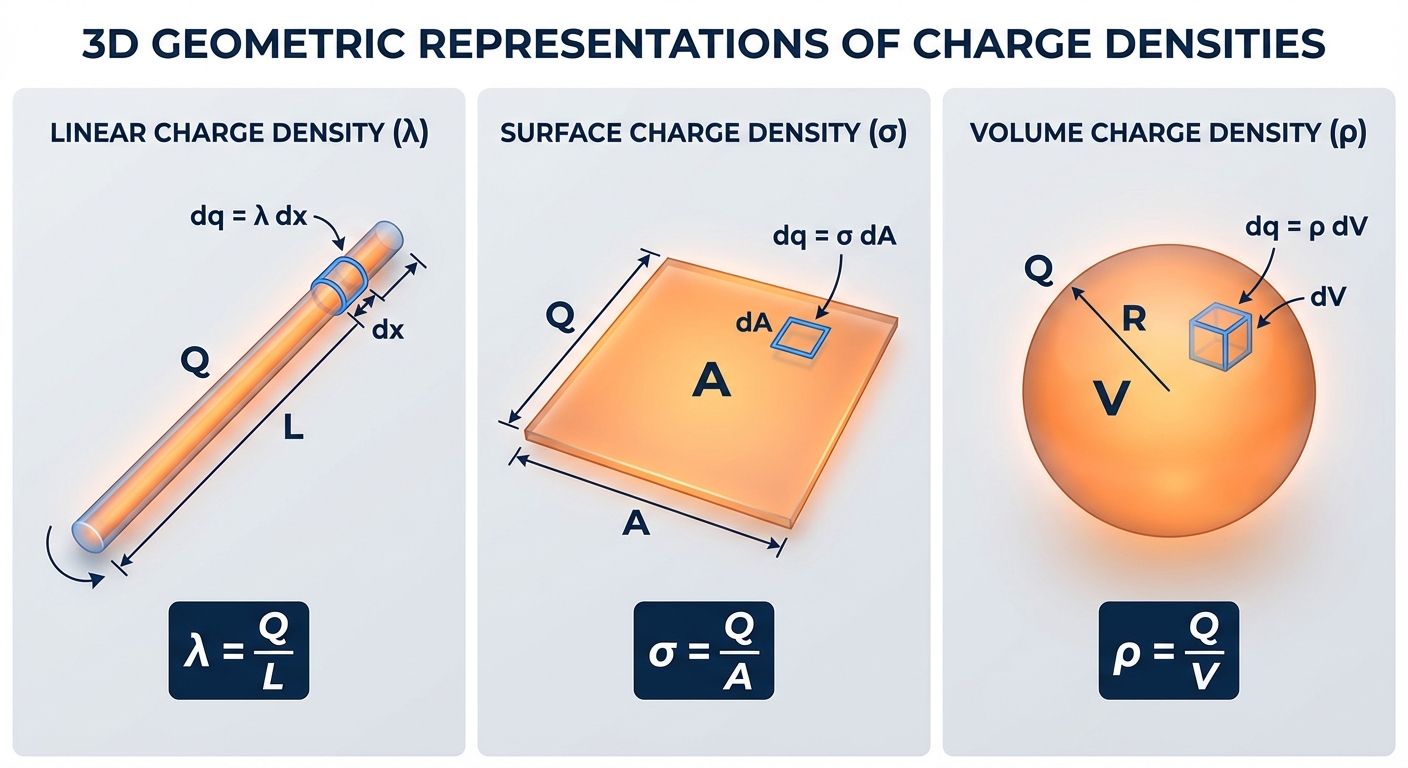
Charge Densities
To solve the integral, you must express $dq$ in terms of spatial coordinates ($dx, dA, dV$) using charge density.
| Type | Symbol | Definition | Relationship ($dq$) |
|---|---|---|---|
| Linear | $\lambda$ (lambda) | Charge per unit length | $dq = \lambda \, dl$ |
| Surface | $\sigma$ (sigma) | Charge per unit area | $dq = \sigma \, dA$ |
| Volume | $\rho$ (rho) | Charge per unit volume | $dq = \rho \, dV$ |
For uniform distributions, density is Total Charge / Total Size (e.g., $\lambda = Q/L$). For non-uniform distributions, density is a function of position (e.g., $\lambda(x) = \alpha x$).
Common Mistakes & Pitfalls
- Ignoring Vector Direction: This is the #1 error. Students often calculate the scalar force ($kq1q2/r^2$) and just add the numbers together. You must decompose forces into $x$ and $y$ vectors before adding.
- Shell Theorem Confusion: A charged spherical shell acts like a point charge only for points outside the shell. Inside a uniformly charged conducting shell, the net force on a test charge is zero. Do not apply $kq/r^2$ blindly inside a shell.
- Sign confusion in the formula: It is highly recommended to substitute absolute values of charge into Coulomb's law to find the magnitude, and determine the direction logicially (like repels like) based on the diagram. Plugging negative signs into the math often leads to confusion about whether "-" means "left" or "attraction".
- Unit Conversion: Remember that $\mu\text{C}$ (microcoulombs) is $10^{-6}$ C and $\text{nC}$ (nanocoulombs) is $10^{-9}$ C. Failing to convert to Coulombs will make your answer off by orders of magnitude.
- Induction vs. Conduction: Remember that Induction creates a net charge on an object opposite to the source, while Conduction creates a net charge same as the source.