AP Physics 2: Foundations of Electrostatics
Unit 2: Electric Force, Field, and Potential
The Nature of Electric Charge
Electrostatics is the study of electromagnetic phenomena that occur when there are no moving charges (static equilibrium). At the heart of this study is the fundamental property known as Electric Charge.
Fundamental Properties
Matter is made of atoms, which consist of protons, neutrons, and electrons. The interactions between these particles dictate electrical behavior.
- Two Types of Charge: By convention, we call them positive (+) and negative (-).
- Protons carry positive charge.
- Electrons carry negative charge.
- Neutrons are neutral (zero charge).
- Interaction Rule: Like charges repel; opposite charges attract.
Quantization of Charge
Electric charge is quantized, meaning it comes in discrete packets. You cannot have a charge of just any value; it must be an integer multiple of the elementary charge ($e$).
q = n \cdot e
Where:
- $q$ is the total net charge.
- $n$ is an integer (positive or negative).
- $e$ is the elementary charge constant.
Key Constants:
- Elementary Charge: $e = 1.60 \times 10^{-19} \, \text{C}$
- Mass of Electron: $m_e = 9.11 \times 10^{-31} \, \text{kg}$
- Mass of Proton: $m_p = 1.67 \times 10^{-27} \, \text{kg}$
Note: The unit of charge is the Coulomb (C). A Coulomb is actually a massive amount of charge; in AP Physics problems, you will often see micro-Coulombs ($\mu\text{C} = 10^{-6} \, \text{C}$) or nano-Coulombs ($n\text{C} = 10^{-9} \, \text{C}$).
Conservation of Electric Charge
The Law of Conservation of Charge states that the net electric charge of an isolated system remains constant. Charge cannot be created or destroyed, only transferred from one object to another.
Algebraically, if you have a system of objects, the sum of initial charges equals the sum of final charges:
\sum q{initial} = \sum q{final}
Conductors vs. Insulators
The ability of charge to move through a material depends on its atomic structure.
- Conductors: Materials where electrons are loosely bound to nuclei and can move freely throughout the material (e.g., metals like copper, gold, aluminum).
- Equilibrium Condition: On a charged conductor in electrostatic equilibrium, all excess charge resides on the surface, and the electric field inside is zero.
- Insulators: Materials where electrons are tightly bound to specific atoms and cannot move freely (e.g., rubber, glass, plastic). Charges placed on an insulator tend to stay where they are put.
Methods of Charging
There are three primary ways to manipulate charge:
- Friction (Triboelectric Effect): Rubbing two neutral insulators together transfers electrons based on electron affinity (e.g., rubbing a balloon on hair).
- Conduction (Contact): Touching a charged object to a neutral object. Electrons physically flow to equilibrate the repulsive forces.
- Induction: Charging a conductor without physical contact. This involves polarizing the object and then providing a path (ground) for electrons to escape or enter.
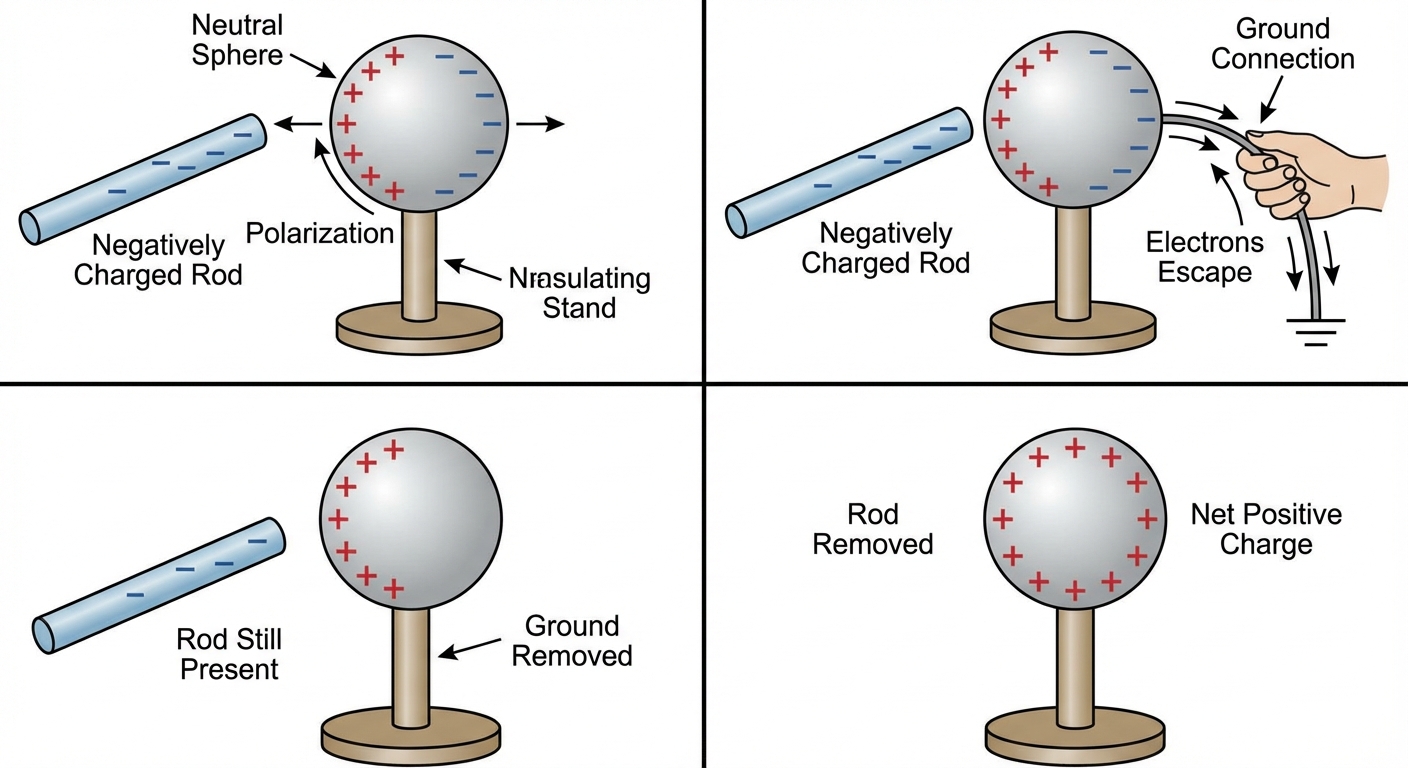
The Induction Process (Step-by-Step):
- Bring a charged rod (e.g., negative) near a neutral conducting sphere. Electrons in the sphere are repelled to the far side (polarization).
- Connect the far side of the sphere to the ground (grounding).
- Electrons flow from the sphere into the ground to escape the repulsive force of the rod.
- Disconnect the ground wire, then remove the rod. The sphere now has a net positive charge.
Coulomb’s Law and Electric Force
The magnitude of the electric force between two point charges is directly proportional to the product of the magnitudes of the charges and inversely proportional to the square of the distance between them.
The Formula
FE = k \frac{|q1 q_2|}{r^2}
Where:
- $F_E$ is the magnitude of the electric force (Newtons, N).
- $q1, q2$ are the magnitudes of the charges (Coulombs, C).
- $r$ is the distance between the centers of the two charges (meters, m).
- $k$ is Coulomb's constant.
Constants:
k = 8.99 \times 10^9 \, \frac{\text{N}\cdot\text{m}^2}{\text{C}^2}
Sometimes written using vacuum permittivity ($\epsilon0$): k = \frac{1}{4\pi\epsilon0}
Vector Nature and Superposition
Coulomb's Law provides the magnitude. You must determine the direction conceptually:
- Repulsive: If charges have the same sign ($++$ or $--$), the force vector points away from the other charge.
- Attractive: If charges have opposite signs ($+-$), the force vector points toward the other charge.
The Principle of Superposition:
If multiple point charges are present, the net force on a specific charge is the vector sum of the individual forces exerted by all other charges.
\vec{F}{net} = \vec{F}1 + \vec{F}2 + \vec{F}3 + …
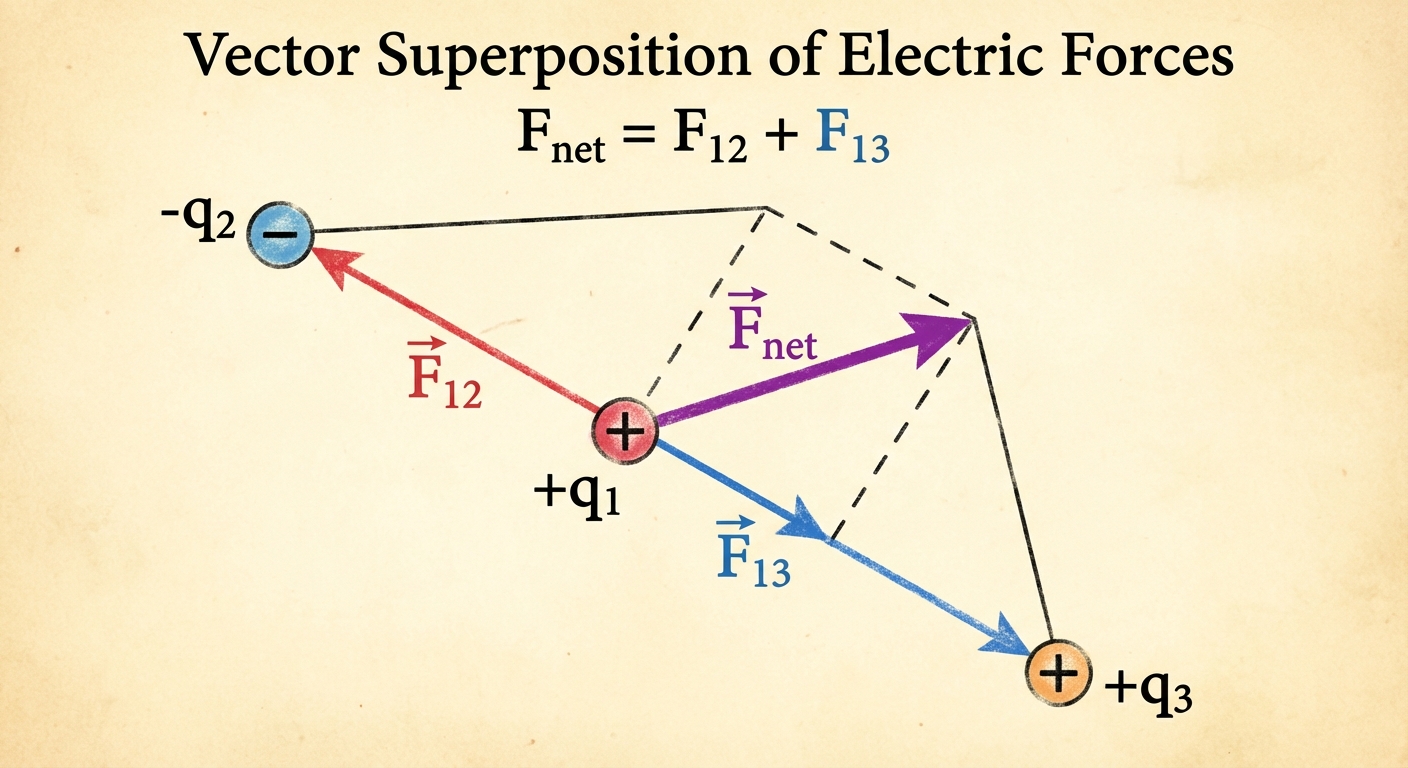
Worked Example: 1D Superposition
Scenario: Three charges are arranged on a line.
- $q_1 = +2 \mu\text{C}$ at $x = 0$
- $q_2 = -3 \mu\text{C}$ at $x = 2 \text{ m}$
- $q_3 = +5 \mu\text{C}$ at $x = 5 \text{ m}$
- Task: Find the net force on $q_2$.
Solution:
Identify forces acting on $q_2$:
- $F{12}$: Force from $q1$ on $q2$. Signs are (+)(-), so it is attractive. $q1$ pulls $q_2$ left (negative direction).
- $F{32}$: Force from $q3$ on $q2$. Signs are (+)(-), so it is attractive. $q3$ pulls $q_2$ right (positive direction).
Calculate Magnitudes:
- F_{12} = k \frac{|(2\times 10^{-6})(-3\times 10^{-6})|}{(2)^2} = (9\times 10^9) \frac{6\times 10^{-12}}{4} = 0.0135 \, \text{N}
- F_{32} = k \frac{|(5\times 10^{-6})(-3\times 10^{-6})|}{(3)^2} = (9\times 10^9) \frac{15\times 10^{-12}}{9} = 0.0150 \, \text{N} (Note: distance is $5-2=3$)
Vector Sum:
- $F{net} = -F{12} + F_{32}$ (assuming right is positive)
- $F_{net} = -0.0135 + 0.0150 = +0.0015 \, \text{N}$
- The net force is 0.0015 N to the right.
Comparison: Electric vs. Gravitational Force
| Feature | Coulomb's Law ($F_E$) | Newton's Gravity ($F_g$) |
|---|---|---|
| Formula | $k \frac{ | q1 q2 |
| Property | Charge ($q$) | Mass ($m$) |
| Type | Attractive OR Repulsive | Always Attractive |
| Strength | Very Strong ($k \approx 10^9$) | Very Weak ($G \approx 10^{-11}$) |
| Range | Infinite ($1/r^2$) | Infinite ($1/r^2$) |
Common Mistakes & Pitfalls
- Sign Errors in Vectors: Students often plug negative signs directly into Coulomb's Law. Recommendation: Calculate the absolute magnitude first using $|q|$, then assign positive/negative direction based on drawing a Free Body Diagram (FBD).
- Forgetting to Convert Units: Charges are often given in $\mu C$ (micro) or $nC$ (nano), and distances in $cm$. You MUST convert to Coulombs and meters before using $k \approx 9 \times 10^9$.
- Newton's Third Law Violations: Students often think a large charge exerts a stronger force on a small charge than the small charge exerts on the large one. Fact: The forces are an action-reaction pair. They are equal in magnitude and opposite in direction ($|F{1 \to 2}| = |F{2 \to 1}|$).
- Inverse Square Law: Remember the relationship is $1/r^2$. If you double the distance, the force becomes $1/4$ as strong, not $1/2$.