Unit 7 Study Notes: Particle Physics & Solid State Applications
The Standard Model of Particle Physics
Classical physics treats matter as solid, continuous, or made of simple spheres. However, at subatomic scales, we use The Standard Model. This theoretical framework describes the fundamental building blocks of the universe and the forces through which they interact. It is currently the most accurate description of the micro-world.
Fundamental Forces
There are four fundamental forces in the universe. In AP Physics 2, you must understand their relative strengths and ranges, even though gravity is negligible at the particle scale.
| Force | Relative Strength | Range | Force Carrier (Boson) | Acts On |
|---|---|---|---|---|
| Strong Nuclear | 1 (Strongest) | Subatomic ($\approx 10^{-15}$ m) | Gluon | Quarks (Hadrons) |
| Electromagnetism | $10^{-2}$ | Infinite | Photon | Electrically charged particles |
| Weak Nuclear | $10^{-6}$ | Subatomic ($\approx 10^{-18}$ m) | W and Z Bosons | Quarks and Leptons |
| Gravity | $10^{-39}$ (Weakest) | Infinite | Graviton (hypothetical) | Mass/Energy |
Elementary Particles
Matter is composed of particles that cannot be split into smaller parts. These are divided into Fermions (matter particles) and Bosons (force carriers).
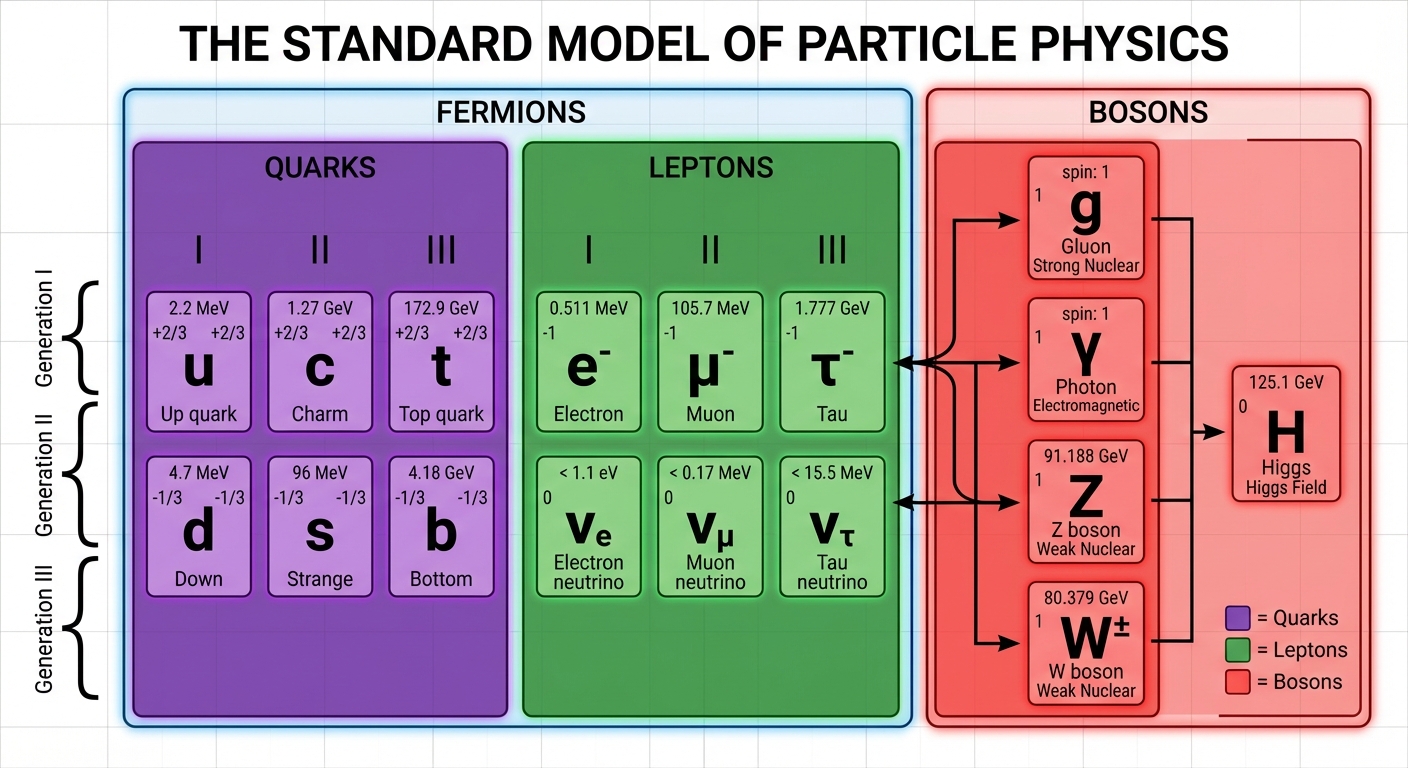
1. Quarks
Quarks are fundamental constituents of matter. They never exist in isolation due to a phenomenon called color confinement; they are always found in groups called Hadrons.
- Flavors: There are six flavors of quarks grouped into three generations:
- Up ($u$), Down ($d$)
- Charm ($c$), Strange ($s$)
- Top ($t$), Bottom ($b$)
- Charge: Up-type quarks have a charge of $+2/3e$; Down-type quarks have a charge of $-1/3e$.
Common Hadrons:
- Baryons: Made of 3 quarks.
- Proton: $uud$ (Total charge: $+2/3 + 2/3 - 1/3 = +1$)
- Neutron: $udd$ (Total charge: $+2/3 - 1/3 - 1/3 = 0$)
- Mesons: Made of a quark and an antiquark. (e.g., Pion).
2. Leptons
Leptons are fundamental particles that do not interact via the Strong Force. They can exist freely.
- Electron family: Electron ($e^-$) and Electron Neutrino ($\nu_e$)
- Muon family: Muon ($\mu^-$) and Muon Neutrino ($\nu_\mu$)
- Tau family: Tau ($\tau^-$) and Tau Neutrino ($\nu_\tau$)
Note: Neutrinos have tiny (non-zero) mass and no electric charge. They interact very weakly with matter, making them difficult to detect.
3. Antimatter
Every particle has a corresponding antiparticle with the same mass but opposite charge. Antimatter is denoted with a bar over the symbol (e.g., $\bar{u}$ is an anti-up quark) or a change in charge sign (e.g., $e^+$ is a positron, the electron's antiparticle).
Conservation Laws in Particle Physics
In any reaction (decay or collision), specific quantities must be conserved. If a proposed reaction violates any of these, it cannot occur.
- Conservation of Charge ($Q$): The total net charge before and after must be equal.
- Conservation of Baryon Number ($B$):
- Quarks have $B = +1/3$.
- Antiquarks have $B = -1/3$.
- Protons and Neutrons (Baryons) have $B = +1$.
- Mesons and Leptons have $B = 0$.
- Conservation of Lepton Number ($L$):
- Leptons (electron, muon, tau, neutrinos) have $L = +1$.
- Antileptons have $L = -1$.
- Non-leptons have $L = 0$.
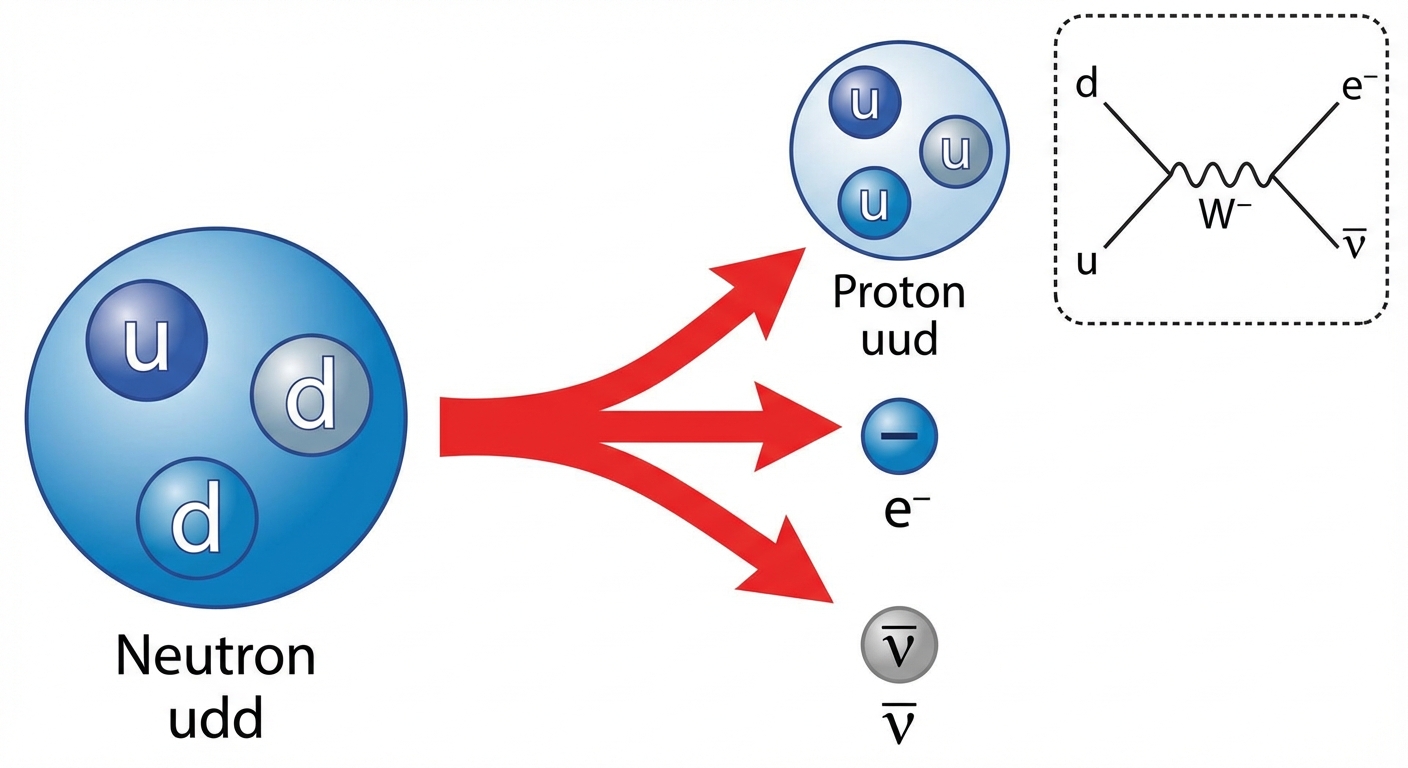
Example: Beta-Minus Decay
A neutron decays into a proton. To conserve charge, an electron is emitted. To conserve Lepton number, an electron antineutrino is emitted.
- Charge: $0 \rightarrow (+1) + (-1) + 0$ (Conserved)
- Baryon Number: $1 \rightarrow 1 + 0 + 0$ (Conserved)
- Lepton Number: $0 \rightarrow 0 + (+1) + (-1)$ (Conserved)
Semiconductors and Technology
While the Standard Model deals with the smallest particles, Modern Physics also applies quantum mechanics to solids to create the technology powering your computer. This relies on Band Theory.
Energy Band Theory
In a single atom, electrons inhabit discrete energy levels. In a solid crystal structure (like silicon), atoms are so close together that these discrete levels blur into continuous Energy Bands.
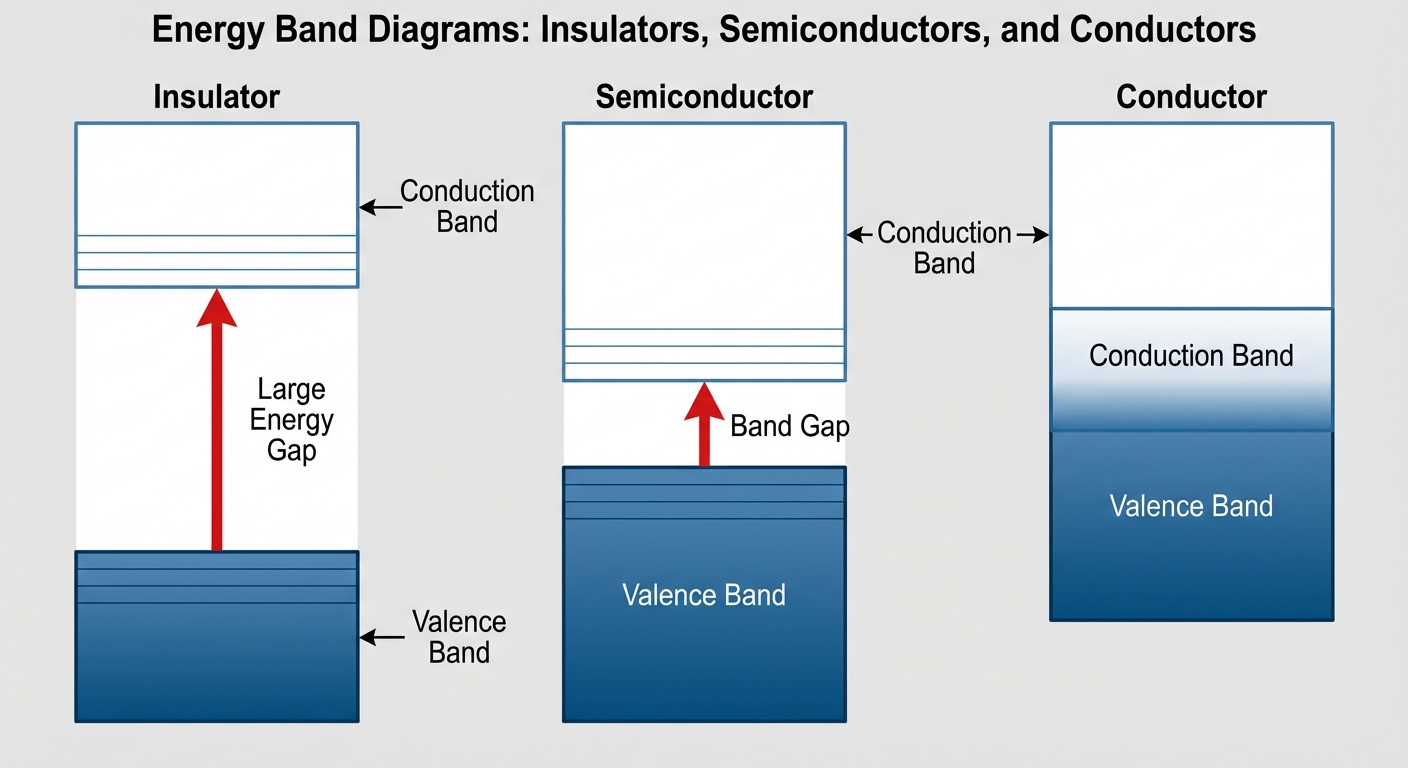
- Valence Band: The lower energy band containing electrons bound to the atom (involved in bonding).
- Conduction Band: The higher energy band where electrons are free to move and conduct electricity.
- Band Gap ($E_g$): The energy difference between the valence and conduction bands. No electrons can exist here.
| Material | Band Gap Characteristics |
|---|---|
| Conductor (Metals) | Valence and Conduction bands overlap. Electrons move freely. |
| Insulator | Large Band Gap ($> 5$ eV). Electrons cannot jump to the conduction band easily. |
| Semiconductor | Small Band Gap ($\approx 1$ eV for Silicon). Electrons can jump the gap if given a small amount of energy (heat or light). |
Doping: N-Type and P-Type
Pure silicon is not a great conductor. We improve it by doping—adding impurities to change its electrical properties.
- N-Type (Negative): Silicon is doped with an element with 5 valence electrons (like Phosphorus).
- Provides extra free electrons in the conduction band.
- Majority carriers: Electrons.
- P-Type (Positive): Silicon is doped with an element with 3 valence electrons (like Boron).
- Creates "Holes" (missing electrons) in the valence band. Holes act like positive charge carriers.
- Majority carriers: Holes.
Crucial Concept: Doped semiconductors are electrically neutral. An N-type crystal has extra moveable electrons, but the donor nuclei have extra protons that balance the charge exactly.
The P-N Junction and Applications
When P-type and N-type materials are joined, they form a P-N Junction. Electrons from the N-side fill holes on the P-side near the boundary, creating a non-conductive Depletion Coordinate and an internal electric field.
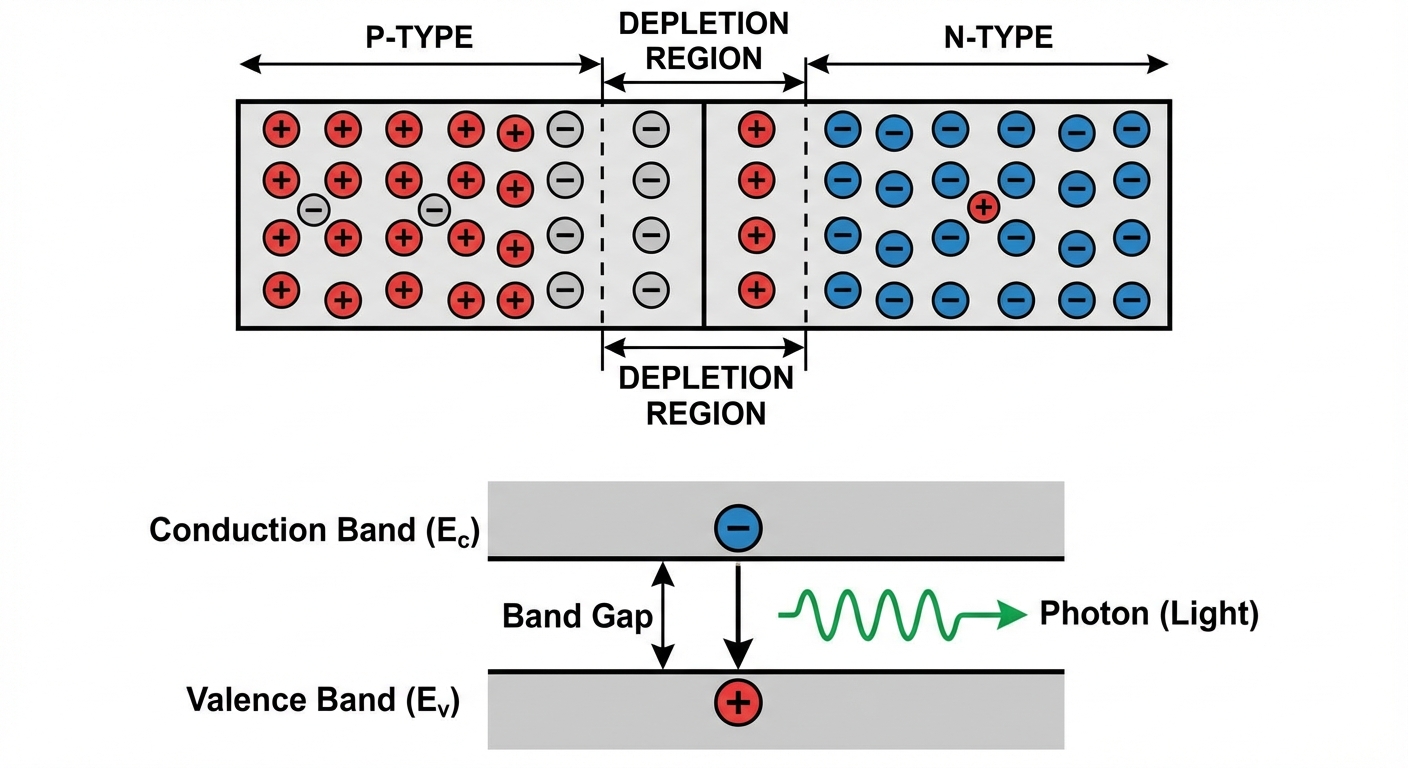
1. Light Emitting Diodes (LEDs)
When current flows through a P-N junction (forward bias), electrons from the conduction band drop into holes in the valence band.
- The energy lost by the electron is emitted as a photon.
- The color depends on the Band Gap energy ($E_g$).
- $h$: Planck's constant ($6.63 \times 10^{-34} \text{ J}\cdot\text{s}$)
- $c$: Speed of light
- $\lambda$: Wavelength of light
2. Photovoltaic Cells (Solar Panels)
This is the reverse of an LED. Light hits the P-N junction. If the photon energy $hf$ is greater than the band gap $E_g$:
- The photon is absorbed.
- An electron is knocked from the valence band to the conduction band (creating an electron-hole pair).
- The internal electric field pushes the electron to the N-side and the hole to the P-side, creating a voltage/current.
Common Mistakes & Pitfalls
Fundamentally Wrong Particles:
- Mistake: Thinking protons and neutrons are fundamental particles.
- Correction: Protons and neutrons are composite particles (Baryons) made of quarks. Electrons are fundamental.
Missing the Neutrino:
- Mistake: Writing beta decay as $n \rightarrow p + e^-$.
- Correction: This violates lepton conservation. You must include the antineutrino ($\bar{\nu}_e$).
Charged Semiconductors:
- Mistake: Thinking N-type material is negatively charged and P-type is positively charged.
- Correction: They are neutral. "N-type" just refers to the sign of the charge carrier (negative electrons) that is free to move, not the net charge of the material.
Blue vs. Red LEDs:
- Mistake: Thinking Red LEDs have a higher band gap.
- Correction: $E = hf$. Blue light has a higher frequency (and energy) than red light. Therefore, Blue LEDs require a semiconductor with a larger band gap than Red LEDs.