Chemical Reactions
1/36
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
37 Terms
chemical reaction
produce new substances by changing the way atoms are arranged
*break and make bonds
signs that a chemical reaction has occurred
color change, change in temperature, production of gas (bubbles), and production of a solid
color change example
rusty nail
temperature change
endothermic- heat added, exothermic- heat released
gas produces
bubbling ex. alkaseltzer
production of a solid (a precipitate)
a solid is produced, no heat was removed, so it is not a phase change
three main types of chemical reactions
synthesis (creating something), decomposition, and combustion
describe what happens to atoms involved in a chemical reaction
-bonds break in the reactants and new bonds form in the products
-the reactants and product has the same atoms
-then the atoms are rearranged
Tina made a volcano by pouring vinegar over a "mountain" of baking soda. The wet baking soda bubbled and foamed. Did a chemical reaction occur?
It is a chemical reaction since gas was produced.
Equilibrium
A state of balance
All chemical reactions involve _________________________________.
energy
Energy is used to _________________________ bonds in reactants.
break
Energy is ___________________________ when new bonds form in products.
released
exothermic reactions
release energy
endothermic reactions
absorb energy
law of conservation of energy
Energy cannot be created or destroyed
If energy cannot be destroyed during a chemical reaction, what happens to the energy that is absorbed in an endothermic reaction?
stored in the bonds of the products as chemical energy
What happens to the excess energy in the reactants of an exothermic energy?
When the reaction happens, the excess energy is released.
endothermic reaction
products have more stored chemical energy than the reactants
excess energy is released to the surrounding when the reactions occur
-example home heating system-energy is released during combustion in the furnace is used to heat the home
exothermic reactions
products have less stored chemical energy than the reactants
pure substances
single kind of matter and has specific set of properties
single kind of matter
elements and compounds
specific set of properties
chemical properties and physical properties
chemical properties
flammability, reactivity (oxidation), radioactivity, combustion, toxicity, new substance created, new properties
physical properties
texture, color, odor, density, luster, flexibility, solubility, physical state, conduction (heat/electric)
chemical reactions
matter, substance, compounds, physical and chemical properties
states of matter
solid, liquid, gas, phase change, melting, freezing, vaporization, condensation
heat/thermal
temperature, Celsius, heat, kinetic, molecular motion, thermal energy, mass
subscript
A number in a chemical formula that tells the number of atoms in a molecule
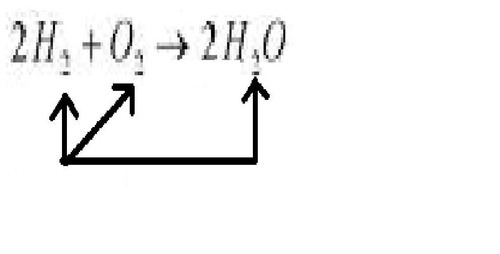
chemical equation
a way to describe a chemical reaction using chemical formulas and other symbols
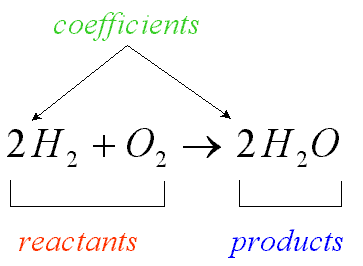
reactants
substance that present at the beginning of a chemical reaction
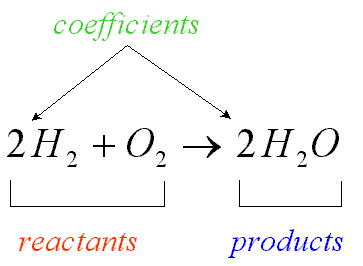
products
substances formed (produced) by the chemical reaction
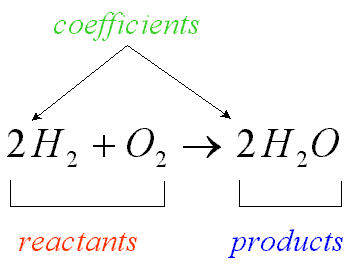
yield sign
the arrow in the middle of a chemical equation that separates reactants and products
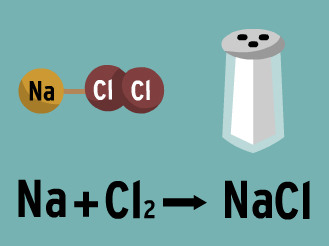
coefficient
A number in front of a chemical formula in an equation that indicates how many molecules or atoms of each reactant and product are involved in a reaction.
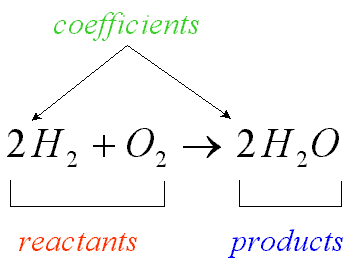
chemical symbol
A one or two letter representation of an element
precipitate
A solid that forms from a solution during a chemical reaction.