0Days
0Hours
0Minutes
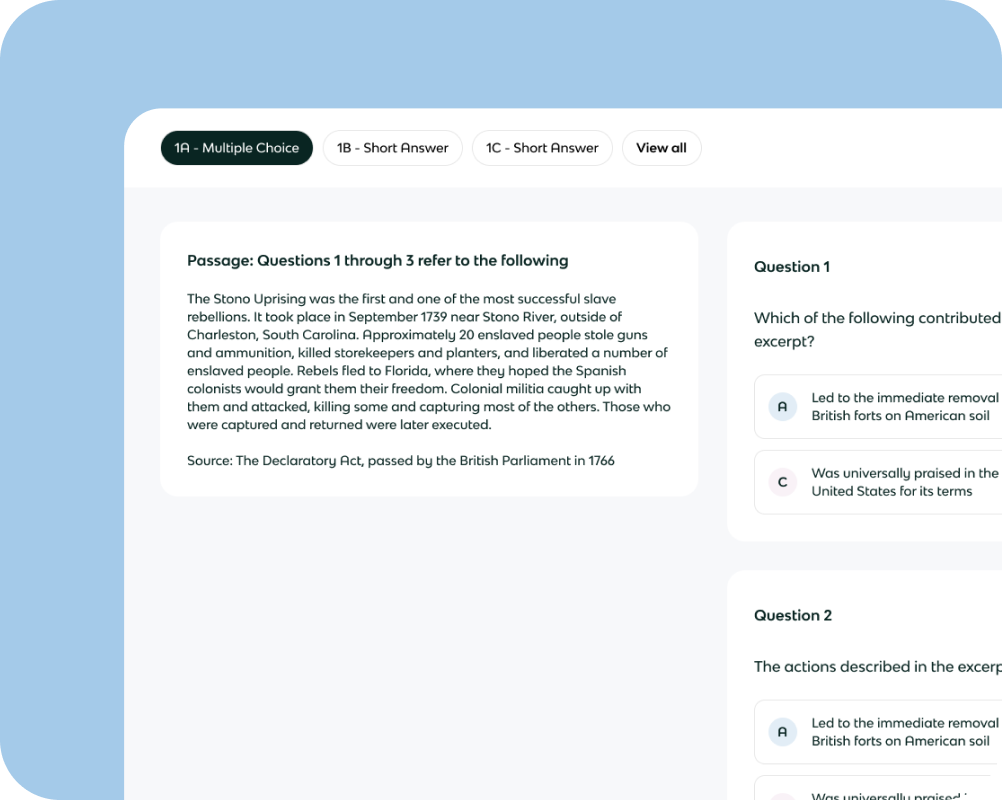
Stoichiometric Relationships
Prepared with
Atomic Structure
Prepared with
Periodicity
Prepared with
Chemical Bonding and Structure
Prepared with
Energetics/Thermochemistry
Prepared with
Chemical Kinetics
Prepared with
Equilibrium
Prepared with
Acids and Bases
Prepared with
Redox Processes
Prepared with
Organic Chemistry
Prepared with
Measurement and Data Processing
Prepared with
Atomic Structure
Prepared with
The Periodic Table - the transition metals
Prepared with
Chemical Bonding and Structure
Prepared with
Energetics/thermochemistry
Prepared with
Chemical kinetics
Prepared with
Equilibrium
Prepared with
Acids and bases
Prepared with
Redox processes
Prepared with
Organic chemistry
Prepared with
Materials
Prepared with
Biochemistry
Prepared with
Energy
Prepared with
Medicinal chemistry
Prepared with