Unit 6: Fundamentals of Thermodynamics and Energy
Endothermic and Exothermic Processes
Thermodynamics begins with the fundamental relationship between chemical reactions and energy flow. In AP Chemistry, the universe is divided into two distinct parts: the System (the chemical reaction or phase change being studied) and the Surroundings (everything else, including the container, the solvent, and the thermometer).
Direction of Heat Flow ($q$)
The First Law of Thermodynamics states that energy is conserved; it cannot be created or destroyed, only transferred. This transfer of thermal energy is denoted as heat ($q$).
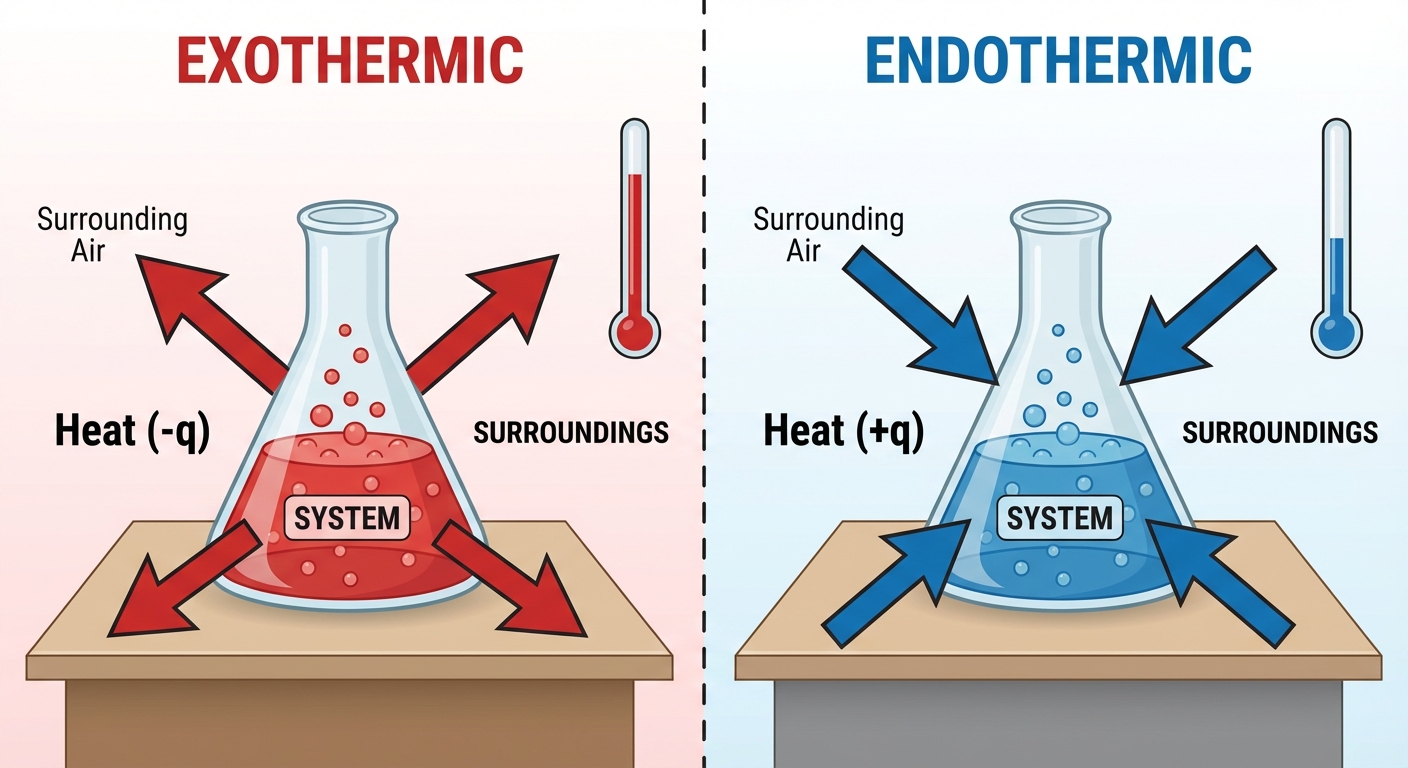
Exothermic Process: Heat flows out of the system into the surroundings.
- The system loses energy.
- The surroundings get hotter (temperature increases).
- Sign Convention: $q$ is negative ($q < 0$).
- Example: Combustion reactions, hand warmers, freezing water.
Endothermic Process: Heat flows into the system from the surroundings.
- The system gains energy.
- The surroundings get cooler (temperature decreases).
- Sign Convention: $q$ is positive ($q > 0$).
- Example: Ice melting, photosynthesis, instant cold packs.
Bond Energy and Enthalpy
A common misconception is that breaking bonds releases energy. This is false.
- Breaking Bonds: Requires energy (Endothermic). You must put energy in to overcome the attraction between atoms.
- Forming Bonds: Releases energy (Exothermic). Atoms move to a lower, more stable energy state.
The overall enthalpy change ($\Delta H$) of a reaction depends on the balance between these two steps:
Energy Diagrams
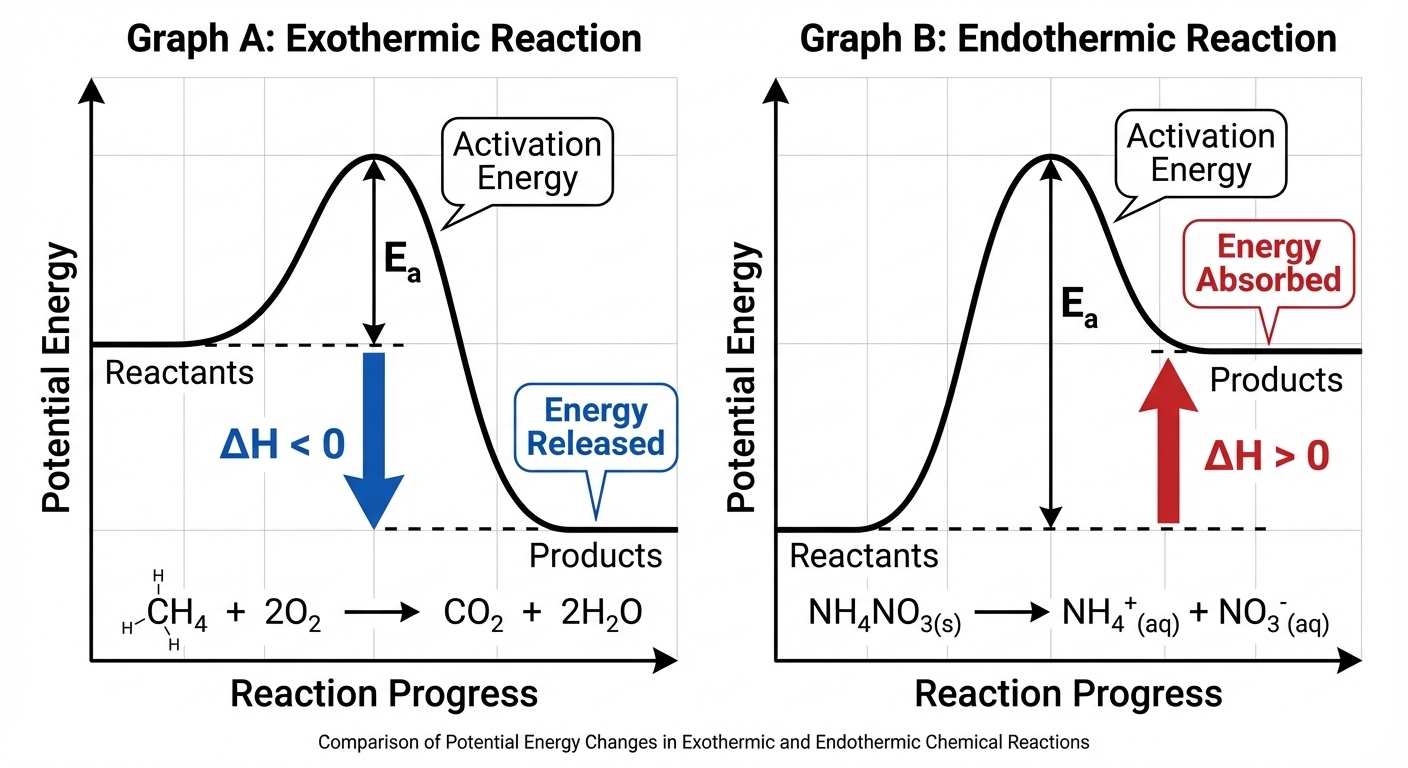
Potential energy diagrams (or reaction profiles) visualize the energy changes during a reaction. The y-axis represents Potential Energy (PE), and the x-axis represents the Reaction Coordinate (progress).
Key Features of the Diagram
- Reactants & Products: Plotted as horizontal lines at their respective energy levels.
- Activation Energy ($E_a$): The energy barrier that must be overcome for the reaction to occur. It is the energy difference between the reactants and the Transition State (the peak of the curve).
- Enthalpy Change ($\Delta H$): The difference in potential energy between products and reactants.
Visual Differences
- Exothermic: The product line is lower than the reactant line. Energy is released. \Delta H < 0
- Endothermic: The product line is higher than the reactant line. Energy is absorbed. \Delta H > 0
Heat Transfer and Thermal Equilibrium
Heat is the transfer of energy due to a temperature difference. It describes the process, not a property possessed by an object (objects possess internal energy, not "heat").
Kinetic Molecular Theory of Heat
On a microscopic level, temperature is a measure of the average kinetic energy (KE) of the particles.
- Direction: Heat naturally flows from matter with higher average KE (higher Temperature) to matter with lower average KE (lower Temperature).
- Collisions: Fast-moving particles collide with slow-moving particles, transferring momentum and energy.
Thermal Equilibrium
When two objects at different temperatures are in contact, heat transfer continues until they reach the same temperature. At Thermal Equilibrium:
- Specific temperatures are equal: $T{final, 1} = T{final, 2}$.
- Average Kinetic Energy of particles in both objects is equal.
- According to the Law of Conservation of Energy (assuming an isolated system):
Heat Capacity and Calorimetry
Calorimetry is the experimental technique used to measure heat transfer.
Specific Heat Capacity ($c$)
Specific Heat Capacity is the amount of energy required to raise the temperature of 1 gram of a substance by 1 degree Celsius (or 1 Kelvin).
- Symbol: $c$ or sometimes $s$
- Units: $J/(g\cdot ^\circ C)$ or $J/(g\cdot K)$
- Meaning: High specific heat (like water) means a substance resists temperature changes. Low specific heat (like metals) means temperature changes rapidly.
The Calorimetry Equation
Use this formula when a substance is changing temperature without changing phase:
Where:
- $q$ = heat energy (Joules)
- $m$ = mass (grams)
- $c$ = specific heat capacity
- $\Delta T$ = change in temperature ($T{final} - T{initial}$)
Worked Example: Coffee Cup Calorimetry
Scenario: A 50.0 g piece of metal at 100.0 $^{\circ}$C is dropped into 100.0 g of water at 22.0 $^{\circ}$C inside an insulated coffee cup. The final temperature of the mixture is 28.0 $^{\circ}$C. Calculate the specific heat of the metal ($c{metal}$). Use $c{water} = 4.18 \; J/g^{\circ}C$.
Solution:
- Calculate heat gained by water ($q{water}$):
- Apply Conservation of Energy:
- Solve for $c{metal}$:
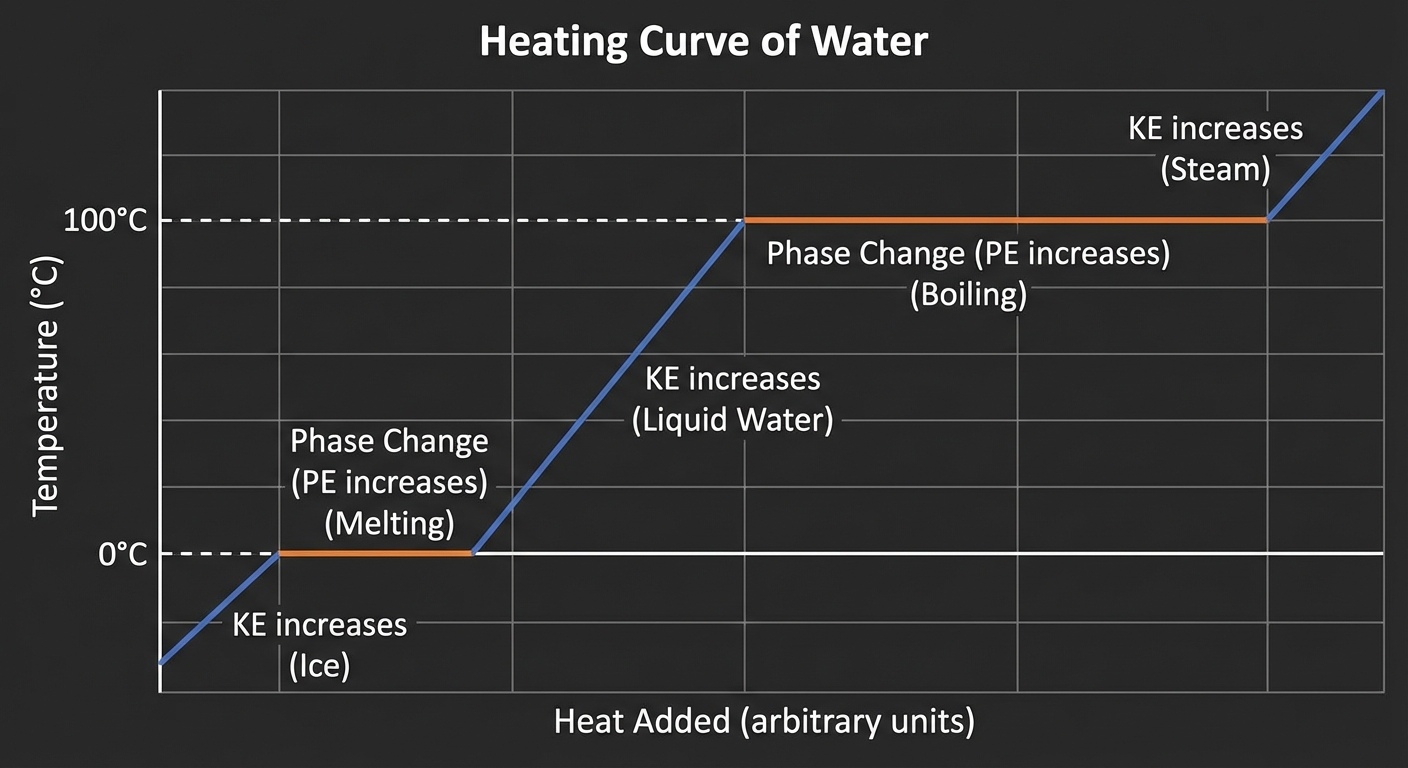
Energy of Phase Changes
Temperature does not change during a phase change. The energy added goes toward increasing the potential energy (overcoming intermolecular forces) rather than kinetic energy.
Enthalpy of Fusion and Vaporization
To calculate heat during a phase change, use appropriate enthalpy values:
- Heat of Fusion ($\Delta H_{fus}$): Energy to melt solid to liquid (or freeze liquid to solid).
- Heat of Vaporization ($\Delta H_{vap}$): Energy to boil liquid to gas (or condense gas to liquid).
Formulas:
Heating Curves
A heating curve plots Temperature vs. Heat Added (or Time).
- Sloped Lines: Single phase exists. Temperature changes. Kinetic energy increases. Use $q=mc\Delta T$.
- Flat Lines (Plateaus): Phase change occurs (equilibrium between two phases). Temperature is constant. Potential energy increases. Use $q=n\Delta H$.
Common Mistakes & Pitfalls
- Mixing up System and Surroundings: Always identify what the "system" is. In a calorimetry problem, the water is usually the surroundings. If the water gets hot ($+q$), the reaction was exothermic ($-q$).
- Units of Energy: $\Delta H$ values are usually given in kJ/mol, while Specific Heat calculations usually result in Joules. You must convert one to match the other (divide J by 1000 to get kJ).
- Using $q=mc\Delta T$ during a Phase Change: If the problem mentions melting or boiling, $\Delta T = 0$. You cannot use the specific heat formula. You must use $\Delta H{fus}$ or $\Delta H{vap}$.
- Bond Breaking signs: Students often think breaking bonds releases energy (like an explosion). In chemistry, input of energy is always required to break a bond. The explosion energy comes from the subsequent formation of stronger bonds.
- Confusing Heat and Temperature: Temperature is average kinetic energy; Heat is the transfer of energy. Objects do not "have" heat.