Mastering Chemical Reactions and Equations
Mastering Chemical Reactions and Equations
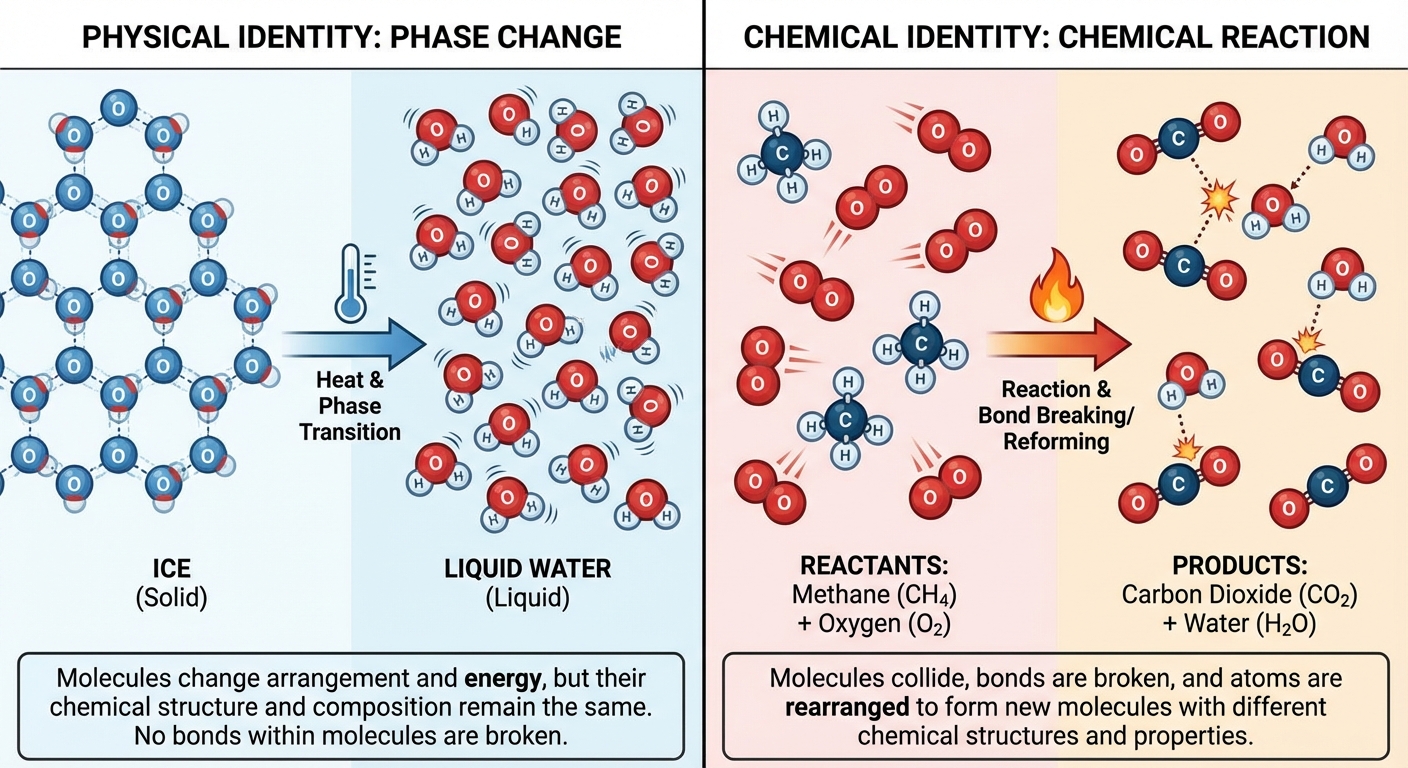
Physical and Chemical Changes
Before we can represent reactions, we must distinguish between changes that alter the identity of a substance and those that do not. In AP Chemistry, this distinction is often assessed through data, observations, or particulate models.
Defining the Distinction
The primary difference lies in the nature of the bonds or forces being overcome.
Physical Change: A change in the properties of a substance without observing a change in its chemical composition.
- Process: Generally involves overcoming Intermolecular Forces (IMFs) or separating components of a mixture.
- Examples: Phase changes (melting, boiling, sublimation), formation of mixtures, separation processes (distillation, filtration, chromatography).
- Equation view: $H2O(s) \rightarrow H2O(l)$. Notice the formula remains the same.
Chemical Change: A process where one or more substances are converted into different substances with different characteristic properties.
- Process: Involves the breaking and forming of Intramolecular bonds (covalent or ionic bonds).
- Examples: Combustion, oxidation (rusting), neutralization, precipitation, decomposition.
- Equation view: $2H2O(l) \rightarrow 2H2(g) + O_2(g)$. New formulas are created.
Evidence of Chemical Change
While we cannot see atoms rearranging with the naked eye, macroscopic evidence suggests a chemical reaction has occurred:
- Production of a precipitate (mixing two liquids creates a solid).
- Color change (unexpected changes, not just dilution).
- Evolution of gas (bubbling/fizzing without external heating).
- Temperature change (transfer of heat energy, either exothermic or endothermic).
The "Gray Area": Dissolution
Is dissolving salt in water physical or chemical?
- Argument for Physical: The water can be evaporated to recover the salt unchanged.
- Argument for Chemical: Ionic bonds in the lattice are broken; ion-dipole interactions form.
- AP Verdict: Dissolution is usually classified as a physical change solely because the substance (the salt) retains its chemical identity, but you should acknowledge the thermodynamic processes (bond breaking/forming) involved.
Introduction for Reactions & Balancing
Chemical equations are the sentences of chemistry. They describe exactly what happens during a reaction.
The Law of Conservation of Mass
Matter is neither created nor destroyed. Therefore, a chemical equation must be balanced; the number of atoms of each element on the reactant side must equal the number of atoms on the product side.
Anatomy of an Equation
- Reactants: Left side of the arrow.
- Products: Right side of the arrow.
- Coefficients: Large numbers in front (e.g., the "2" in $2Al$). These represent the mole ratio.
- Subscripts: Small numbers (e.g., the "3" in $AlCl_3$). These determine chemical identity and cannot be changed when balancing.
- States of Matter: Crucial for Net Ionic Equations.
- $(s)$ Solid / Precipitate
- $(l)$ Liquid (pure, e.g., water)
- $(g)$ Gas
- $(aq)$ Aqueous (dissolved in water)
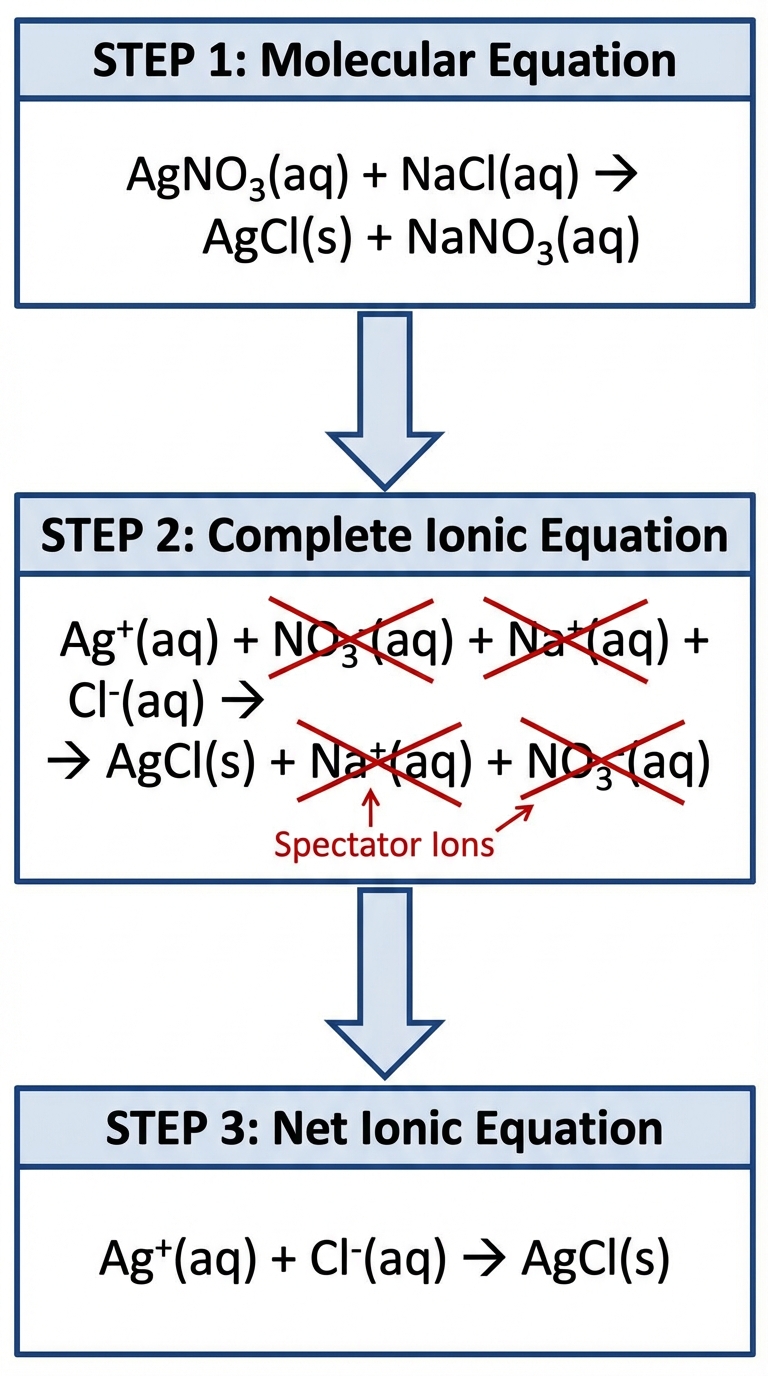
Net Ionic Equations
This is a high-frequency topic on the AP Exam. You must be able to translate a molecular equation into a net ionic equation to show only the species actually participating in the reaction.
The Three Levels of Equations
Let's assume we mix solutions of Lead(II) Nitrate and Potassium Iodide.
Molecular Equation: Shows entire compounds.
Complete Ionic Equation: Shows all soluble ionic compounds dissociated into ions.
Net Ionic Equation: Removes Spectator Ions (ions that appear unchanged on both sides).
The Golden Rules for Dissociation
When moving from Molecular to Complete Ionic, what do you split apart?
| SPLIT IT (Strong Electrolytes) | KEEP IT TOGETHER |
|---|---|
| Soluble Salts (Check solubility rules) | Solids $(s)$, Precipitates |
| Strong Acids ($HCl, HBr, HI, HNO3, H2SO4, HClO4$) | Liquids $(l)$ (e.g., $H_2O$) |
| Strong Bases (Group 1 hydroxides, Sr/Ba/Ca hydroxides) | Gases $(g)$ |
| Weak Acids/Bases (e.g., $HF, HC2H3O_2$) |
Note Example: The reaction of acetic acid with sodium hydroxide.
- Incorrect: $H^+ + OH^- \rightarrow H_2O$ (Acetic acid is weak!)
- Correct: $HC2H3O2(aq) + OH^-(aq) \rightarrow C2H3O2^-(aq) + H_2O(l)$
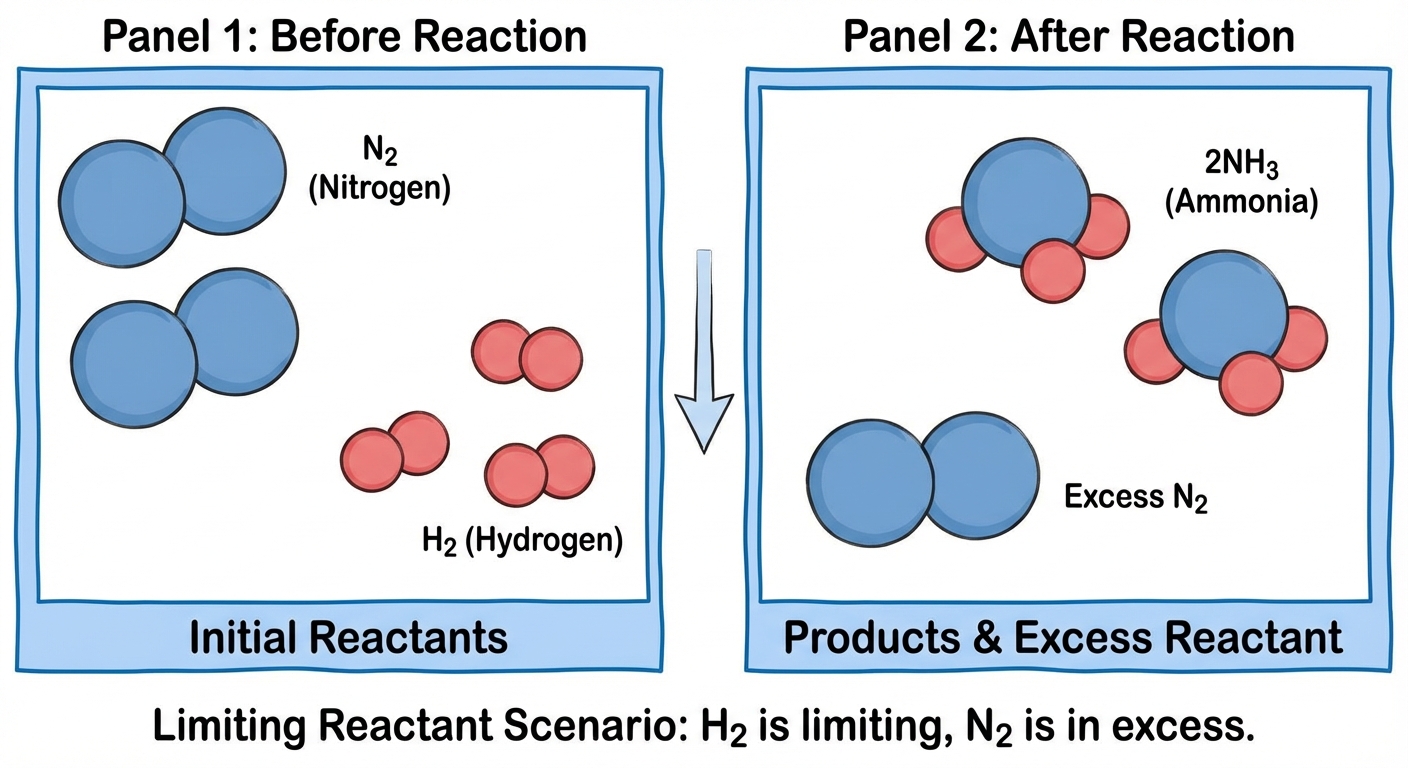
Representations of Reactions (Particulate Models)
AP questions frequently ask you to draw or analyze "particle diagrams." These are visual representations of what occurs at the atomic level.
Critical Elements to Analyze
When drawing or interpreting these diagrams, check these three factors:
Conservation of Mass:
Count the atoms. If you start with 4 nitrogen atoms, you must end with 4 nitrogen atoms, even if they are bonded differently.Limiting Reactant vs. Excess:
Reactions rarely happen in perfect stoichiometric ratios.- If the ratio is 1:1 and you have 3 particles of A and 5 particles of B, then A is the limiting reactant.
- Vital: The final diagram MUST show the product and the leftover excess reactant.
States of Matter & Density:
- Gas: Particles far apart, filling the box.
- Liquid: Particles touching but disordered.
- Solid: Particles packed typically in a regular array.
Tips for Drawing
- Use a legend (e.g., Open circle = Hydrogen, Filled circle = Chlorine).
- Ensure diatomic elements exist as pairs when unbonded ($O2, H2$, etc.).
- If drawing a soluble salt (e.g., $NaCl$), draw the ions separated and surrounded by water molecules (if requested), oriented according to polarity.
Common Mistakes & Pitfalls
- Splitting Weak Acids: Students often write $H^+$ for acetic acid or hydrofluoric acid in net ionic equations. Don't. If it's a weak acid, write the whole formula.
- Ignoring Charge Balance: A net ionic equation must generally balance mass AND charge.
- Wrong/Unbalanced: $Fe^{3+} + e^- \rightarrow Fe^{2+}$ (Mass balanced, charge is +3 vs +2).
- Correct: $Fe^{3+} + e^- \rightarrow Fe^{2+}$ (Wait, the first one was unbalanced in charge logic).
- Real Example: $Cu(s) + Ag^+(aq) \rightarrow Cu^{2+}(aq) + Ag(s)$. Mass is balanced. Charge is (+1) $\to$ (+2). Incorrect.
- Corrected: $Cu(s) + 2Ag^+(aq) \rightarrow Cu^{2+}(aq) + 2Ag(s)$.
- State Symbol Negligence: Omitting $(aq)$ or $(s)$ prevents you from knowing what splits and what doesn't. Always write them down.
- Misinterpreting "Boiling": Seeing bubbles when water boils is not a chemical change; no new substance is formed, only the phase changes. Don't confuse physical changes with chemical gas evolution.