AP Biology Unit 1: The Chemistry of Macromolecules
Introduction: Monomers, Polymers, and Chemical Reactions
Biological systems rely on four main classes of large biological molecules, known as macromolecules: carbohydrates, lipids, proteins, and nucleic acids. With the exception of lipids, these macromolecules are chain-like molecules called polymers.
A polymer is a long molecule consisting of many similar or identical building blocks linked by covalent bonds. The repeating units that serve as the building blocks of a polymer are smaller molecules called monomers.
Synthesis and Breakdown Mechanisms
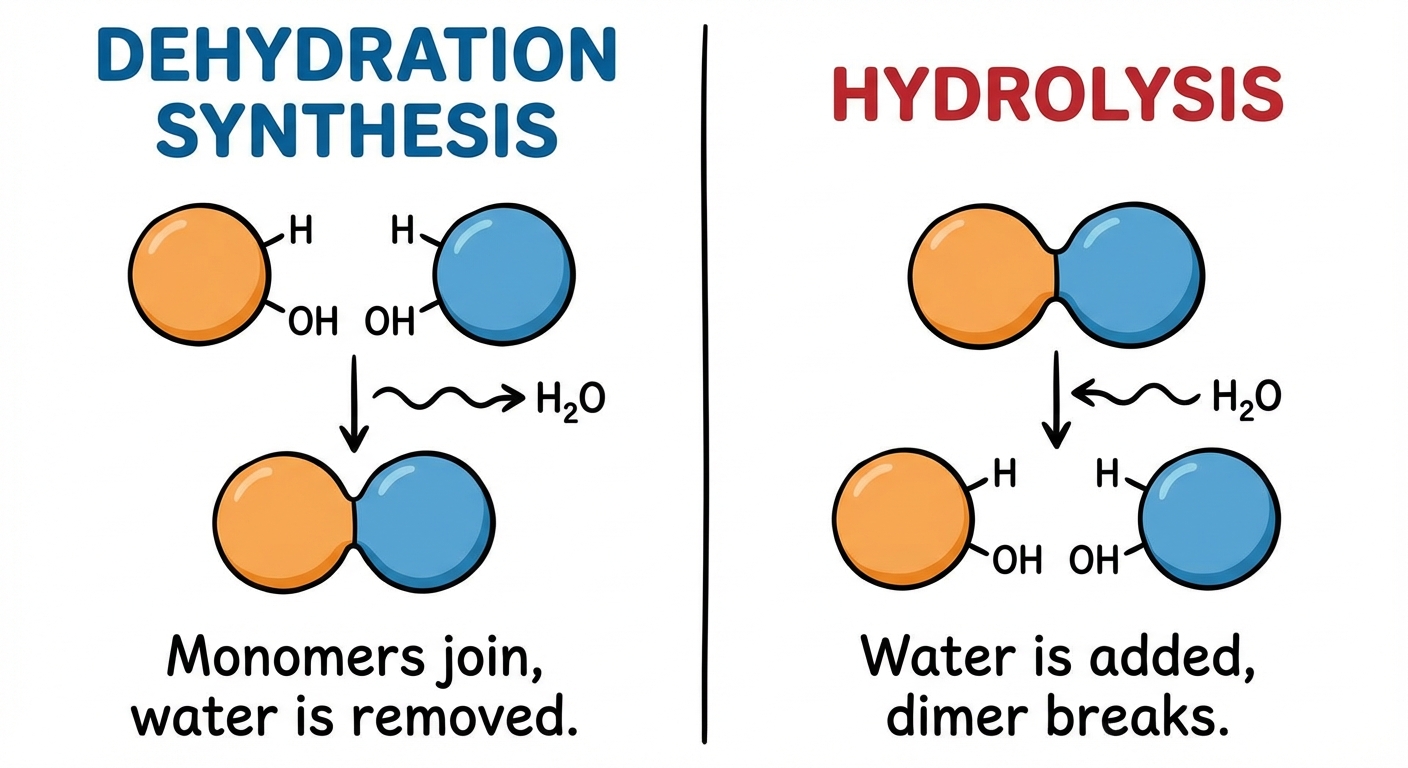
Life builds up and breaks down these molecules using two specific chemical reactions facilitated by enzymes. Understanding the role of water in these reactions is critical for the AP Biology exam.
Dehydration Synthesis (Condensation Reaction):
- Function: Connects monomers to create polymers.
- Mechanism: A reaction in which two molecules are covalently bonded to each other with the loss of a water molecule ($H_2O$). One monomer provides a hydroxyl group ($-OH$), while the other provides a hydrogen ($-H$).
- Result: New covalent bond formation and water is released as a byproduct.
Hydrolysis (Greek hydro = water, lysis = break):
- Function: Breaks down polymers into monomers (e.g., digestion).
- Mechanism: The bond between monomers is broken by the addition of a water molecule. A hydrogen from water attaches to one monomer and the hydroxyl group attaches to the other.
- Result: Covalent bond breakage.
Carbohydrates: Fuel and Building Material
Carbohydrates include sugars and polymers of sugars. They consist of Carbon, Hydrogen, and Oxygen, generally in a ratio of $1:2:1$ ($CH_2O$).
Structure and Classification
- Monosaccharides: The simplest sugars (monomers). Most common is glucose ($C6H{12}O_6$). They serve as major fuel for cells and raw material for building molecules.
- Disaccharides: Two monosaccharides joined by a glycosidic linkage (a covalent bond formed via dehydration synthesis).
- Polysaccharides: The polymers of sugars, which have storage and structural roles.
Storage vs. Structural Polysaccharides
The function of a polysaccharide is determined by its sugar monomers and the positions of its glycosidic linkages. This usually depends on whether the glucose monomers are in the $\alpha$ (alpha) or $\beta$ (beta) ring form.
| Type | Name | Structure | Function |
|---|---|---|---|
| Storage | Starch | Polymer of $\alpha$-glucose monomers within plants. | Plant energy storage. Plants store surplus starch as granules. |
| Storage | Glycogen | Highly branched polymer of glucose in animals. | Animal energy storage (liver and muscle cells). |
| Structural | Cellulose | Polymer of $\beta$-glucose monomers. Linear and unbranched. | Major component of rapid plant cell walls. Hydrogen bonds between parallel strands make it tough. |
| Structural | Chitin | Carbohydrate with nitrogen-containing appendage. | Exoskeletons of arthropods and fungal cell walls. |
Lipids: Hydrophobic Molecules
Lipids are the one class of large biological molecules that does not include true polymers. They are grouped together because they share one important trait: they mix poorly, if at all, with water (hydrophobic). They consist mostly of hydrocarbon regions.
Fats (Triglycerides)
A fat is constructed from two types of smaller molecules: glycerol and fatty acids.
- Linkage: Three fatty acids are joined to glycerol by an ester linkage, creating a triacylglycerol.
Saturation Types:
- Saturated Fatty Acids: Have no double bonds between carbon atoms composing a chain (saturated with Hydrogen). They are solid at room temperature (e.g., butter).
- Unsaturated Fatty Acids: Have one or more double bonds ($C=C$), usually creating a cis double bond which creates a "kink" or bend in the chain. They are liquid at room temperature (e.g., olive oil) because the kinks prevent packing tightly.
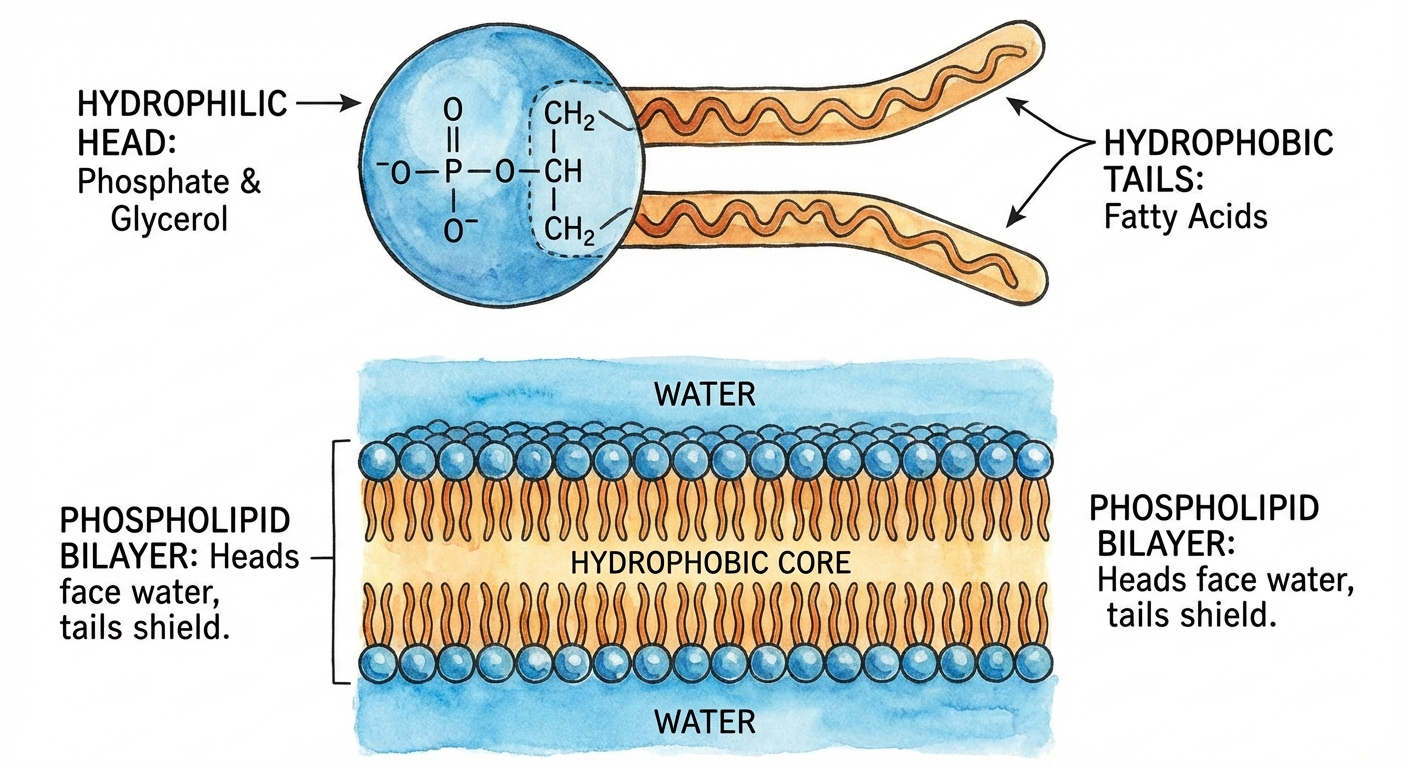
Phospholipids
Phospholipids are essential for cells because they are major constituents of cell membranes.
- Structure: Attachment of two fatty acids and a phosphate group to glycerol.
- Properties: They are amphipathic, meaning they have both a hydrophilic region and a hydrophobic region.
- Heads: Hydrophilic (attracted to water), polar.
- Tails: Hydrophobic (repelled by water), nonpolar.
When added to water, they self-assemble into a bilayer, shielding their hydrophobic tails from water. This forms the boundary between the cell and its external environment.
Steroids
Lipids characterized by a carbon skeleton consisting of four fused rings. Cholesterol is a crucial steroid in animals, acting as a common component of cell membranes and a precursor for other steroids (like sex hormones).
Proteins: Diversity and Complexity
Proteins account for more than 50% of the dry mass of most cells and are instrumental in almost everything organisms do. Their functions include enzymatic catalysis, defense, storage, transport, hormonal regulation, and structural support.
Amino Acids
Proteins are polymers of amino acids. There are 20 different amino acids.
- Structure: An organic molecule with an $\alpha$-carbon in the center, bonded to:
- An Amino group ($-NH_2$)
- A Carboxyl group ($-COOH$)
- A Hydrogen atom
- A variable group, symbolized by R (the side chain).
The physical and chemical properties of the R-group determine the unique characteristics of a particular amino acid (e.g., acidic, basic, polar/hydrophilic, nonpolar/hydrophobic).
Peptide Bonds and Polypeptides
Amino acids are linked by covalent bonds called peptide bonds via dehydration synthesis (Carboxyl group of one reacts with Amino group of another). The polymer is called a polypeptide.
- Directionality: A polypeptide chain has a unique linear sequence. It has a free amino group at one end (the N-terminus) and a free carboxyl group at the opposite end (the C-terminus).
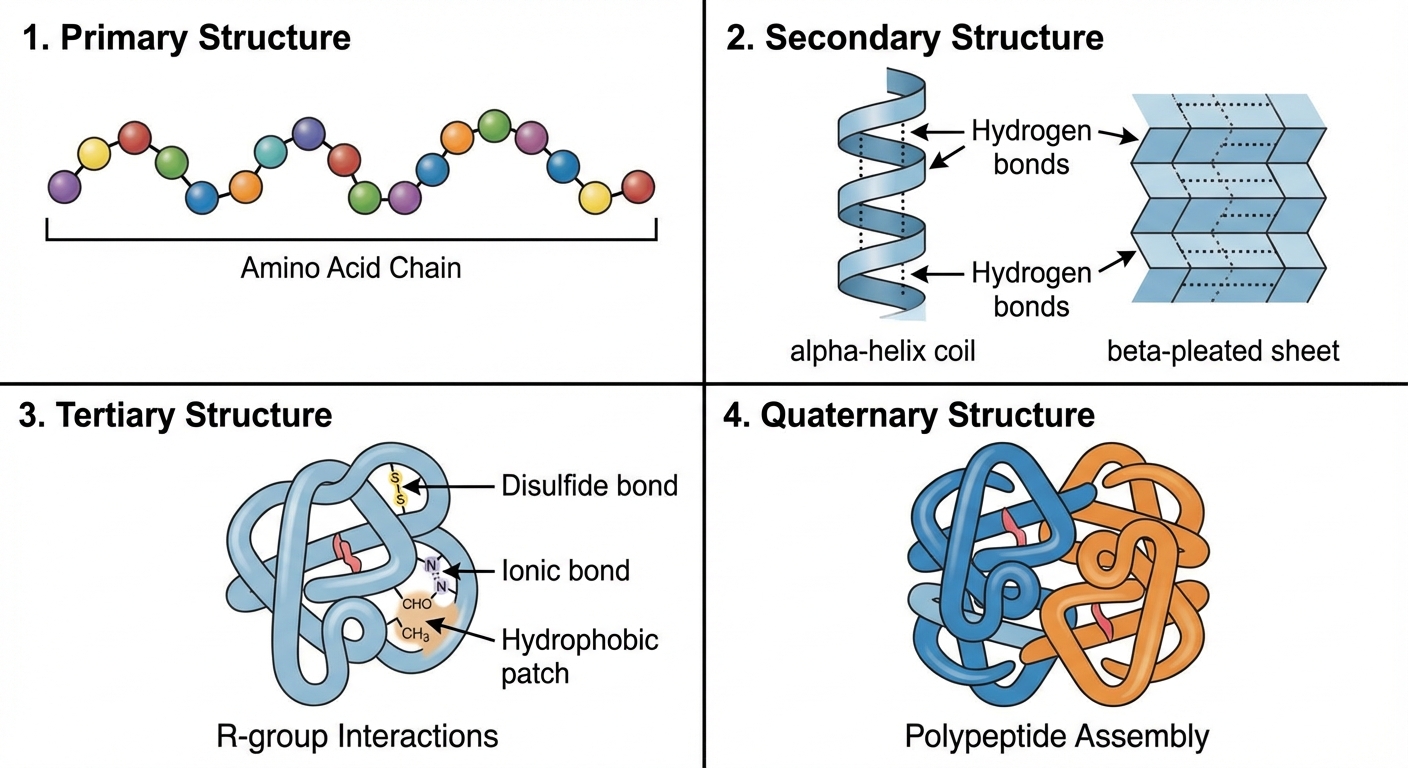
Levels of Protein Structure
A functional protein is not just a polypeptide chain, but one or more polypeptides strictly twisted, folded, and coiled into a molecule of unique shape.
- Primary Structure: The unique sequence of amino acids. Determined by inherited genetic information (DNA). Even a slight change here (mutation) can affect ability to function (e.g., Sickle-Cell Disease).
- Secondary Structure: Coils and folds in the polypeptide chain due to hydrogen bonding between the repeating constituents of the polypeptide backbone (not the side chains!).
- $\alpha$-helix (coil)
- $\beta$-pleated sheet (folds)
- Tertiary Structure: The overall shape of a polypeptide resulting from interactions between the side chains (R-groups) of the various amino acids. Interactions include:
- Hydrophobic interactions (nonpolar groups cluster inside)
- Van der Waals interactions
- Hydrogen bonds (between polar side chains)
- Ionic bonds (between +/- side chains)
- Disulfide bridges (covalent bonds between sulfurs in Cysteine monomers)
- Quaternary Structure: Arises when a protein consists of two or more polypeptide chains aggregated into one functional macromolecule (e.g., Hemoglobin).
Nucleic Acids: Information Store
Nucleic acids store, transmit, and help express hereditary information.
Nucleotides
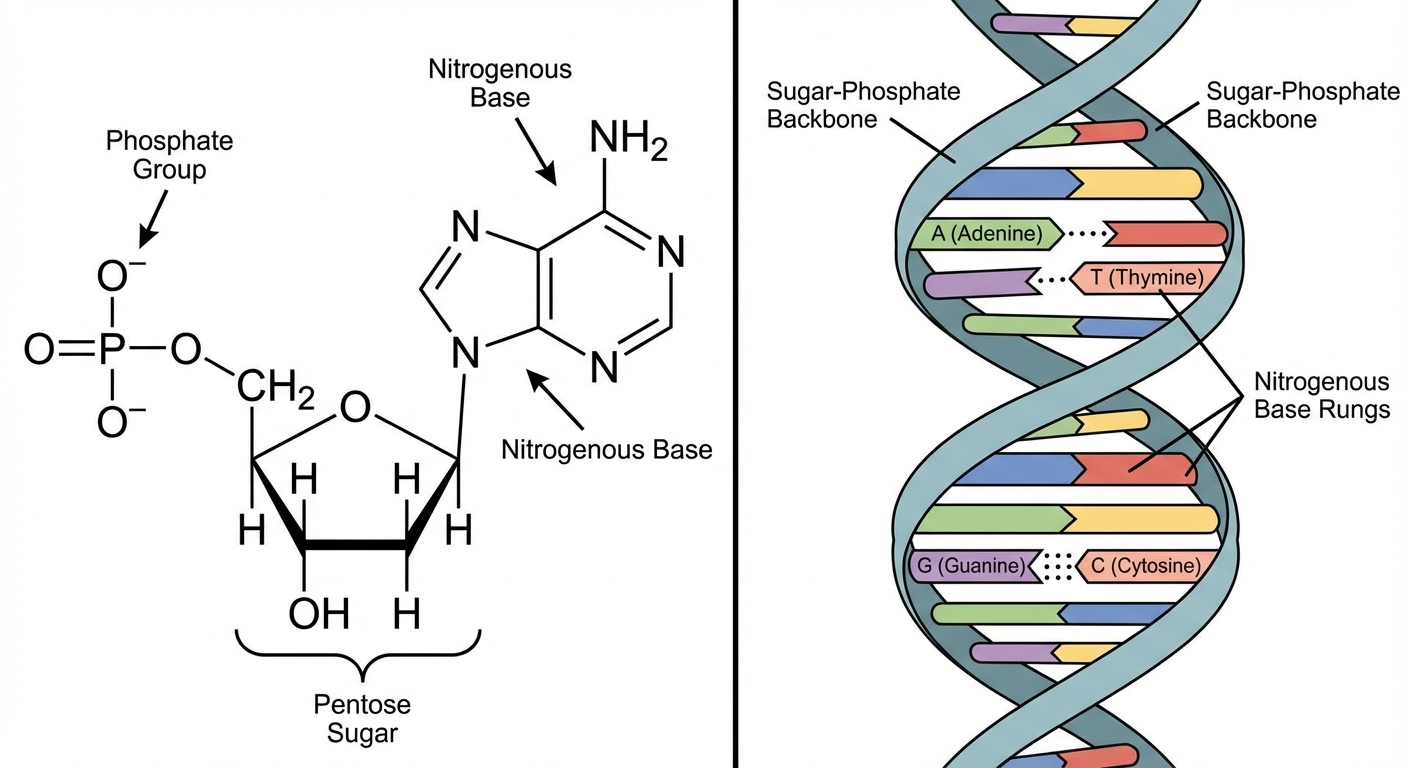
Nucleic acids are polymers called polynucleotides. Each polynucleotide is made of monomers called nucleotides.
Nucleotide Structure (3 parts):
- A five-carbon sugar (Pentose).
- A nitrogen-containing (nitrogenous) base.
- One or more phosphate groups.
DNA vs. RNA
| Feature | DNA (Deoxyribonucleic Acid) | RNA (Ribonucleic Acid) |
|---|---|---|
| Sugar | Deoxyribose (lacks oxygen at 2' carbon) | Ribose |
| Bases | Adenine (A), Guanine (G), Cytosine (C), Thymine (T) | Adenine (A), Guanine (G), Cytosine (C), Uracil (U) |
| Structure | Double helix; Antiparallel strands | Usually single-stranded |
| Function | Stores genetic information | Transmits genetic info; Catalytic roles |
Directionality and Bonding
- Phosphodiester Linkage: The bond connecting nucleotides. Formed between the phosphate group on the 5' carbon of one sugar and the -OH group on the 3' carbon of the neighboring sugar.
- 5' to 3' Direction: A DNA strand has built-in directionality along its sugar-phosphate backbone, from 5' to 3'.
- Base Pairing: In DNA, the two backbones run in opposite 5' $\rightarrow$ 3' directions (antiparallel). Interaction is specific:
- Adenine (A) pairs with Thymine (T) via 2 hydrogen bonds.
- Guanine (G) pairs with Cytosine (C) via 3 hydrogen bonds.
Common Mistakes and Pitfalls
- Confusing Hydrolysis and Dehydration Synthesis: Remember that "Hydro" means water and "lysis" means to break. If water is added to break a bond, it is hydrolysis. If water is removed to make a bond, it is dehydration synthesis.
- Bond Types: Students often confuse peptide bonds with glycosidic linkages.
- Peptide = Protein (Amino Acids)
- Glycosidic = Carbohydrate (Sugars)
- Ester = Lipids (Fatty Acids)
- Phosphodiester = Nucleic Acids
- Protein Folding Agents: A common error is assuming secondary structure involves R-groups. Secondary structure is purely hydrogen bonding in the backbone. Tertiary structure is where R-groups interact.
- Lipids as Polymers: Avoid calling lipids "polymers" in free-response questions (FRQs). While they are macromolecules, they are not composed of repeating monomer units in the same way proteins or carbs are.
- DNA Polarity: Neglecting the 3' and 5' ends. Many biological processes (replication, transcription) rely entirely on this directionality. Always check which end is which in diagrams.