APES Unit 6: Nonrenewable Energy Resources
Introduction to Energy Resources
Energy drives every ecological and economic system on Earth. In AP Environmental Science, understanding Unit 6: Energy Resources and Consumption requires you to analyze where our energy comes from, how we use it, and the consequences of that usage. The vast majority of the world's energy currently comes from nonrenewable resources—energy sources that exist in a fixed amount and cannot be easily replaced on a human time scale.
Renewable vs. Nonrenewable
- Nonrenewable Energy: Resources that exist in a finite quantity on Earth. Once consumed, they are gone for millions of years. Examples include Fossil Fuels (Coal, Oil, Natural Gas) and Nuclear Fuels (Uranium).
- Renewable Energy: Resources that can be replenished naturally at or near the rate of consumption (Solar, Wind, Geothermal, Hydroelectric).
- Potentially Renewable: Biomass (wood, charcoal) is renewable only if we do not consume it faster than it grows back.
Global Energy Consumption
The distribution of energy use is not uniform across the globe. It is heavily influenced by geography, availability of reserves, and a country's level of economic development.
1. Developed vs. Developing Nations
- Commercial Energy Sources: Energy sources that are bought and sold (e.g., coal, oil, natural gas, nuclear). These are heavily used in developed nations (US, Canada, Western Europe).
- Subsistence Energy Sources: Energy sources gathered by individuals for their own immediate needs. The most common is biomass (wood, charcoal, animal manure). These are heavily used in developing nations (rural areas of Africa, Asia, Latin America).
2. Trends in Consumption
As developing nations industrialize (increase in GDP), their demand for energy increases, triggering a shift from subsistence fuels to commercial fossil fuels. This increases efficiency but also drastically increases global carbon emissions.
Note on Efficiency: We often use the EROEI (Energy Return on Energy Investment) to measure efficiency.
A higher number is better. If it takes 1 unit of energy to extract 10 units of oil, the EROEI is 10.
Fuel Types and Uses
Fossil fuels are formed from ancient biological material buried under sediment and subjected to intense heat and pressure over millions of years.
1. Coal
Coal is a solid fuel formed primarily from the remains of trees, ferns, and other plant materials preserved 280-360 million years ago. It is the most abundant fossil fuel.
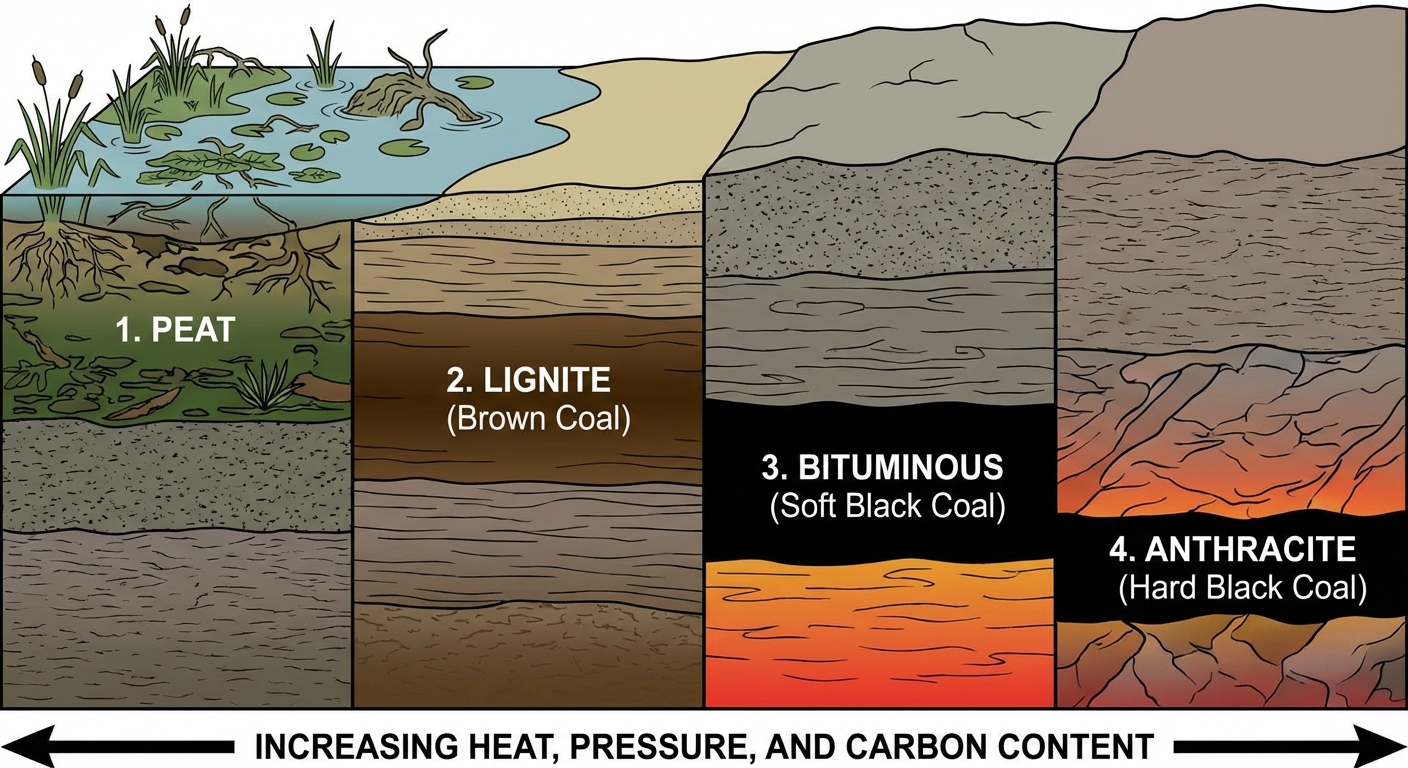
Types of Coal (Ranked by Age and Energy Density):
- Peat: Not yet coal. Partially decomposed organic matter. Low heat, high moisture. Found in bogs.
- Lignite (Brown Coal): Low heat content, low sulfur, limited supplies in most areas.
- Bituminous (Soft Coal): High heating capacity and high sulfur content. Extensively used for electricity generation.
- Anthracite (Hard Coal): The highest heating capacity and low sulfur content. It is the hardest, oldest, and Cleanest burning coal, but also the most expensive and rare.
Pros/Cons:
- Pros: Abundant, energy-dense, easy to transport, low upfront cost for power plants.
- Cons: Dirty combustion (, , Particulate Matter), mining destruction (strip mining), ash disposal issues.
2. Petroleum (Crude Oil)
Petroleum is a fluid mixture of hydrocarbons, water, and sulfur that occurs in underground deposits. It originated from ancient marine phytoplankton.
Refining:
Crude oil is useless until it is refined. It undergoes Fractional Distillation, where the oil is boiled and separated into different products based on their boiling points.
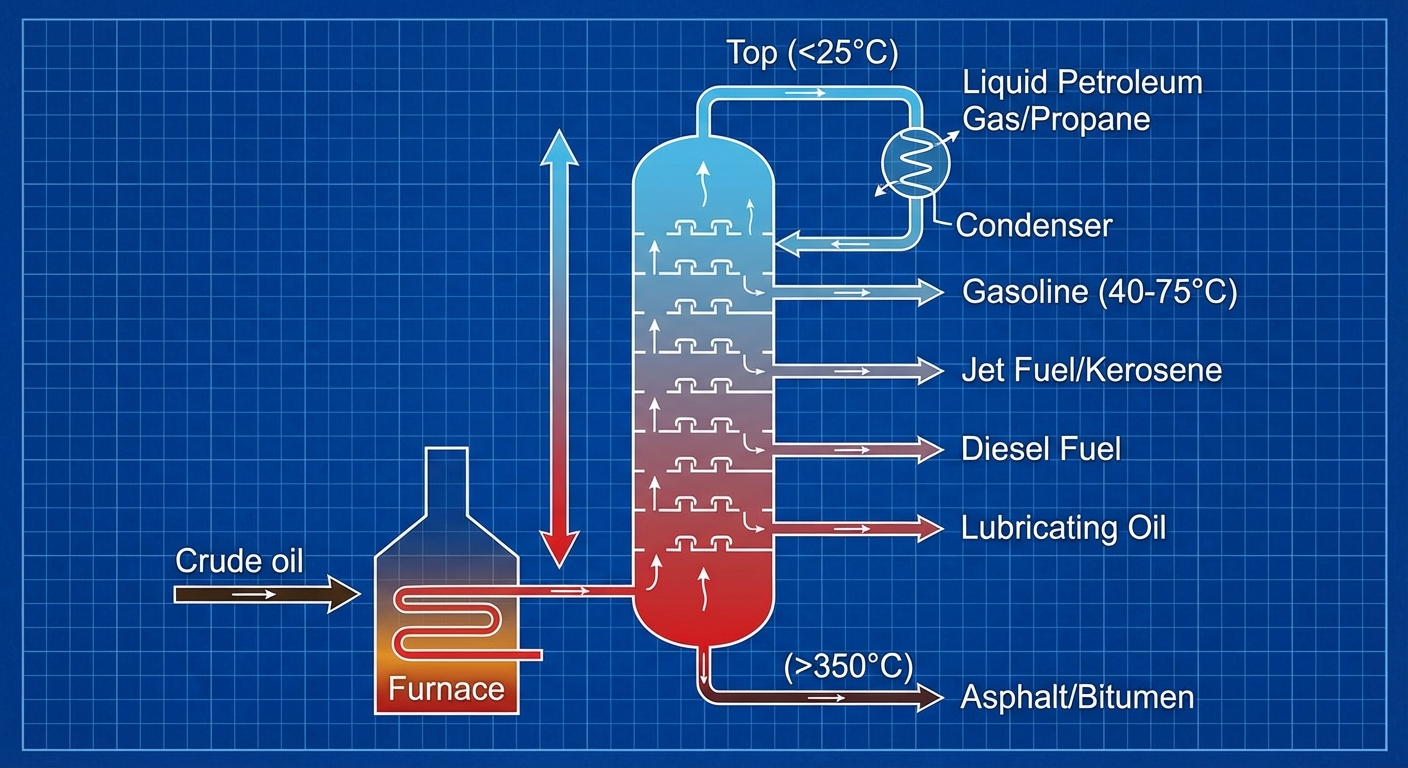
- Top of tower (Lowest boiling point): Gases, Gasoline, Aviation fuel.
- Bottom of tower (Highest boiling point): Diesel, Heating oil, Asphalt/Bitumen.
Uses: Transportation (gasoline/diesel) is the primary use, followed by the production of plastics (petrochemicals).
3. Natural Gas
Natural gas is composed primarily of Methane () (80-95%) and lighter hydrocarbons (Ethane, Propane).
- Uses: Heating homes (furnaces), cooking, and increasingly for electricity generation.
- Extraction: Often associated with oil drilling, but increasingly accessed via Hydraulic Fracturing (Fracking) to extract gas from shale rock.
- Status: It is considered the "cleanest" fossil fuel because it emits almost no particulate matter or and produces roughly 60% as much as coal when burned.
- Risk: Methane leaks during extraction are dangerous because Methane is a greenhouse gas 25x more potent than .
4. Cogeneration
Cogeneration (Combined Heat and Power - CHP) is the use of a fuel to generate electricity and produce heat. In a standard plant, heat is wasted. In CHP, the steam used to turn the turbine is captured and used to heat the building. This boosts efficiency from ~35% to ~90%.
Nuclear Power
Nuclear energy is nonrenewable because it relies on Uranium ore (), which is a finite mineral resource. It produces electricity through Fission (splitting atoms), not combustion (burning).
How It Works
- Fission: A neutron strikes a large atomic nucleus (), splitting it into smaller parts. This releases massive heat and more neutrons.
- Steam Generation: This heat is used to boil water into steam.
- Electricity: The steam turns a turbine, which turns a generator.
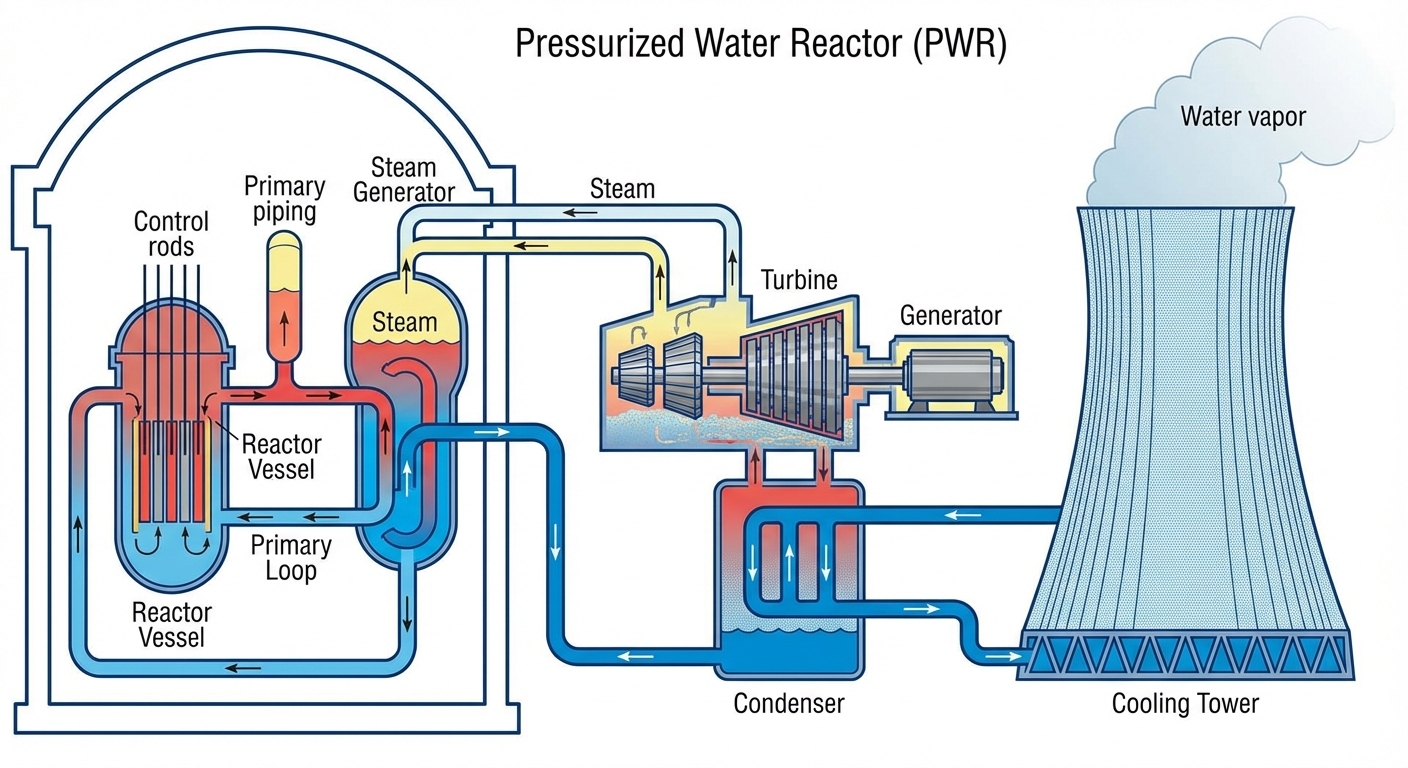
Key Reactor Components:
- Fuel Rods: Tubes containing Uranium pellets.
- Control Rods: Inserted between fuel rods to absorb excess neutrons and slow down or stop the reaction.
- Coolant: Water circulating through the core to remove heat.
The Trade-off
- Advantages: No air pollution during operation (), very high energy density, ample supply.
- Disadvantages: Radioactive waste (must be stored for thousands of years), thermal pollution (hot water dumped in rivers), risk of catastrophic accidents, nonrenewable.
Radioactive Decay & Half-Life
A key skill in APES is calculating Half-Life (): the time it takes for one-half of an original radioactive parent atom to decay.
(Where $n$ is the number of half-lives that have passed)
Example Problem:
A sample contains 100g of a radioactive isotope with a half-life of 20 years. How much remains after 80 years?
- Number of half-lives () =
- Calculation:
Major Accidents
You should memorize these three events:
- Three Mile Island (USA, 1979): Partial meltdown due to human error. No direct deaths, but increased fear of nuclear power in the US.
- Chernobyl (Ukraine/USSR, 1986): Worst accident in history. Explosion blowing the roof off the reactor. Massive radiation release across Europe.
- Fukushima Daiichi (Japan, 2011): Tsunami caused by an earthquake flooded backup generators, leading to cooling failure and meltdown.
Common Mistakes & Pitfalls
- Thinking Nuclear Power releases : A common exam trap. Nuclear reactors release water vapor (cooling towers), NOT carbon dioxide. The only associated with nuclear is during the mining/construction phase.
- Confusing Resources vs. Reserves: A "Resource" is the total amount in the ground. A "Reserve" is the amount we can economically extract with current technology.
- Peat is not Coal: Remember that Peat is the precursor to coal. It is not technically coal itself.
- Mixing up Fission and Fusion: Current nuclear plants use Fission (Splitting). Fusion (Combining) powers the sun but is not yet commercially viable on Earth.