Unit 5: Kinetics — Unlocking Reaction Mechanisms
Elementary Reactions
Most chemical reactions do not occur in a single, instantaneous event where all reactant molecules smash together simultaneously. Instead, they occur through a sequence of simpler steps. These individual steps are called elementary reactions (or elementary steps).
An elementary reaction describes a single molecular event, such as a distinct collision between two particles or the radioactive decay of a single particle.
Molecularity
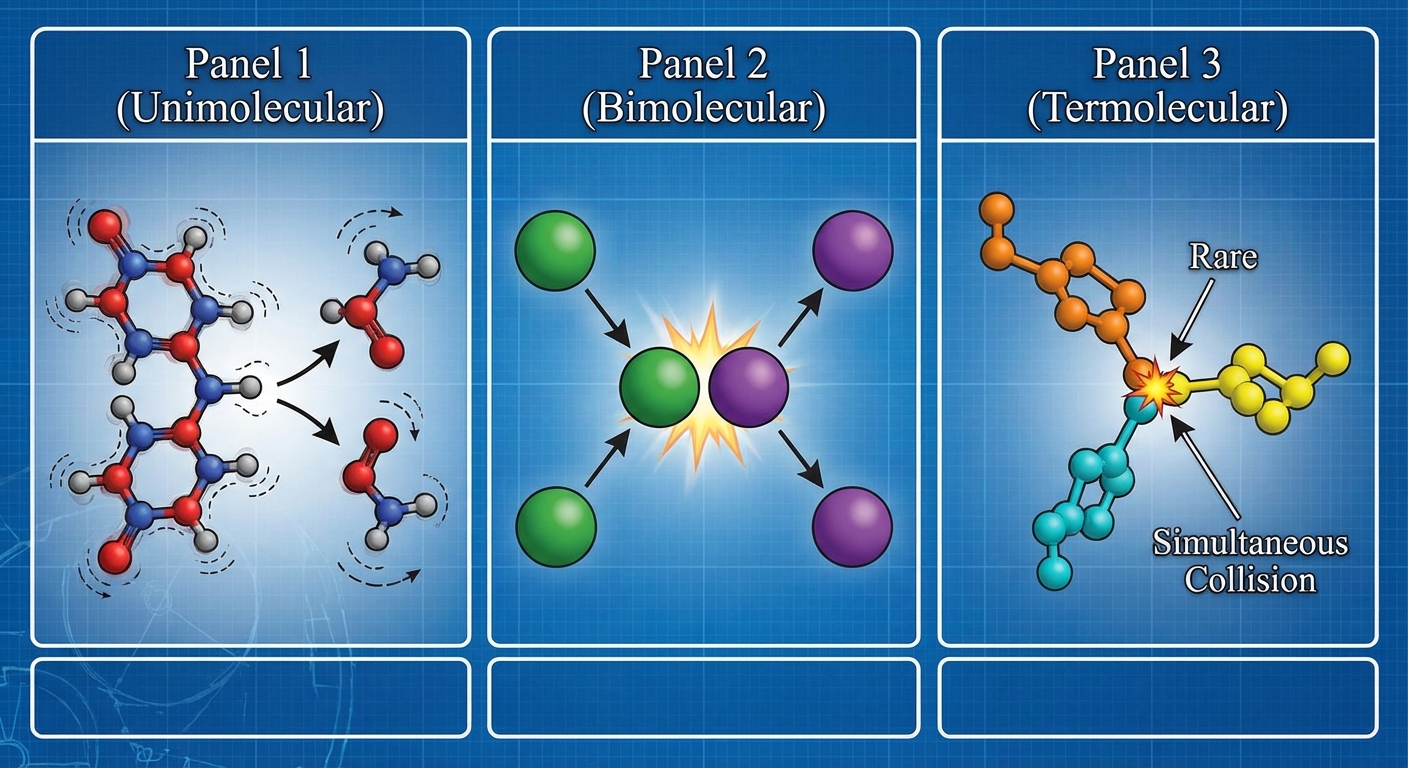
Molecularity refers to the number of reactant particles involved in a single elementary step. This is a crucial concept because, unlike overall reactions, the rate law for an elementary step can be written directly from its molecularity.
| Molecularity | Description | Generic Equation | Rate Law |
|---|---|---|---|
| Unimolecular | A single molecule rearranges or decomposes. | $A \rightarrow products$ | $Rate = k[A]$ |
| Bimolecular | Two particles collide. This is the most common type. | $A + B \rightarrow products$ $2A \rightarrow products$ | $Rate = k[A][B]$ $Rate = k[A]^2$ |
| Termolecular | Three particles collide simultaneously. These are extremely rare due to the low statistical probability of a simultaneous 3-way collision. | $2A + B \rightarrow products$ | $Rate = k[A]^2[B]$ |
The Golden Rule of Elementary Steps
For an overall reaction (e.g., $2N2O5 \rightarrow 4NO2 + O2$), you cannot determine the rate law exponents from the coefficients. However, for a designated elementary step, the stoichiometric coefficients become the exponents in the rate law.
Introduction to Reaction Mechanism
A reaction mechanism is the proposed series of elementary steps that leads from reactants to products. It provides a detailed picture of how the reaction occurs at the molecular level.
Rules for a Valid Mechanism
A proposed mechanism must satisfy two strict requirements to be considered valid:
- Summation: The sum of the elementary steps must add up to the overall balanced chemical equation.
- Rate Law Agreement: The rate law derived from the mechanism (specifically the slow step) must agree with the experimentally determined rate law.
Intermediates vs. Catalysts
Students often confuse these two species because neither appears in the final balanced equation. However, their roles are opposite.
Intermediate: A species that is produced in an early step and consumed in a later step. It is not present in the reactants or the final products.
- Path: Created $\rightarrow$ Destroyed.
- Note: Intermediates are often unstable and short-lived.
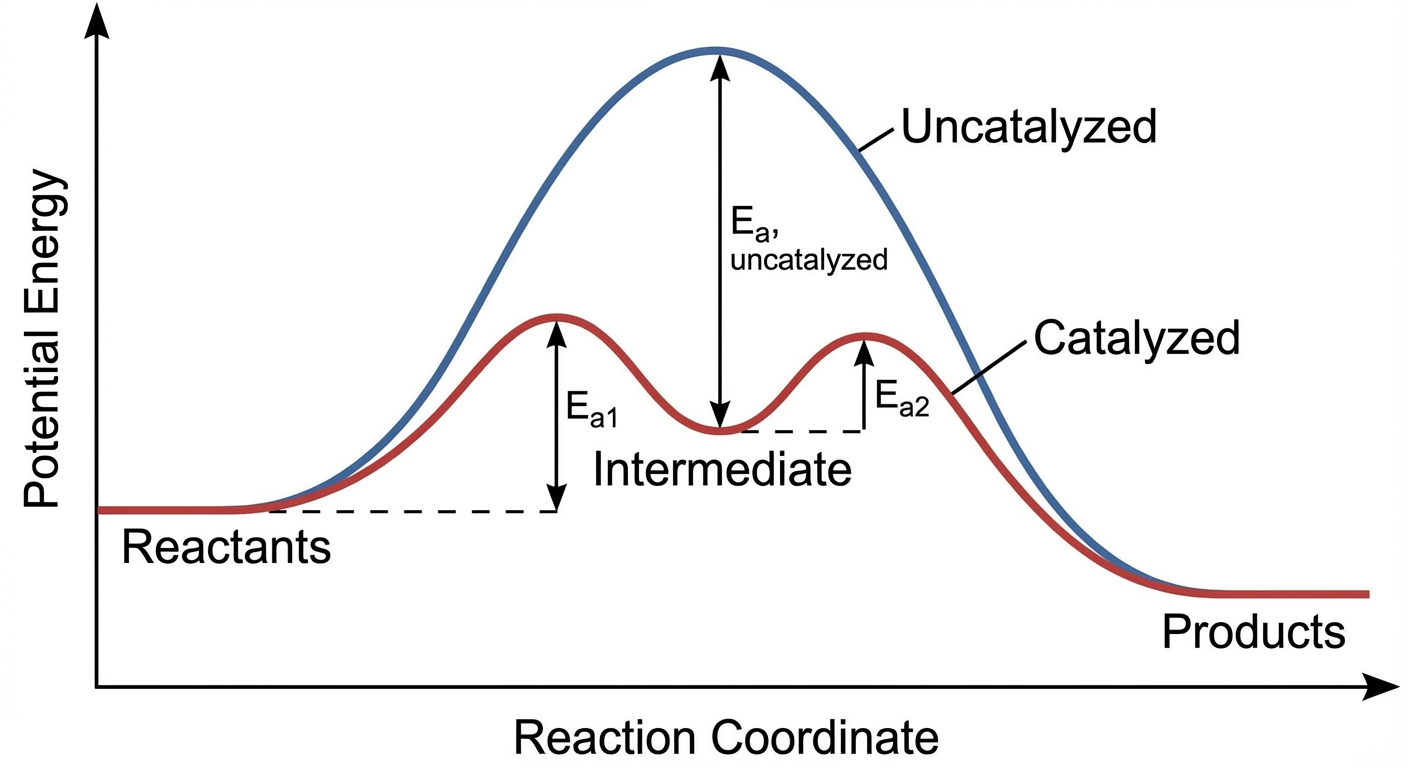
Catalyst: A substance that increases the reaction rate without being consumed overall. It is consumed (used) in an early step and regenerated (produced) in a later step.
- Path: Destroyed (Used) $\rightarrow$ Created (Regenerated).
- Function: It lowers the activation energy ($E_a$) by providing an alternate mechanism.
Example Analysis:
Consider the mechanism:
- $NO + NO \rightarrow N2O2$ (Step 1)
- $N2O2 + O2 \rightarrow 2NO2$ (Step 2)
- Overall Reaction: Add the steps. $2NO + O2 \rightarrow 2NO2$. ($N2O2$ cancels out).
- Intermediate: $N2O2$ (Produced in Step 1, consumed in Step 2).
Reaction Mechanism and Rate Law
The speed of a multi-step reaction is limited by its slowest step, known as the Rate-Determining Step (RDS). The rate law of the overall reaction corresponds to the rate law of this step.
Scenario 1: The First Step is the RDS
This is the simplest scenario. If the first elementary step is the slow step, the overall rate law is simply the rate law of that first step.
Example:
- Step 1: $NO2 + NO2 \rightarrow NO_3 + NO$ (Slow)
- Step 2: $NO3 + CO \rightarrow NO2 + CO_2$ (Fast)
Since Step 1 is the bottleneck, the overall rate is determined by the collision of two $NO2$ molecules.
Scenario 2: The RDS is Not the First Step (Fast Equilibrium)
If the slow step is preceded by a fast, reversible step, the derivation implies that the fast step reaches equilibrium quickly.
Problem: The rate law for the slow step will contain an Intermediate.
Rule: You generally cannot include intermediates in a final rate law expression. You must substitute them algebraically using the equilibrium condition.
Working Example:
- Step 1: $NO + Br2 \rightleftharpoons NOBr2$ (Fast Equilibrium)
- Step 2: $NOBr_2 + NO \rightarrow 2NOBr$ (Slow/RDS)
Write rate for the Slow Step:
Problem: $NOBr_2$ is an intermediate.Use Step 1 Equilibrium:
Rate forward = Rate reverseSolve for the Intermediate:
Substitute back into the Slow Step Rate:
Simplify:
Combine all constants ($k2, k1, k{-1}$) into a new experimental constant $k{obs}$.
Steady-State Approximation
Note: In general AP Chemistry courses, the "Fast Equilibrium" approach (Scenario 2 above) is the standard method for solving these problems. However, the Steady-State Approximation is a more general method used in advanced chemistry when a prior equilibrium cannot be assumed.
The Steady-State Approximation assumes that during the major part of the reaction, the concentration of a reactive intermediate remains small and constant. In other words, the rate of production of the intermediate equals its rate of consumption.
For an intermediate $I$:
Application
Consider the same mechanism as above, but without assuming Step 1 is in perfect equilibrium:
- $A + B \xrightarrow{k_1} I$
- $I \xrightarrow{k_{-1}} A + B$
- $I + C \xrightarrow{k_2} Products$
Rate of formation of I: $k1[A][B]$ Rate of consumption of I: $k{-1}[I] + k_2[I][C]$ (Consumed in both reverse Step 1 and forward Step 2)
Set Formation = Consumption:
Factor out $[I]$:
Solve for $[I]$:
Substitute this into the rate law for product formation ($Rate = k2[I][C]$):
This result is more complex than the fast equilibrium result but reduces to it if $k{-1} \gg k2[C]$.
Common Mistakes & Pitfalls
Confusing Intermediates and Catalysts:
- Mistake: Thinking a catalyst is an intermediate.
- Correction Check: Did it start as a reactant and regenerate at the end? That's a catalyst. Did it appear in the middle and disappear? That's an intermediate.
Applying Coefficients to Overall Rate Laws:
- Mistake: Given $2A + B \rightarrow C$, writing $Rate = k[A]^2[B]$.
- Correction Check: Never do this for the overall reaction unless told it is a "one-step" elementary reaction. Only do this for individual steps inside a mechanism.
Leaving Intermediates in the Final Answer:
- Mistake: Writing a final rate law like $Rate = k[A][I]$.
- Correction Check: If $I$ is not a reactant in the overall balanced equation, it must be substituted out algebraically.