AP Biology Unit 3: Cellular Energetics — Master Guide to Enzymes
Fundamentals of Enzyme Structure
To understand cellular energetics, you must first master the machinery that makes life possible: Enzymes. Most enzymes are proteins (tertiary structure is critical), though some RNA molecules, known as ribozymes, also have catalytic activity.
The Biological Catalyst
An enzyme is a macromolecule that acts as a biological catalyst. A catalyst is an agent that speeds up a chemical reaction without being consumed by the reaction itself. Because they are not used up, enzymes can be reused repeatedly to catalyze the same specific reaction.
The Active Site and Specificity
The functioning of an enzyme relies entirely on its 3D shape (conformation).
- Substrate: The specific reactant that an enzyme acts on.
- Active Site: A specific region on the enzyme—often a pocket or groove—where the substrate binds. The chemical properties of the active site (charge, hydrophobicity, pH) must match the substrate.
Since proteins function based on their shape, enzymes exhibit specificity. For example, sucrase will hydrolyze sucrose but will be completely useless against maltose, even though they are both disaccharides.
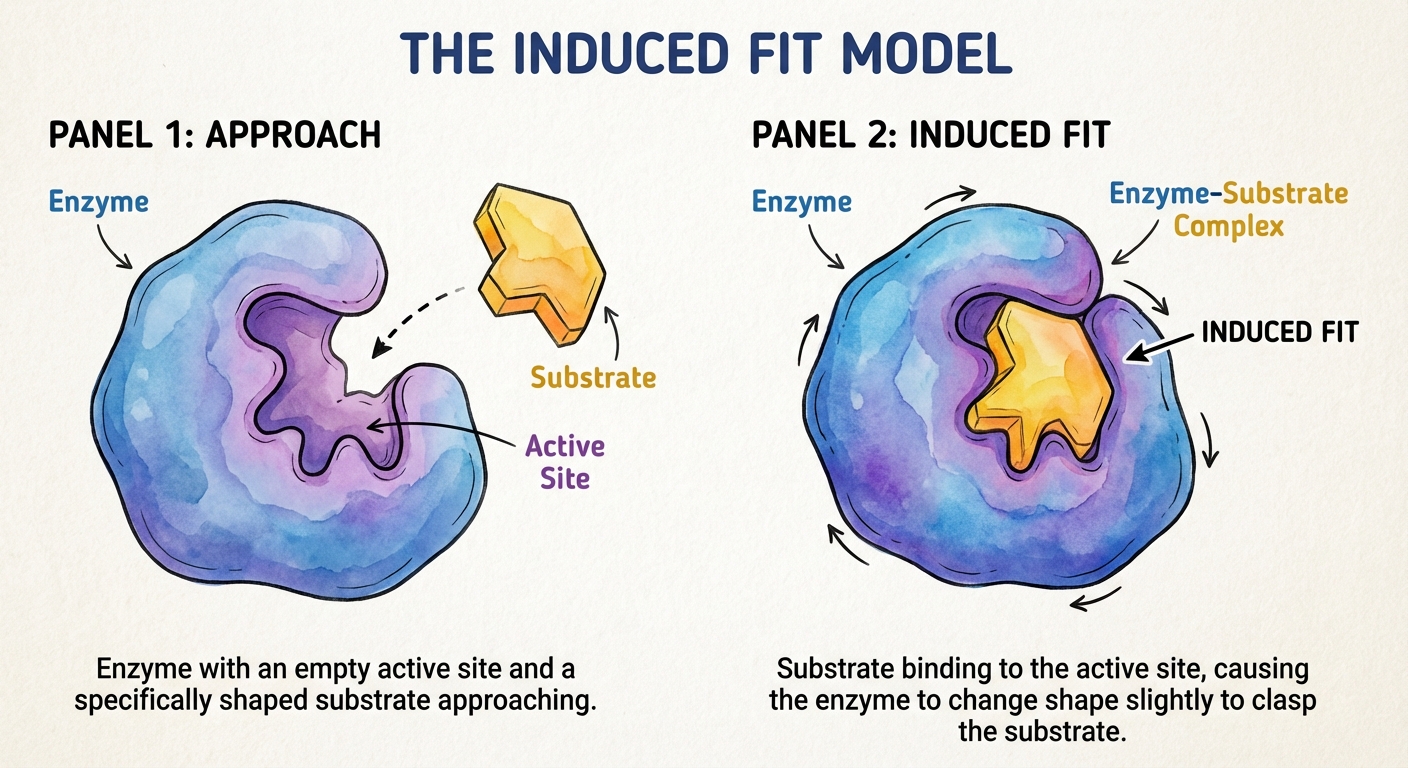
The Induced Fit Model
Historically, scientists described enzyme-substrate interaction as a "Lock and Key" model (perfect fit). However, current research supports the Induced Fit Model.
When a substrate enters the active site, the enzyme changes shape slightly—like a handshake tightening—to fit the substrate more snugly. This tightening brings chemical groups of the active site into positions that enhance their ability to catalyze the reaction.
Mechanism of Enzyme Catalysis
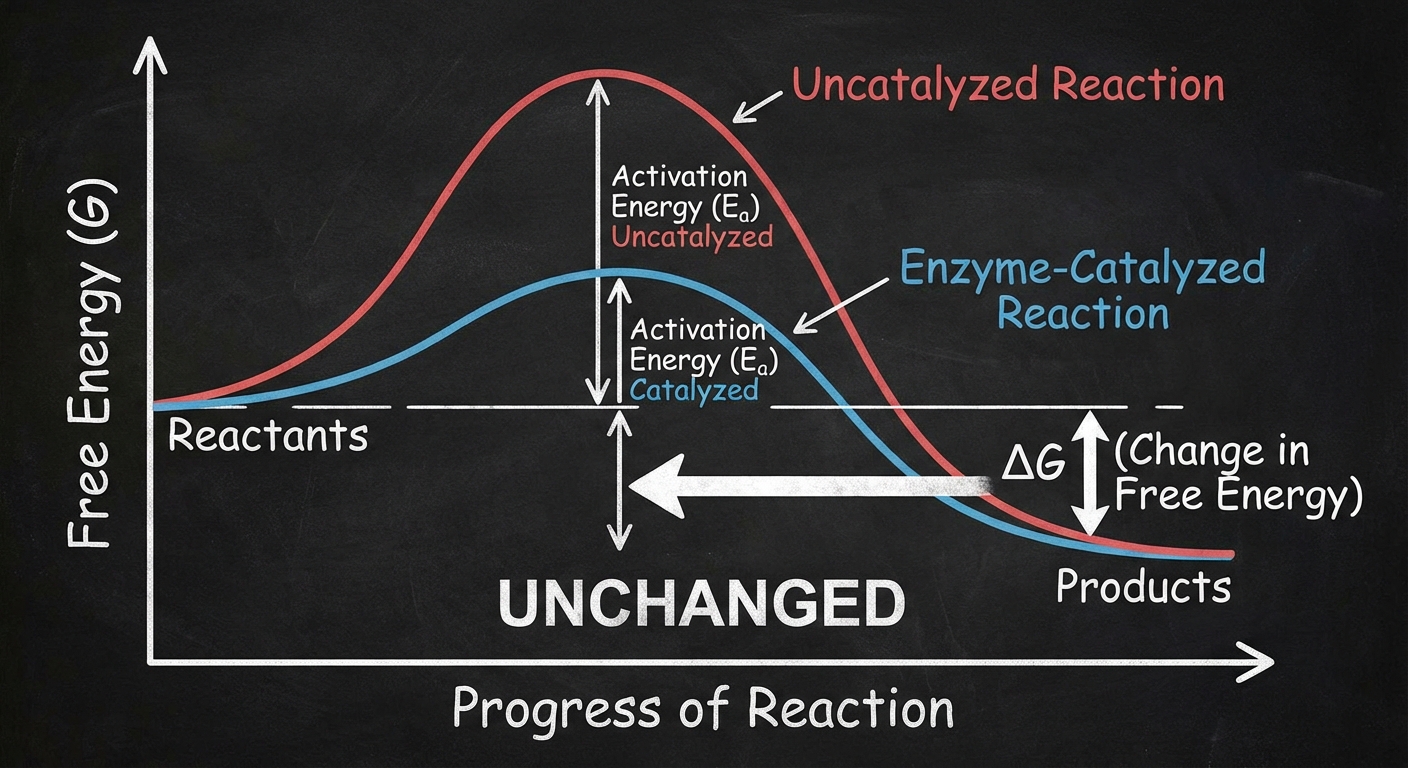
Enzymes speed up metabolic reactions by lowering energy barriers. They do not add energy to a reaction; they simply make it easier for the reaction to occur.
Activation Energy ($E_A$)
Every chemical reaction involves bond breaking and bond forming. To start a reaction, the starting molecules must absorb energy to reach an unstable state known as the transition state.
- Activation Energy ($E_A$): The initial investment of energy required to start a reaction.
- Enzyme Role: Enzymes lower the $E_A$, allowing reactant molecules to reach the transition state at moderate temperatures (like body temperature).
Crucial Thermodynamic Rules:
- Enzymes DO NOT change the change in free energy ($\Delta G$) of the reaction.
- Enzymes DO NOT turn an endergonic reaction into an exergonic one.
- They only accelerate the rate at which equilibrium is reached.
Catalytic Strategies
How exactly does the active site lower $E_A$? The enzyme may:
- Orient substrates: Align two substrates perfectly so a reaction can happen.
- Strain bonds: Physically stretch the substrate molecules toward their transition state form.
- Microenvironment: Create a specific pH or hydrophobic environment within the active site that favors the reaction.
Environmental Impacts on Enzyme Function
Because enzymes are proteins, their efficiency is heavily dependent on the cellular environment. Changes in the environment can disrupt the hydrogen bonds, ionic bonds, and weak interactions that maintain the enzyme's active shape.
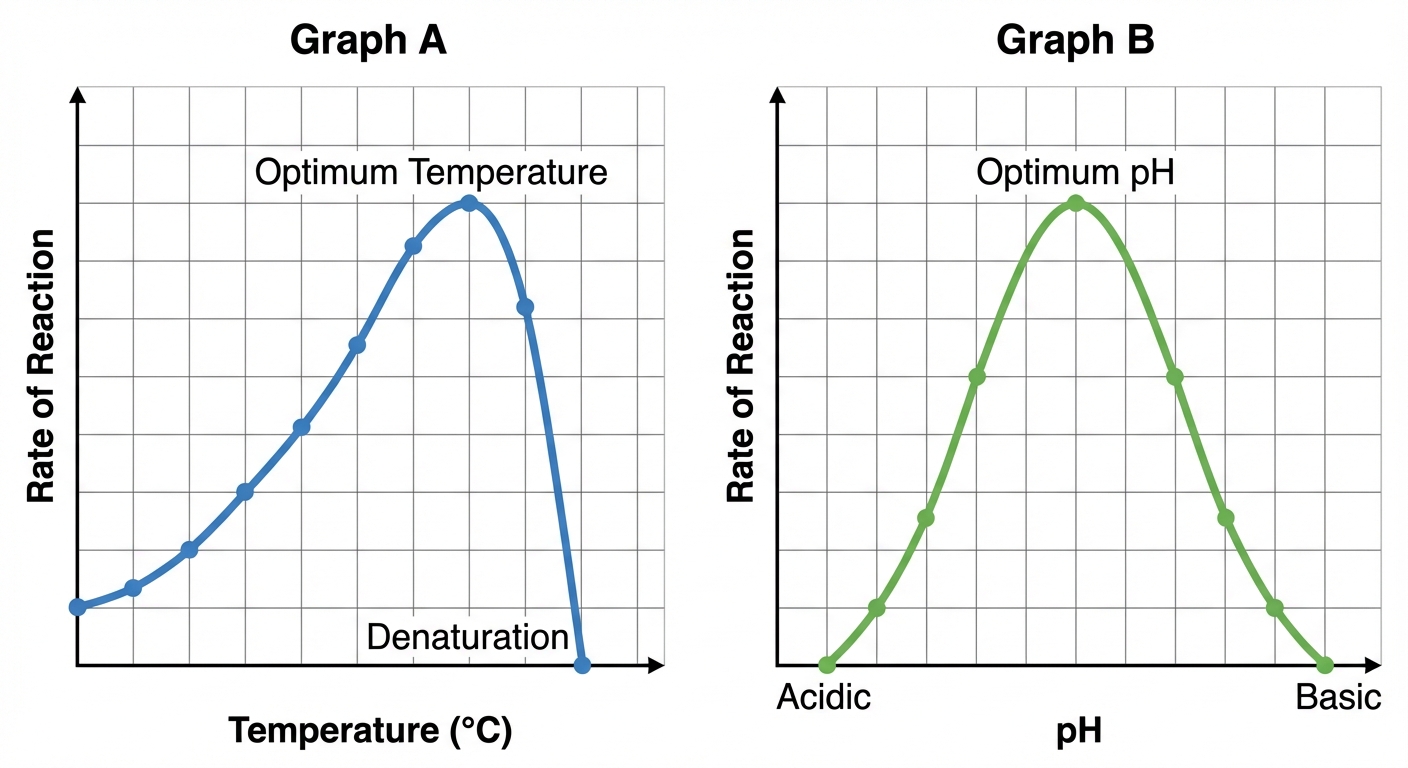
1. Temperature
- Rate Increase: Initially, as temperature rises, reaction rate increases. This is because substrates collide with active sites more frequently due to higher kinetic energy.
- Optimum Temperature: The point at which reaction rate is fastest (e.g., $37^\circ\text{C}$ for human enzymes, $75^\circ\text{C}$ for thermophilic bacteria).
- Denaturation: If temperature goes beyond the optimum, the thermal agitation disrupts the weak bonds stabilizing the protein's tertiary structure. The protein loses its shape and function. Denaturation at high temperatures is usually irreversible.
2. pH
- Every enzyme has an optimum pH.
- Deviating from this optimum (too acidic or too basic) disrupts hydrogen bonding and ionic interactions between R-groups (side chains) of the amino acids.
- Example: Pepsin (stomach) works best at pH 2, while Trypsin (intestine) works best at pH 8.
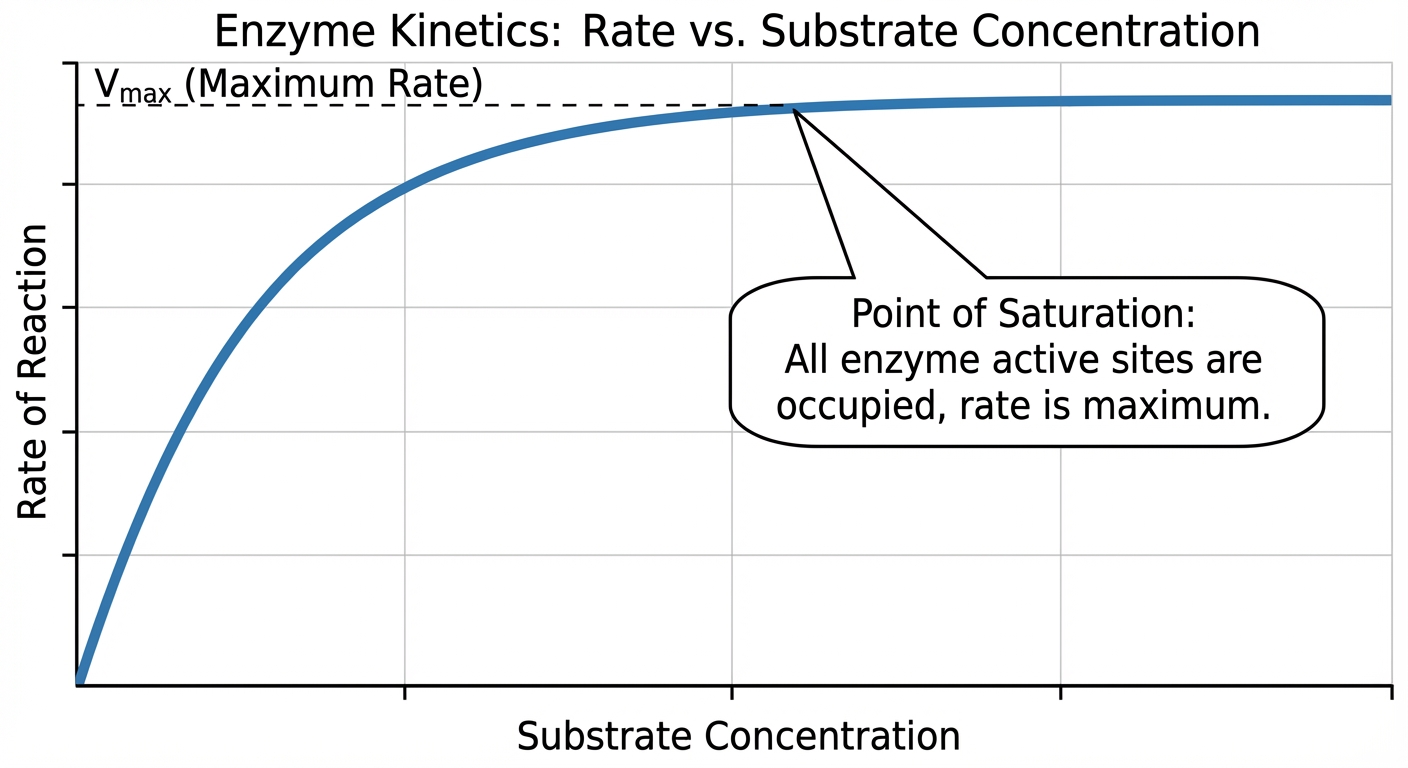
3. Substrate Concentration
Assuming a fixed amount of enzyme:
- Linear Phase: At low substrate concentrations, adding more substrate linearly increases the reaction rate.
- Saturation Point: Eventually, every active site is occupied continuously. The enzyme is "saturated."
- Plateau: At saturation, adding more substrate will not increase the rate further. The only way to increase the rate ($V_{max}$) at this point is to add more enzyme.
Regulation: Inhibition and Cofactors
Cells must control when and where enzymes are active. This is often done via inhibitors.
Cofactors and Coenzymes
Some enzymes require non-protein helpers to function.
- Cofactors: Inorganic ions (e.g., Zinc, Iron, Magnesium).
- Coenzymes: Organic molecules (e.g., Vitamins).
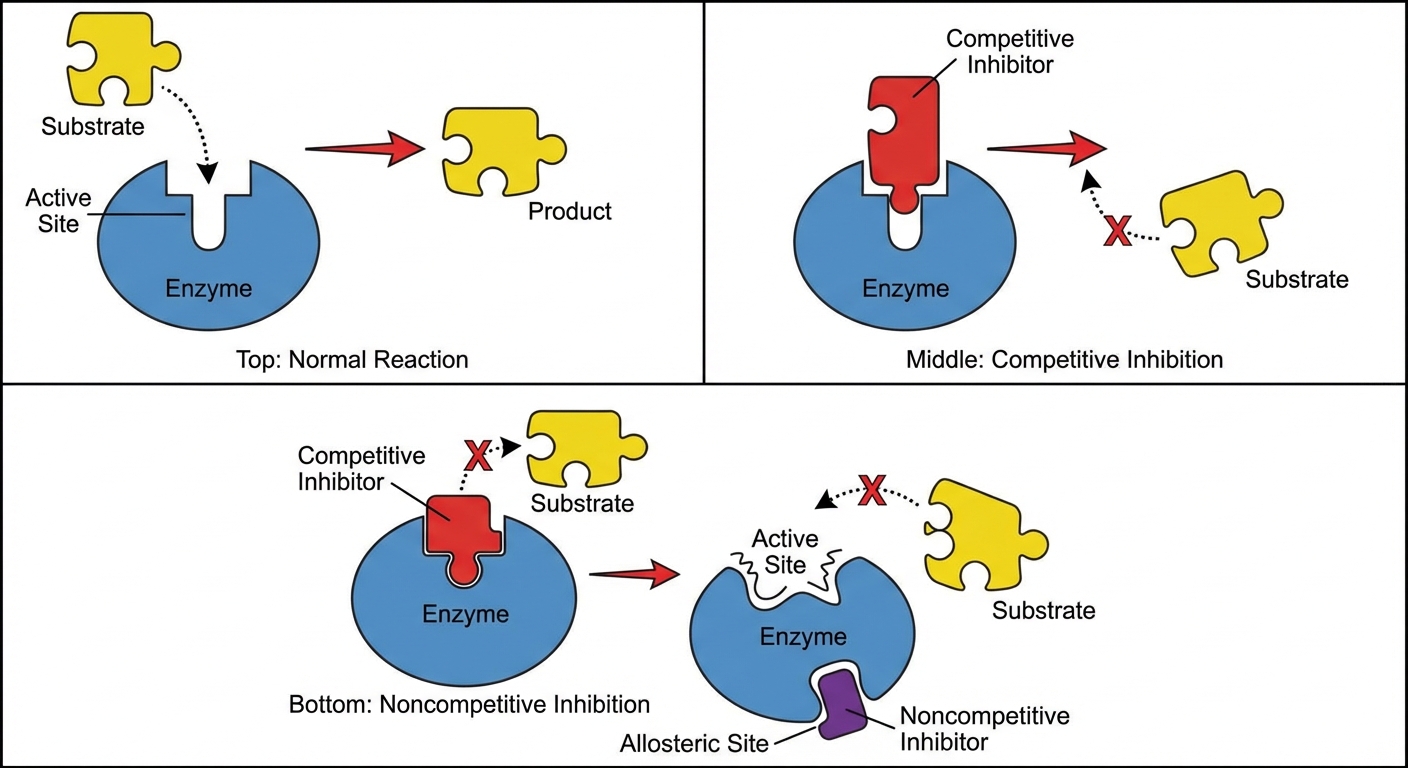
Competitive vs. Noncompetitive Inhibition
This is a high-frequency AP exam topic. You must understand how these affect the reaction.
| Feature | Competitive Inhibition | Noncompetitive Inhibition |
|---|---|---|
| Binding Site | Binds to the Active Site. | Binds to an Allosteric Site (a site other than the active site). |
| Mechanism | Blocks the substrate from entering. | Changes the shape of the enzyme so the active site is no longer functional. |
| Overcoming it | Can be overcome by adding more substrate (outcompeting the inhibitor). | Cannot be overcome by adding more substrate. |
| Effect on Rate | Increases time to reach max rate, but max rate is still possible. | Lowers the maximum possible rate ($V_{max}$). |
Allosteric Regulation
Most naturally occurring regulation is noncompetitive. Allosteric regulation involves a regulatory molecule binding to a protein at one site and affecting the protein's function at another site. This can result in either activation (stabilizing the active form) or inhibition (stabilizing the inactive form).
Common Mistakes & Pitfalls
"Enzymes die" vs. "Enzymes denature"
- Mistake: Saying an enzyme "dies" when heated.
- Correction: Enzymes are not living things; they are molecules. They denature (unfold). Also, remember that cold temperatures generally do not denature enzymes; they just slow down molecular motion. Warming it back up restores function.
Product vs. Rate
- Mistake: Thinking enzymes produce more product.
- Correction: Enzymes do not change the final amount of product (determined by equilibrium); they only decrease the time it takes to get there.
Lock and Key Rigidity
- Mistake: Describing the active site as a rigid, static mold.
- Correction: Always reference Induced Fit. The enzyme is dynamic and flexible.
$\Delta G$ Confusion
- Mistake: Claiming enzymes lower $\Delta G$ or make reactions spontaneous.
- Correction: Enzymes lower Activation Energy ($E_A$). $\Delta G$ remains constant regardless of the enzyme.