Mastering Intermolecular Forces and States of Matter
Unit 3: Intermolecular Forces and Properties
Intermolecular Forces: The Glue of Matter
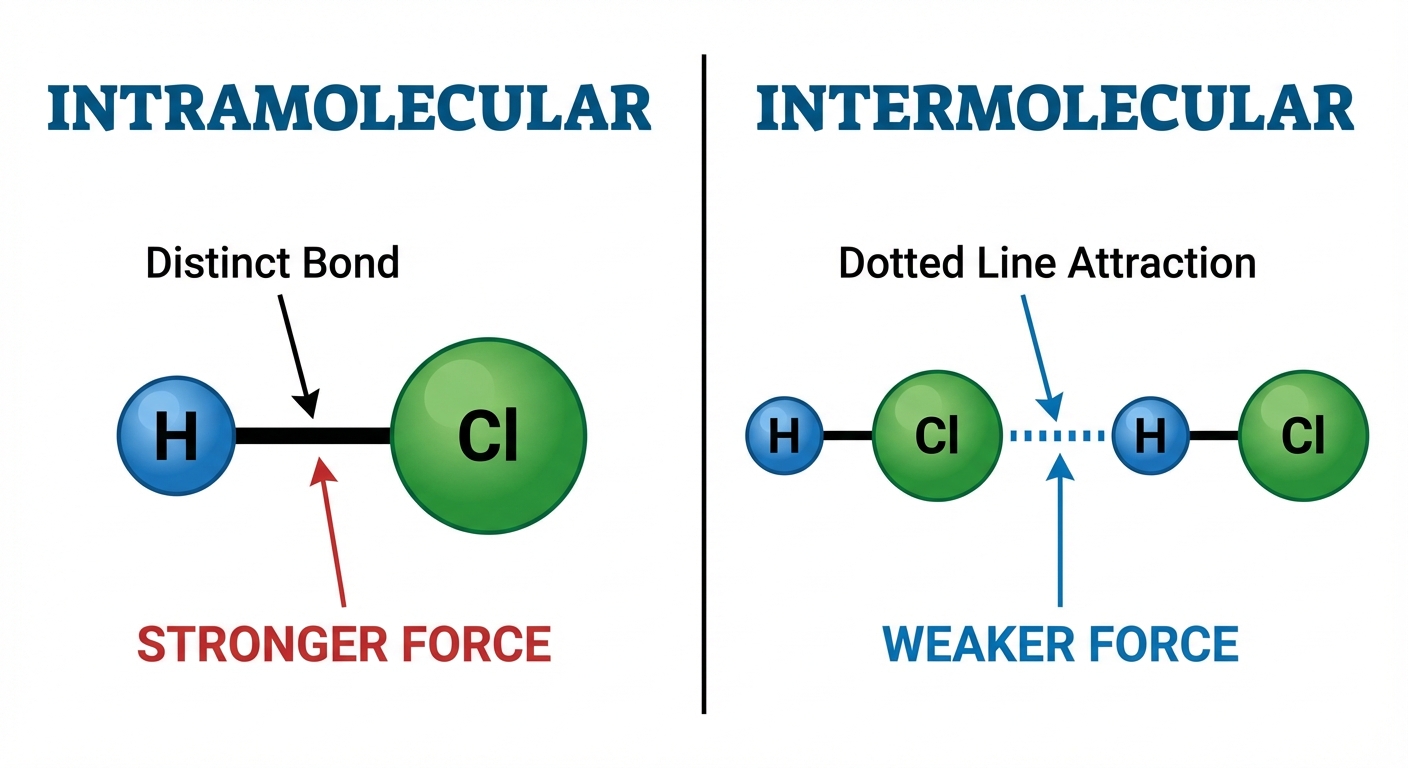
Intermolecular Forces (IMFs) are the attractive forces that exist between molecules. It is crucial to distinguish these from Intramolecular forces (ionic, covalent, metallic bonds), which hold atoms together within a molecule.
While intramolecular forces determine chemical properties (reactivity), intermolecular forces largely determine physical properties (boiling point, melting point, density, and solubility).
1. London Dispersion Forces (LDF)
Also known as induced dipole-induced dipole forces or dispersion forces.
- Definition: Temporary fluctuations in electron distribution create a temporary dipole, which induces a dipole in a neighboring molecule.
- Occurrence: Present in ALL molecules and atoms (polar and nonpolar). However, they are the only force present in nonpolar molecules (e.g., $CH4$, $N2$, Noble gases).
- Key Concept - Polarizability: The strength of LDFs depends on the polarizability of the molecule—how easily the electron cloud can be distorted.
- More electrons $\rightarrow$ Larger electron cloud $\rightarrow$ Higher polarizability $\rightarrow$ Stronger LDFs.
- Consequently, larger molecules (higher molar mass) generally have higher boiling points due to stronger LDFs.
- Shape Factor: Long, cylindrical molecules have stronger LDFs than spherical ones of similar mass because they have more surface area for contact (e.g., n-pentane vs. neopentane).
2. Dipole-Dipole Interactions
- Definition: The electrostatic attraction between the positive end of one permanent dipole and the negative end of another.
- Occurrence: Only in polar molecules.
- Strength: Generally stronger than LDFs (for molecules of similar size). The strength increases as the magnitude of the dipole moment increases.
3. Hydrogen Bonding
- Definition: A particularly strong type of dipole-dipole interaction (not an actual chemical bond).
- Requirements:
- A Hydrogen atom covalently bonded to a highly electronegative atom (N, O, or F).
- An attraction to a lone pair on an electronegative atom (N, O, or F) of a nearby molecule.
- Impact: Responsible for the anomalously high boiling point of water ($H2O$) compared to $H2S$ or $H_2Se$, and the structure of DNA.
4. Ion-Dipole Forces
- Definition: The attraction between an ion and a neutral polar molecule.
- Occurrence: Most commonly seen in solutions (e.g., NaCl dissolved in water).
- Strength: Usually stronger than H-bonding. The strength depends on Coulomb's Law relationships:
- $q_{ion}$ = charge of the ion (higher charge = stronger force)
- $\mu_{dipole}$ = magnitude of the dipole (more polar = stronger force)
- $r$ = distance (smaller ion radius = stronger force)
Comparison of Strength
Generally:
LDF < Dipole-Dipole < Hydrogen Bonding < Ion-Dipole < Ion-Ion (Intramolecular)
Note: A very large molecule with only LDFs can have a higher boiling point than a tiny molecule with Dipole-Dipole forces. Always check size/polarizability first!
Solids, Liquids, and Gases
The state of matter is determined by the competition between the kinetic energy of the particles (temperature) and the strength of the IMFs holding them together.
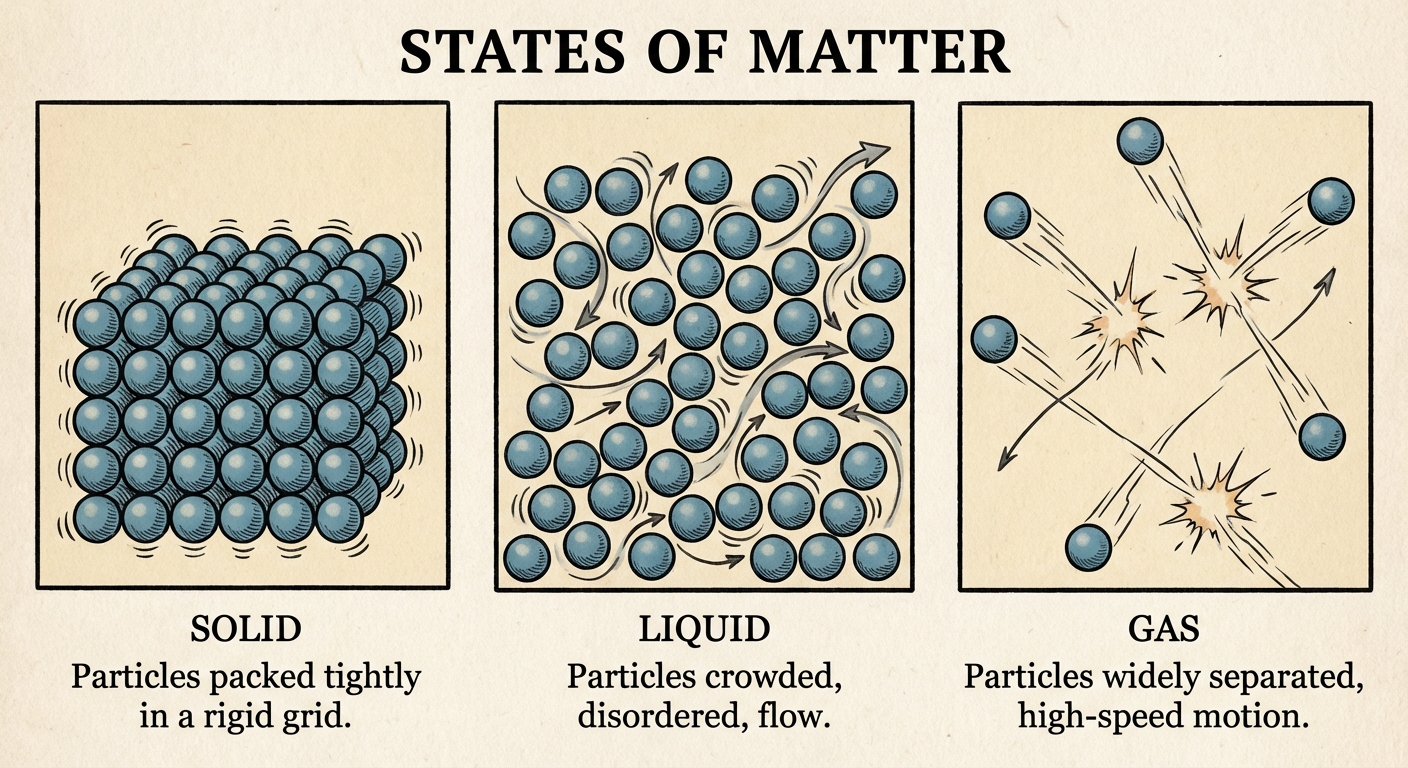
1. General Properties & Particle Behavior
| Property | Solid | Liquid | Gas |
|---|---|---|---|
| Particle Proximity | Very close interaction | Close interaction | Far apart (mostly empty space) |
| Motion | Vibrational only (fixed positions) | Random motion, sliding past one another | different speeds, random, continuous |
| Shape/Volume | Definite shape & volume | Indefinite shape, definite volume | Indefinite shape & volume |
| Compressibility | Negligible | Negligible | High |
2. Properties Determined by IMFs
For liquids and solids, stronger IMFs result in specific trends. If Force $\uparrow$, then:
- Boiling Point & Melting Point: Increase. More energy is required to overcome the attraction between particles to change phase.
- Surface Tension: Increase. Stronger inward forces pull surface molecules together.
- Viscosity: Increase. Stronger attraction makes it harder for molecules to slide past one another (flow resistance).
- Vapor Pressure: Decreases. Stronger IMFs hold molecules in the liquid phase, creating fewer gas particles above the surface.
Important Relationship: Vapor pressure and boiling point are inversely related. High vapor pressure $\implies$ Weak IMFs $\implies$ Low boiling point.
Properties of Solids
Solids can be categorized by their internal arrangement and the forces holding them together.
Amorphous vs. Crystalline
- Amorphous Solids: Disordered arrangement; no distinct melting point (e.g., glass, rubber, some plastics).
- Crystalline Solids: Highly regular, repeating arrangement of components (unit cells).
Types of Crystalline Solids
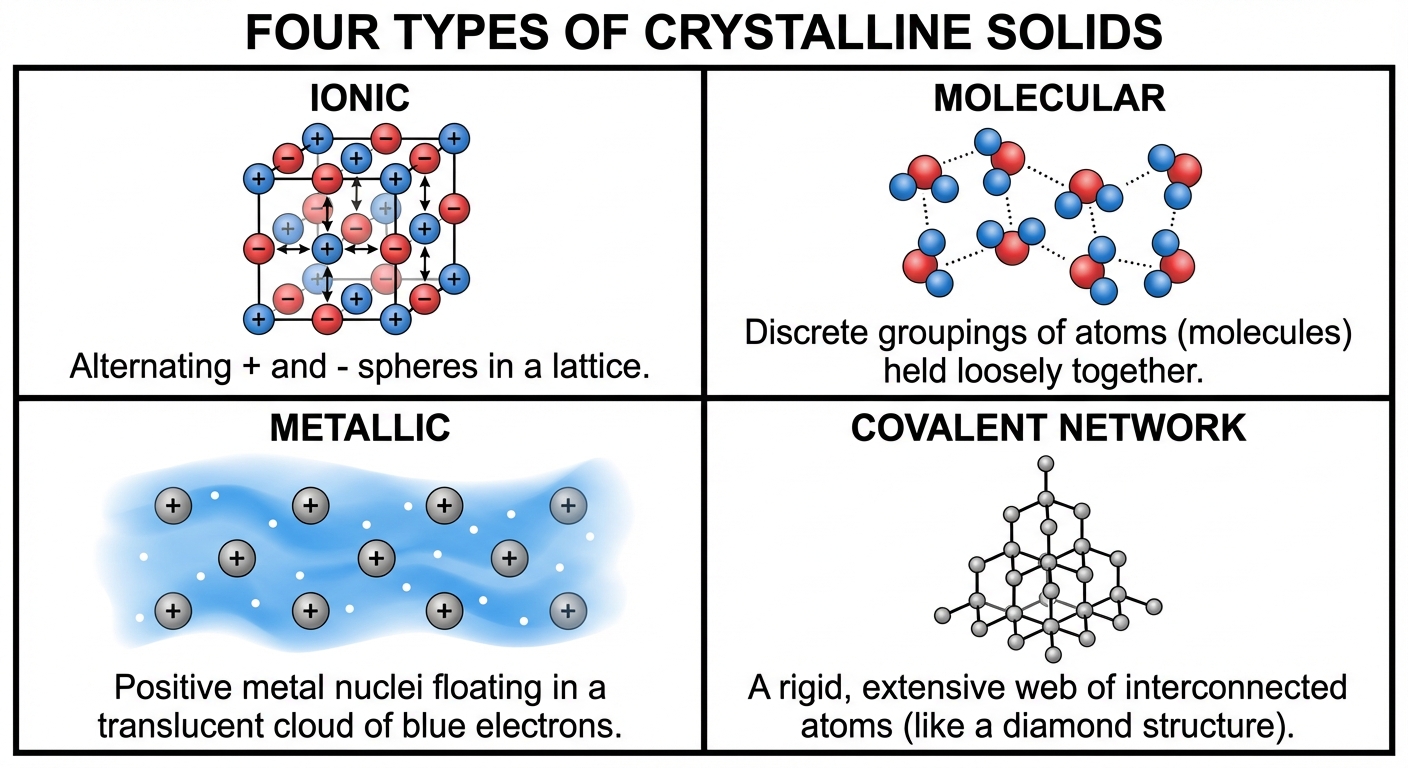
1. Ionic Solids
- Constituent Particles: Ions (Cations and Anions).
- Holding Force: Ionic Bonds (Electrostatic attraction). Highly dependent on Lattice Energy (Coulomb's Law).
- Properties:
- High Melting/Boiling points.
- Hard but brittle (repulsion occurs when layers slide).
- Conductivity: Poor conductors as solids (ions fixed), but excellent conductors when molten or dissolved in water (ions free to move).
- Examples: NaCl, MgO, KI.
2. Molecular Solids
- Constituent Particles: Discrete molecules (atoms or neutral molecules).
- Holding Force: Intermolecular Forces (LDF, Dipole-Dipole, H-bonds).
- Properties:
- Low to moderate Melting/Boiling points (easy to melt).
- Usually soft.
- Non-conductive (no free electrons or ions).
- Examples: Ice ($H2O$), Dry Ice ($CO2$), Sugar ($C{12}H{22}O{11}$), Iodine ($I2$).
3. Covalent Network Solids
- Constituent Particles: Atoms bonded in a continuous 2D or 3D network.
- Holding Force: Covalent Bonds (Intramolecular).
- Properties:
- Extremely high Melting points.
- Very hard.
- Generally non-conductive (Exception: Graphite).
- Common Examples (Memorize these!):
- Diamond (C): Tetrahedral ($sp^3$), hard insulator.
- Graphite (C): Sheets of hexagons ($sp^2$), soft, slippery, conducts electricity due to delocalized $\pi$ electrons between sheets.
- Silicon Dioxide ($SiO_2$): Quartz/sand.
- Silicon Carbide ($SiC$).
4. Metallic Solids
- Constituent Particles: Metal atoms.
- Holding Force: Metallic Bonding ("Sea of Electrons"). Valence electrons are delocalized over the entire crystal.
- Properties:
- Variable melting points.
- Malleable and Ductile: Atoms can slide past without breaking the bond because the electron sea adjusts.
- Excellent Conductors: Highly mobile valence electrons transfer heat and electricity.
- Examples: Cu, Fe, Ag, Alloys (Brass, Steel).
Mnemonics & Study Tips
- H-Bonding: Remember "Hydrogen just wants to have FON". H must be bonded directly to Fluorine, Oxygen, or Nitrogen.
- "Like Dissolves Like": Polar solvents dissolve polar solutes; Nonpolar solvents dissolve nonpolar solutes. This is an application of IMF compatibility.
Common Mistakes to Avoid
Breaking Bonds vs. Overcoming Forces:
- Wrong: "When water boils, the O-H bonds break."
- Right: "When water boils, the intermolecular hydrogen bonds break. The $H_2O$ molecule remains intact."
- Rule: Phase changes involve overcoming IMFs, not chemical bonds.
LDF Electron Count vs. Mass:
- While usually correlated, the strength of LDF is caused by the number of electrons (polarizability), not the mass itself. When explaining, always credit the electron cloud distortion or polarizability.
Confusing Network Covalent with Molecular:
- $CO2$ looks like $SiO2$ in formula, but they are radically different. $CO2$ is a discrete nonpolar molecule (gas/molecular solid). $SiO2$ is a covalent network (sand).
Graphite vs. Diamond:
- Remember that Graphite is the only common covalent network solid that conducts electricity. Do not generalize "covalent solids never conduct" without acknowledging this exception.