AP Chemistry Unit 8: Mechanisms and Calculations of Buffer Solutions
Properties of Buffers
A buffer solution is a specific aqueous system that resists changes in pH upon the addition of small amounts of strong acid or strong base. It is crucial to understand that a buffer does not necessarily maintain a neutral pH (7); rather, it maintains a constant pH relative to its specific chemical composition.
Composition of a Buffer
To function effectively, a buffer must be able to neutralize both added acid ($H^+$) and added base ($OH^-$). Therefore, it must contain two components:
- An acidic component (to react with added $OH^-$)
- A basic component (to react with added $H^+$)
Crucially, these two components must not react with each other. This is achieved by using a conjugate acid-base pair.
There are two primary ways to create a buffer:
- Direct Preparation: Mixing a weak acid ($HA$) with a salt containing its conjugate base ($A^-$), or a weak base ($B$) with a salt containing its conjugate acid ($HB^+$).
- Example: Acetic acid ($HC2H3O2$) mixed with Sodium Acetate ($NaC2H3O2$).
- Partial Neutralization: Reacting a weak acid with a strong base (or weak base with a strong acid) such that the weak species is in excess. This generates the conjugate in situ.
- Example: Reacting 1.0 mole of $HC2H3O_2$ with 0.5 moles of $NaOH$. The result is a mixture of approximately 0.5 mole unreacted acid and 0.5 mole produced conjugate base.
How Buffers Resist pH Change
The mechanism relies on Le Châtelier’s Principle. The buffer components act as a "sink" for protons or hydroxide ions.
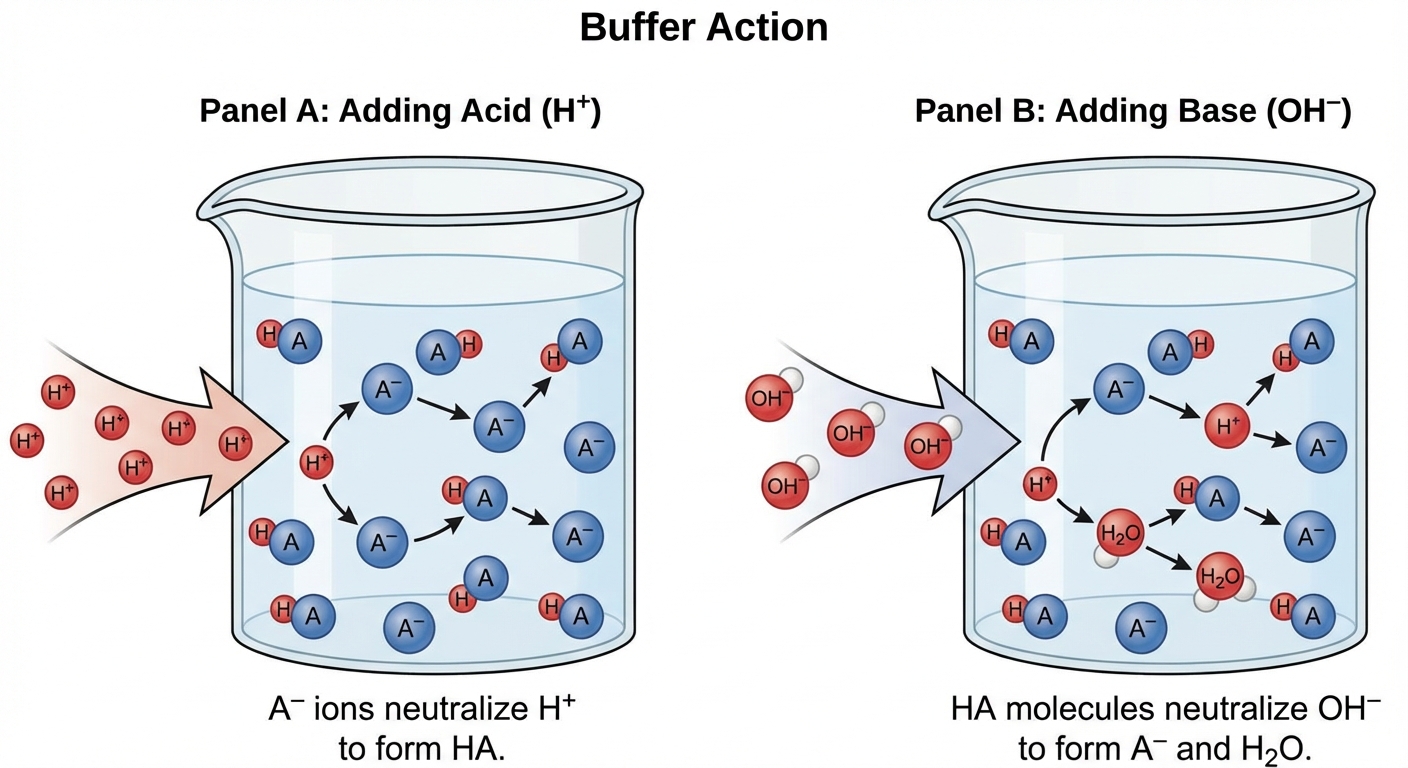
Adding Strong Acid
When a strong acid (source of $H^+$) is added, the conjugate base ($A^-$) in the buffer consumes the protons:
Because the strong $\text{H}^+$ is converted into the weak acid ($HA$), the free $\text{H}^+$ concentration usually increases only slightly, preventing a drastic drop in pH.
Adding Strong Base
When a strong base (source of $OH^-$) is added, the weak acid ($HA$) in the buffer consumes the hydroxide:
The strong $OH^-$ is replaced by the weak conjugate base ($A^-$), preventing a drastic spike in pH.
The Henderson-Hasselbalch Equation
While ICE tables can be used for buffer calculations, the Henderson-Hasselbalch equation provides a more direct relationship between pH, pKa, and the ratio of concentrations. It is derived from the $K_a$ expression.
The Formula
For a buffer system consisting of a weak acid ($HA$) and its conjugate base ($A^-$):
Where:
- $pKa = -\log(Ka)$ of the weak acid.
- $[A^-]$ is the concentration of the conjugate base.
- $[HA]$ is the concentration of the weak acid.
Note: Because this is a ratio, you can often use moles directly instead of molarity, provided the volume is the same for both species.
Key Relationships
- When $[A^-] = [HA]$: The log term becomes $\log(1)$, which is zero. Therefore, . This occurs at the half-equivalence point of a titration.
- When $[A^-] > [HA]$: The log term is positive, so $pH > pK_a$.
- When $[A^-] < [HA]$: The log term is negative, so $pH < pK_a$.
Worked Example
Problem: Calculate the pH of a buffer solution that is 0.50 M in acetic acid ($HC2H3O2$) and 0.50 M in sodium acetate ($NaC2H3O2$). Given $K_a$ for acetic acid is $1.8 \times 10^{-5}$.
Solution:
- Find $pKa$:
- Apply Henderson-Hasselbalch:
Answer: The pH is 4.74.
Buffer Capacity
Buffer capacity refers to the amount of acid or base a buffer can neutralize before the pH changes significantly. It is a measure of the "strength" or endurance of the buffer.
Factors Affecting Capacity
1. Magnitude of Concentrations
The absolute concentrations of the acid and conjugate base determine capacity.
- A buffer with 1.0 M $HA$ and 1.0 M $A^-$ has a high capacity. It can absorb many protons or hydroxides.
- A buffer with 0.01 M $HA$ and 0.01 M $A^-$ has a low capacity. It will be exhausted quickly upon adding external acid/base.
Analogy: Think of a sponge. A large sponge (high concentration) and a small sponge (low concentration) can both be made of the same material (same $pK_a$), but the large sponge can hold significantly more water.
2. The Base/Acid Ratio
A buffer is most resistant to pH changes when the ratio of $\frac{[A^-]}{[HA]}$ is close to 1.
- If the ratio is 1:1, the buffer has equal capacity to resist both acid and base.
- If the ratio deviates significantly (e.g., 10:1), the buffer is good at resisting one but poor at resisting the other.
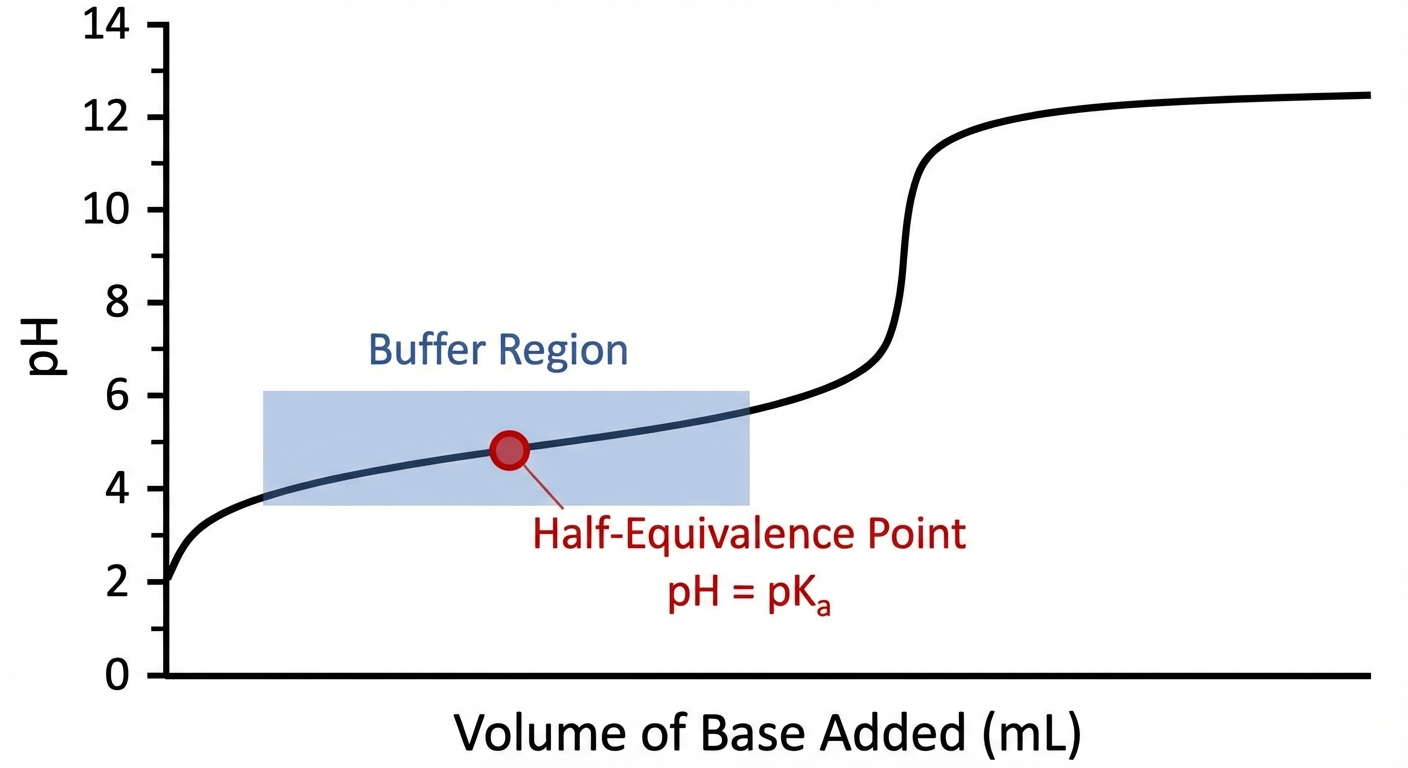
Effective Buffer Range
A buffer is generally considered effective only within a specific pH range:
Outside of this range, the ratio of $\frac{[Base]}{[Acid]}$ is greater than 10:1 or less than 1:10, meaning one component is depleted, and the system loses its ability to stabilize pH effectively.
Summary of Common Mistakes & Pitfalls
- "The Magic 7" Mistake: Students often misuse the formula $pH = 7$ for buffers. Remember, a buffer stabilizes pH at a value determined by the $pK_a$, not necessarily at neutral (7).
- Stoichiometry vs. Equilibrium: When adding a strong acid or base to a buffer, you perform a calculation in two steps:
- Step 1 (Stoichiometry): Assume the strong acid/base reacts completely with the buffer component. Use an ICF (Initial, Change, Final) table with moles.
- Step 2 (Equilibrium): Use the new amounts of $HA$ and $A^-$ in the Henderson-Hasselbalch equation to find the new pH.
- Inverse Log Error: In the Henderson-Hasselbalch equation, the ratio is always $\frac{[Base]}{[Acid]}$. A common error is flipping this to $\frac{[Acid]}{[Base]}$, which gives the wrong sign for the log term.
- Identification Errors: Not recognizing that a Weak Acid + Strong Base can make a buffer if the weak acid is in excess. If the moles are equal, you have reached the equivalence point (salt hydrolysis), not a buffer.