AP Chemistry Unit 1: Atomic Structure, Moles, and Periodicity
Moles and Molar Mass
The mole is the fundamental unit of measurement in chemistry, serving as a bridge between the microscopic world of atoms and the macroscopic world of grams.
The Mole Concept
- Definition: A mole (mol) is defined as the quantity of a substance that contains exactly Avogadro's Number of particles (atoms, molecules, ions, or formula units).
- Avogadro's Number ($N_A$):
- Molar Mass: The mass of one mole of a substance, expressed in grams per mole ($g/mol$). This is numerically equivalent to the average atomic mass found on the periodic table (measured in amu).
Converting Quantities
The ability to convert between mass, moles, and particle count is the foundation of stoichiometry.
- Mass to Moles:
- Moles to Particles:
- Moles to Gas Volume (at STP only):
At Standard Temperature and Pressure ($0^{\circ}C$ and $1 \text{ atm}$), 1 mole of any ideal gas occupies 22.4 Liters.
AP Exam Tip: Always be careful with dimensional analysis. If you are given specific molecules (like $O2$), remember that 1 mole of $O2$ contains $6.022 \times 10^{23}$ molecules, but $2 \times (6.022 \times 10^{23})$ atoms.
Elemental Composition
Percent Composition describes the percent by mass of each element in a compound.
Example: Find the % mass of Nitrogen in Calcium Nitrate, $Ca(NO3)2$.
- Molar Mass of $Ca(NO3)2 = 40.08 + 2(14.01) + 6(16.00) = 164.10 \text{ g/mol}$
- Mass of Nitrogen part = $2 \times 14.01 = 28.02 \text{ g}$
- $\% N = (28.02 / 164.10) \times 100 = \mathbf{17.07\%}$
Empirical vs. Molecular Formulas
- Empirical Formula: The simplest whole-number ratio of atoms in a compound (e.g., $CH_2O$).
- Molecular Formula: The actual number of atoms in a molecule (e.g., $C6H{12}O_6$).
Steps to Determine Formula:
- Mass to Moles: Convert percentages or gram masses to moles for each element.
- Divide by Smallest: Divide all mole values by the smallest mole value calculated.
- Multiply til Whole: If results are not integers (e.g., 1.5), multiply all by a factor (e.g., 2) to get whole numbers.
Mass Spectrometry and Isotopes
Isotopes and Atomic Mass
- Isotopes: Atoms of the same element (same number of protons) with different numbers of neutrons. This results in different mass numbers.
- Average Atomic Mass: The weighted average of the atomic masses of the naturally occurring isotopes of an element. This is the value reported on the Periodic Table.
Mass Spectroscopy
Mass spectrometry is an analytical technique used to determine the relative abundance and mass of isotopes.
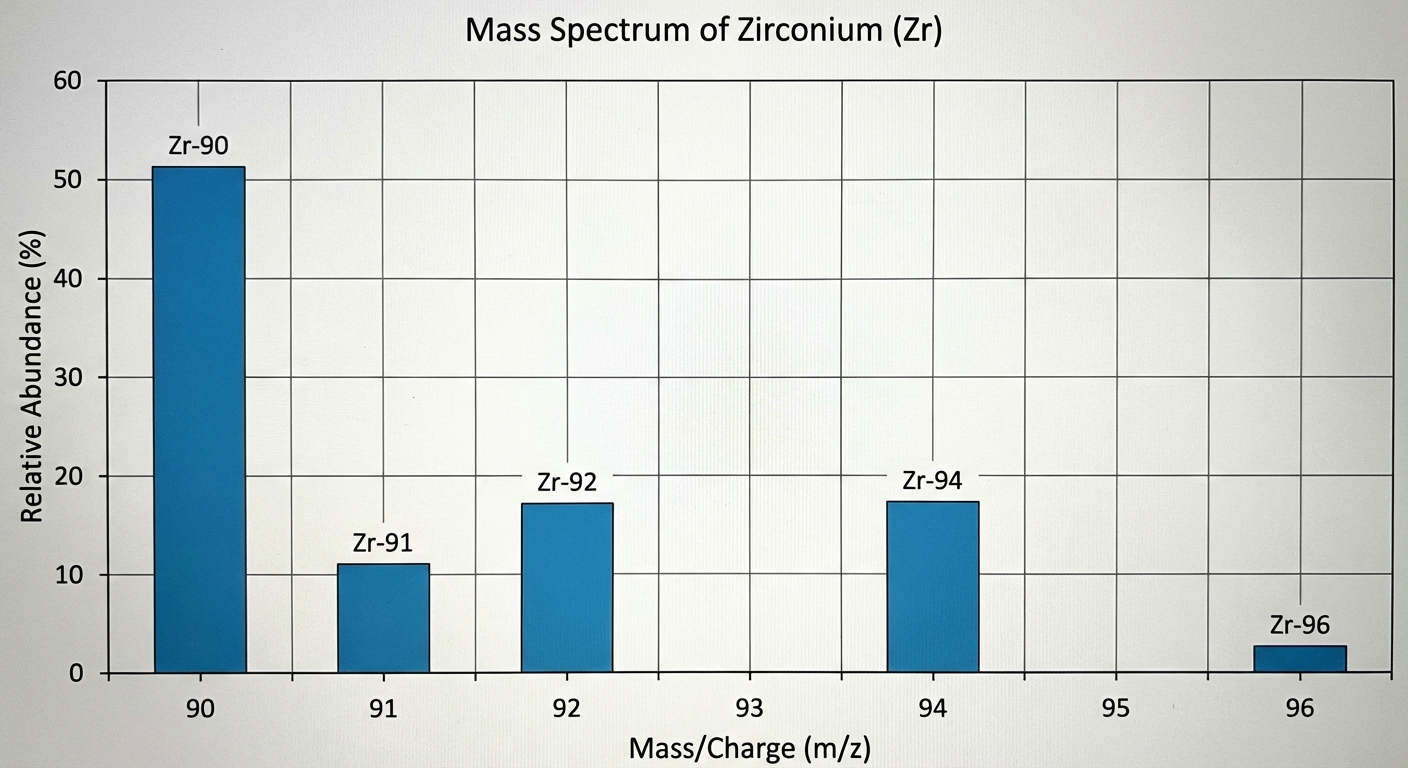
Interpreting the Graph:
- X-axis (m/z): Represents the mass-to-charge ratio. Since charge is usually +1, this essentially represents the mass (amu) of the isotope.
- Y-axis (Relative Intensity): Represents the percent abundance of that isotope.
- Analysis: The number of peaks represents the number of isotopes. The height represents how common that isotope is.
Review Example: If Selenium has peaks at masses 74, 76, 77, 78, 80, and 82, and the peak at 80 is the highest, the average atomic mass will be closest to 80.
Atomic Structure and Electron Configuration
Coulomb’s Law
This is the most important concept in Unit 1 for explaining atomic structure and periodic trends.
- $F_E$: Force of attraction/repulsion.
- $q1, q2$: Magnitudes of the charges (proton count vs. electron charge).
- $r$: Distance between the charged particles.
Key Takeaway:
- Greater Charge ($q$) $\rightarrow$ Stronger attraction (Higher ionization energy).
- Greater Distance ($r$) $\rightarrow$ Weaker attraction (Lower ionization energy).
Electron Configuration
Electrons are arranged in shells ($n$), subshells ($l$), and orbitals.
- Subshells:
- s: 1 orbital (max 2 electrons) — Sphere shape.
- p: 3 orbitals (max 6 electrons) — Dumbbell shape.
- d: 5 orbitals (max 10 electrons).
- f: 7 orbitals (max 14 electrons).
Rules for Filling (The Aufbau Principle):
Electrons fill the lowest energy orbitals first ($1s \rightarrow 2s \rightarrow 2p \rightarrow 3s \rightarrow 3p \rightarrow 4s \rightarrow 3d$…). Note that $4s$ fills before $3d$ due to energy overlaps.
Common Exceptions:
- Chromium ($Cr$): $[Ar] 4s^1 3d^5$ (Half-filled d-subshell is more stable).
- Copper ($Cu$): $[Ar] 4s^1 3d^{10}$ (Fully-filled d-subshell is more stable).
Hund’s Rule & Pauli Exclusion
- Hund’s Rule: Electrons fill degenerate orbitals (orbitals of equal energy, like the three $p$ orbitals) singly before pairing up.
- Pauli Exclusion Principle: No two electrons can have the same quantum state. Electrons in the same orbital must have opposite spins (up and down).
Photoelectron Spectroscopy (PES)
PES provides experimental evidence for the shell and subshell model of the atom.
How PES Works
High-energy photons hit an atom, ejecting electrons. The energy of the photon is used to overcome the binding energy ($BE$) of the electron vs the nucleus.
Interpreting PES Spectra
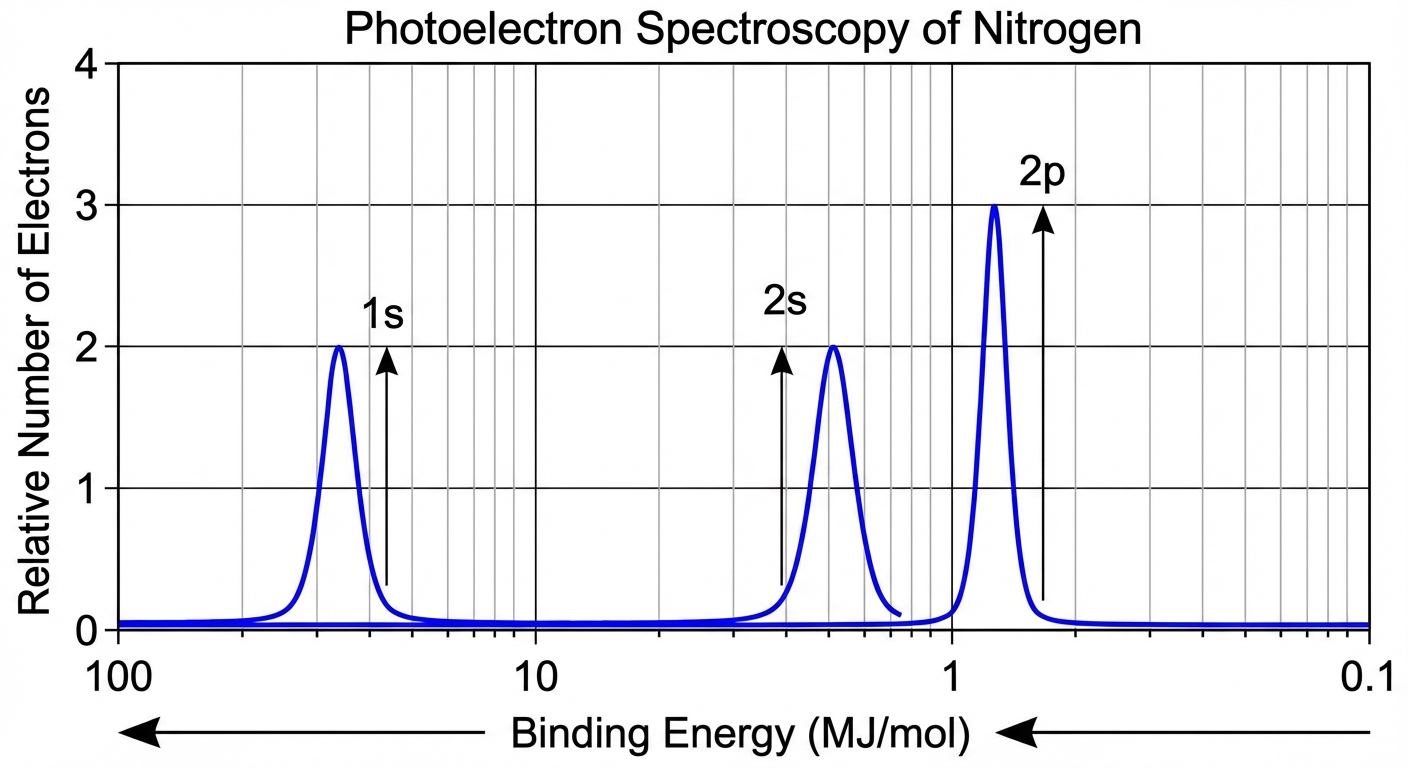
- Position (X-axis): Represents Binding Energy. High energy (left side) corresponds to electrons closest to the nucleus (1s). Low energy (right side) corresponds to valence electrons.
- Peak Height: Proportional to the number of electrons in that subshell. A peak for $2p^6$ will be three times as tall as a peak for $2s^2$.
Example Analysis:
If a PES graph shows:
- Peak A at 100 MJ/mol (Height 2)
- Peak B at 10 MJ/mol (Height 2)
- Peak C at 1 MJ/mol (Height 3)
Configuration: $1s^2 2s^2 2p^3$. The element is Nitrogen.
Periodic Trends
Periodic trends are explained by two opposing factors: Effective Nuclear Charge ($Z_{eff}$) and Shielding/Distance.
1. Effective Nuclear Charge ($Z_{eff}$)
- The net positive charge experienced by valence electrons.
- Trend: Constant down a group, Increases across a period.
2. Atomic Radius
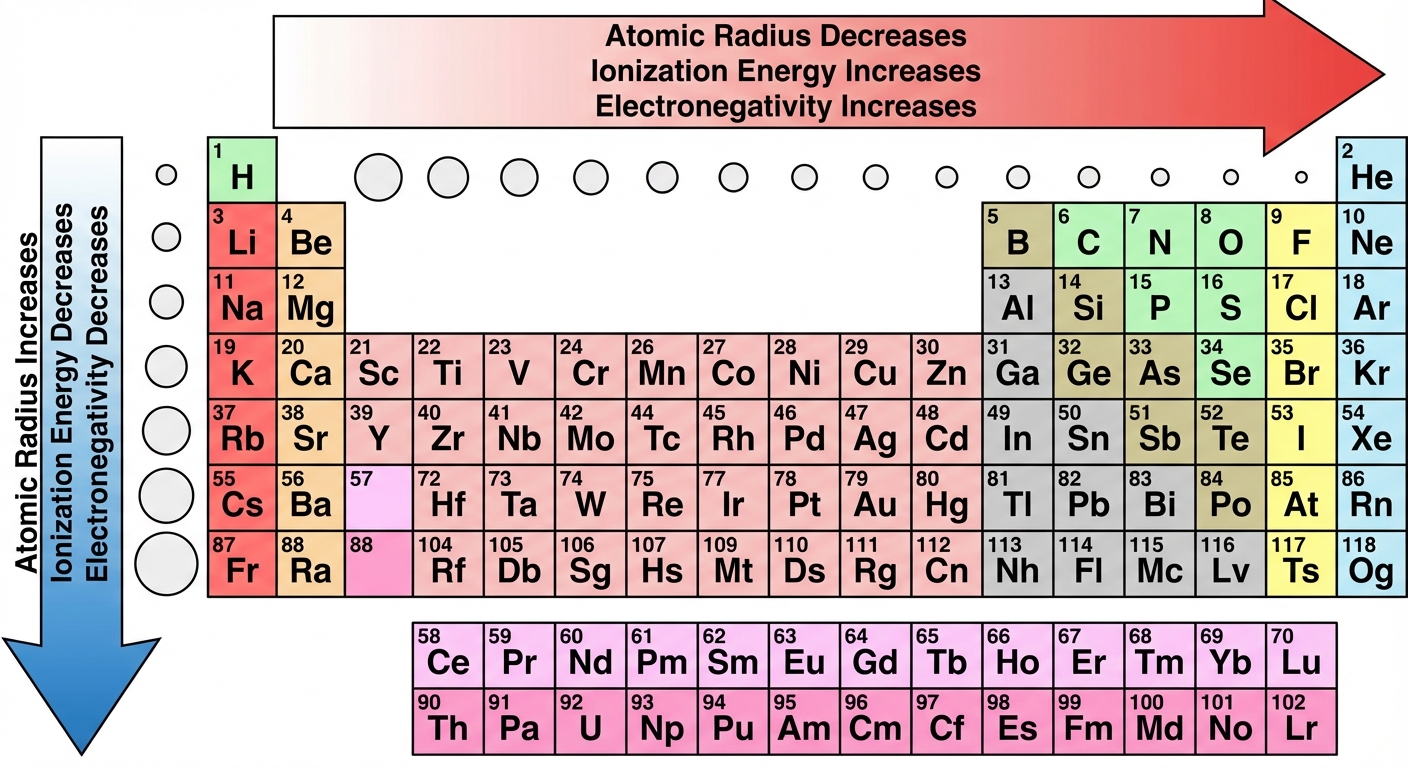
- Trend Across a Period: Decreases. (Protons increase $\rightarrow$ higher attraction pulls shell closer).
- Trend Down a Group: Increases. (More shells are added $\rightarrow$ increased distance and shielding).
3. Ionic Radius
- Cations (+): Smaller than their parent atom. Loss of valence shell and reduced electron-electron repulsion.
- Anions (-): Larger than their parent atom. Added electrons increase electron-electron repulsion, expanding the cloud.
- Isoelectronic Series: For ions with the same electron config (e.g., $O^{2-}, F^-, Na^+, Mg^{2+}$), the one with the most protons is the smallest (strongest pull).
4. Ionization Energy (IE)
- Definition: Energy required to remove the outermost electron from a gas-phase atom.
- Trend Across a Period: Increases. ($Z_{eff}$ increases, radius decreases $\rightarrow$ harder to remove).
- Trend Down a Group: Decreases. (Shielding increases, radius increases $\rightarrow$ easier to remove).
- Important Discontinuities:
- Be vs. B: Boron has lower IE than Beryllium because the $2p$ electron is higher energy/shielded by the $2s$.
- N vs. O: Oxygen has lower IE than Nitrogen because of electron-electron repulsion in the paired $2p$ orbital of Oxygen.
5. Electronegativity
- Definition: The ability of an atom in a molecule to attract shared electrons.
- Trend: Increases bottom-left to top-right.
- Fluorine is the most electronegative element (4.0).
Common Mistakes & Pitfalls
- Mass Number vs. Atomic Mass: Do not confuse Mass Number (Protons + Neutrons, an integer) with Average Atomic Mass (weighted average, a decimal). $C-12$ has a mass number of 12, but Carbon's atomic mass is 12.011.
- Coulomb's Law Justification: When explaining trends across a period, argue using effective nuclear charge ($Z_{eff}$ / proton count). When explaining down a group, argue using number of shells (distance/shielding).
- PES Graph Orientation: Pay attention to the x-axis. Sometimes high energy is on the left, sometimes on the right. Always look for the highest energy peak to identify the $1s$ orbital.
- Molar Mass of Diatomics: "Nitrogen gas" refers to $N_2$, not $N$. Its molar mass is $28.02 \text{ g/mol}$, not $14.01$.
- Removing Electrons from Transition Metals: When forming cations for transition metals (e.g., $Fe$ to $Fe^{2+}$), remove electrons from the s-orbital first, then the d-orbital. ($Fe: [Ar] 4s^2 3d^6 \rightarrow Fe^{2+}: [Ar] 3d^6$).