AP Chemistry Unit 8: Complex Acid-Base Equilibria
Acid-Base Reactions and Buffers
Acid-base reactions in AP Chemistry move beyond simple neutralization properties into the realm of chemical equilibrium. The most critical application of this equilibrium is the concept of Buffers.
The Common Ion Effect
Before understanding buffers, you must understand the Common Ion Effect. This is the shift in equilibrium caused by the addition of a compound having an ion in common with the dissolved substance.
- Example: If you have a solution of acetic acid ($CH3COOH$) and you add sodium acetate ($CH3COONa$), you are adding the common ion acetate ($CH_3COO^-$).
- Le Châtelier's Principle: The addition of product ($CH_3COO^-$) shifts the equilibrium to the left, suppressing the ionization of the acid and increasing the pH.
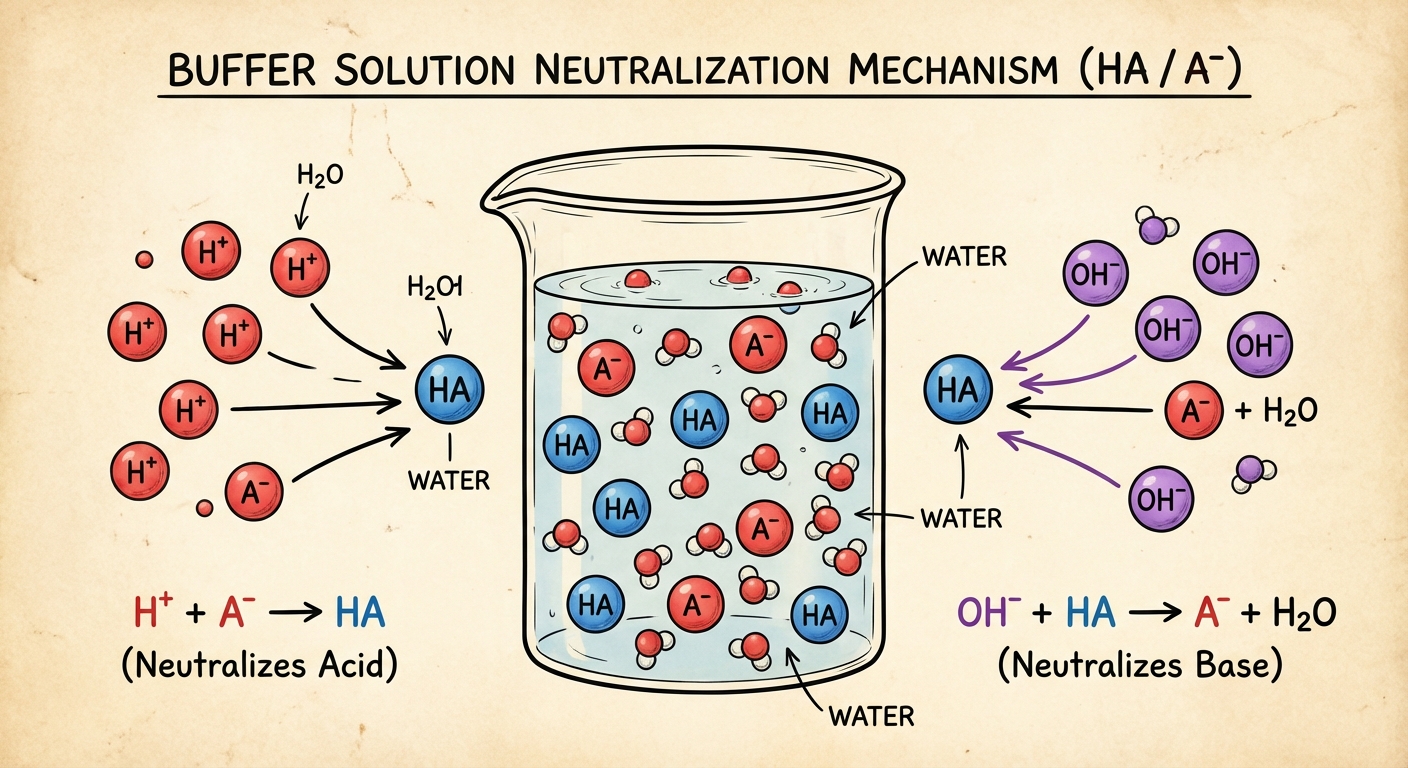
Buffer Solutions
A Buffer is a solution that resists drastic changes in pH when small amounts of strong acid or strong base are added.
Composition of a Buffer:
- A Weak Acid and its Conjugate Base (e.g., $HF$ and $F^-$)
- A Weak Base and its Conjugate Acid (e.g., $NH3$ and $NH4^+$)
Note: A strong acid and its conjugate base do NOT form a buffer because the conjugate base of a strong acid has negligible basicity.
The Henderson-Hasselbalch Equation
This is the governing equation for calculating the pH of buffer solutions. It is derived from the $K_a$ expression.
Where:
- $pKa = -\log(Ka)$
- $[A^-]$ is the concentration of the conjugate base.
- $[HA]$ is the concentration of the weak acid.
Buffer Capacity
Buffer Capacity refers to the amount of acid or base a buffer can neutralize before the pH begins to change appreciably.
- Concentration: Higher concentrations of acid/base components ($[HA]$ and $[A^-]$) result in higher buffer capacity.
- Ratio: A buffer is most effective when the ratio $\frac{[A^-]}{[HA]}$ is close to 1. This occurs when $pH \approx pK_a$.
Acid-Base Titrations
A Titration is a quantitative analytical method used to determine the concentration of an identified analyte by reacting it with a standard solution (titrant) of known concentration.
Key Definitions
- Equivalence Point: The point where the moles of titrant added stoichiometrically equal the moles of substance being analyzed (moles $H^+ = $ moles $OH^-$).
- Endpoint: The point where the indicator changes color. Ideally, the endpoint should occur very close to the equivalence point.
Reaction Types and Net Ionic Equations
Students often lose points by writing molecular equations instead of net ionic equations.
| Titration Type | Net Ionic Equation | Equivalence $pH$ | Explanation |
|---|---|---|---|
| Strong Acid + Strong Base | $H^+ + OH^- \rightarrow H_2O$ | $pH = 7$ | Resulting ions are neutral (spectator ions). |
| Weak Acid + Strong Base | $HA + OH^- \rightarrow A^- + H_2O$ | $pH > 7$ | The conjugate base ($A^-$) hydrolysis produces $OH^-$. |
| Weak Base + Strong Acid | $B + H^+ \rightarrow BH^+$ | $pH < 7$ | The conjugate acid ($BH^+$) hydrolysis produces $H^+$. |
Analyzing Titration Curves
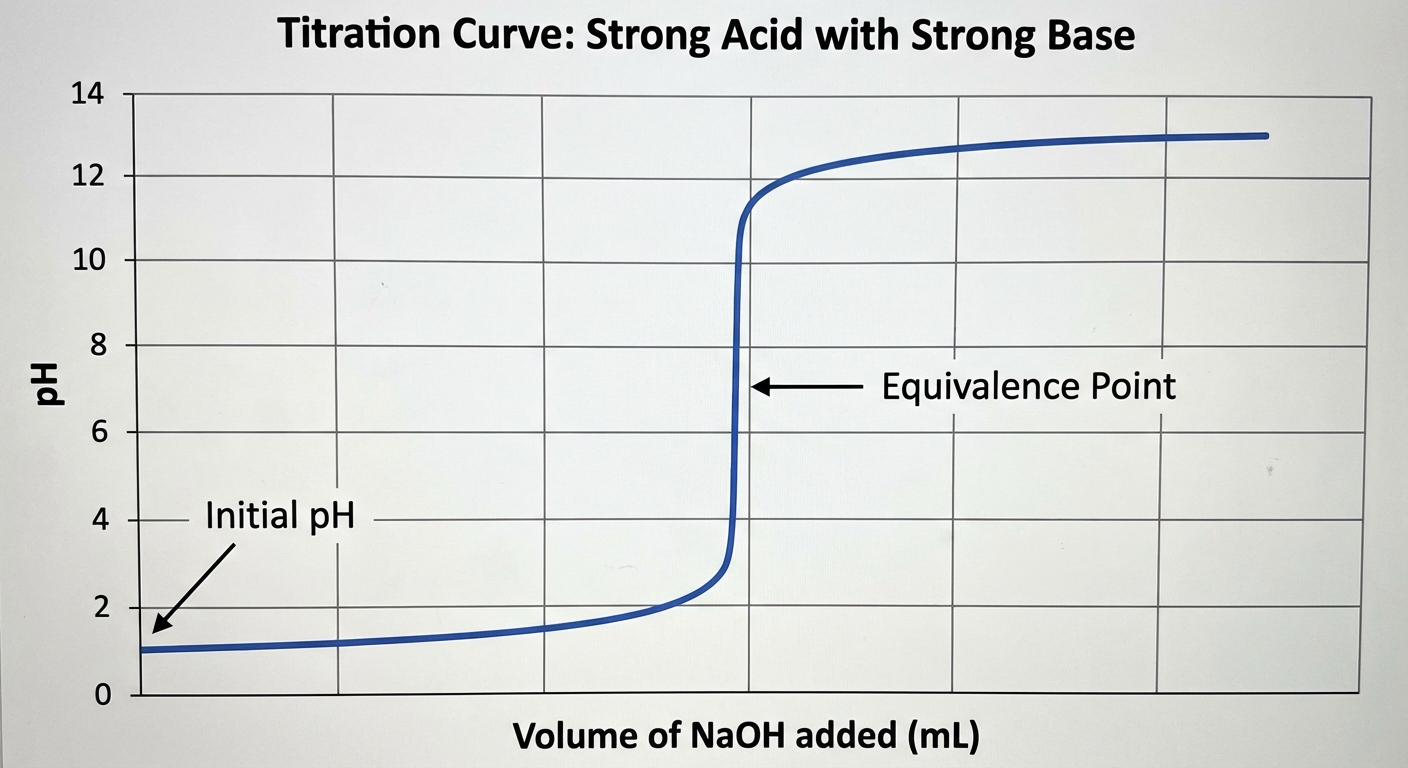
1. Strong Acid - Strong Base Titration
- Initial pH: Very low (determines initial concentration).
- Shape: Flat for a long time, then a very steep, vertical rise near the equivalence point.
- Equivalence Point: Exactly at pH 7.0 at $25^{\circ}C$.
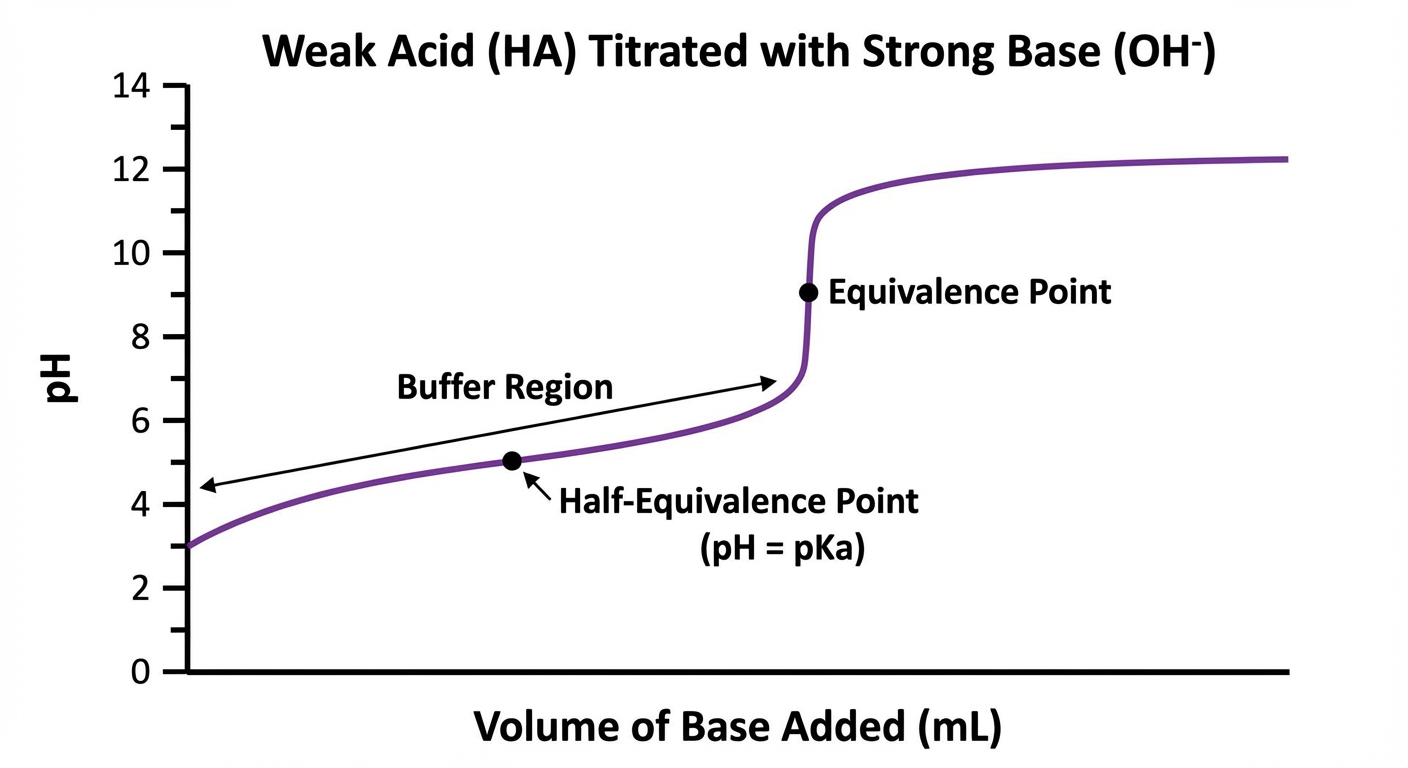
2. Weak Acid - Strong Base Titration
This is the most frequently tested scenario in AP Chemistry.
- Initial pH: Higher than a strong acid (usually pH 3-5).
- Buffer Region: As base is added, $HA$ converts to $A^-$. The solution creates a buffer. The curve rises slowly here.
- Half-Equivalence Point: This is the most critical concept for weak acid titrations. At the volume where half of the acid has been neutralized:
- $[HA] = [A^-]$
- Therefore, $\log(1) = 0$
- Equivalence Point: The pH is basic (above 7). To calculate this pH, you must treat the problem as a weak base equilibrium problem using the concentration of the conjugate base formed.
Calculating pH During Weak-Strong Titration
To solve these problems, follow this strict two-step process:
- Stoichiometry (BCA Table): Use moles. Calculate the reaction between the acid/base to completion. Determine what remains.
- Equilibrium:
- If weak acid + conjugate base remain $\rightarrow$ Use Henderson-Hasselbalch.
- If only conjugate base remains $\rightarrow$ Use $K_b$ and an ICE table (this is the equivalence point).
- If excess strong base remains $\rightarrow$ Calculate $[OH^-]$ from excess moles / total volume.
pH and pKa
Understanding the relationship between relative pH and pKa is essential for predicting the structure and charge of species in solution.
The Relationship Defined
The $pKa$ is a measure of the strength of an acid (lower $pKa$ = stronger acid). The relationship between the solution's pH and the acid's $pK_a$ determines the dominant species.
- If $pH < pK_a$: The environment is too acidic. The species holds onto its proton. Protonated form ($HA$) dominates.
- If $pH > pK_a$: The environment is basic enough to pull the proton off. Deprotonated form ($A^-$) dominates.
- If $pH = pK_a$: $[HA] = [A^-]$.
Application: Polyprotic Acids
For acids with multiple protons (like $H3PO4$), there are multiple $pKa$ values ($pK{a1}, pK{a2}, pK{a3}$).
- A titration curve for a polyprotic acid will show multiple "humps" or equivalence points.
- The dominant species changes as pH crosses each specific $pK_a$ threshold.
Common Mistakes & Pitfalls
- "The Equivalence Point is always pH 7."
- Correction: This is ONLY true for Strong Acid/Strong Base titrations. For Weak/Strong, the conjugate salt hydrolyzes, changing the pH.
- Molarity vs. Moles in Titrations.
- Correction: When combining solutions during a titration, you have a new total volume. Always do stoichiometry in moles first, then divide by the new total volume to get concentration for equilibrium calculations.
- Mixing up $Ka$ and $Kb$.
- Correction: At the equivalence point of a weak acid titration, you have the conjugate base. You must convert the given $Ka$ to $Kb$ using $Kw = Ka \times K_b$ before constructing your ICE table.
- Misidentifying the Buffer Region.
- Correction: Remember that the buffer region occurs before the equivalence point. Once you pass equivalence, the pH is determined solely by the excess strong titrant.