Comprehensive Guide to Classification of Chemical Transformations
Fundamental Reaction Classifications
In AP Chemistry, understanding the nature of chemical changes is foundational. A chemical reaction involves the breaking of intramolecular bonds (within molecules) and the formation of new ones, resulting in substances with new properties.
While reactions are often complex, categorize them into broad patterns helps predict products. The three major overarching categories in the AP curriculum are Precipitation, Acid-Base, and Oxidation-Reduction (Redox). However, we first review the fundamental patterns.
Synthesis and Decomposition
Synthesis (Combination): Two or more substances combine to form a single product.
- General Form:
- Example: Formation of ammonia (Haber Process).
Decomposition: A single compound breaks down into two or more simpler substances.
- General Form:
- Example: Decomposition of Hydrogen Peroxide.
Combustion
Combustion is a specific type of redox reaction where a substance reacts vigorously with oxygen gas ($O_2$), releasing energy in the form of light and heat.
- Hydrocarbon Combustion: When a hydrocarbon ($CxHy$) or carbohydrate acts as fuel, the products are always carbon dioxide ($CO2$) and water vapor ($H2O$).
Aqueous Reactions and Precipitation
Many reactions in chemistry occur in an aqueous environment (dissolved in water). Understanding how ions interact in solution is critical for writing Net Ionic Equations.
Double Replacement (Metathesis)
In these reactions, the cations and anions of two different compounds switch places.
For a reaction to actually occur (and not just result in a physical mixture of ions), one of the products must drive the reaction by leaving the solution phase as a:
- Precipitate (Solid)
- Gas
- Weak Electrolyte (usually Water)
Solubility Rules (The "SNAP" Rule)
While you do not need to memorize the entire solubility table for the AP exam, you MUST know which ions generally remain soluble (do not form precipitates).
Memory Aid: SNAP
- Sodium ($Na^+$)
- Nitrate ($NO_3^-$)
- Ammonium ($NH_4^+$)
- Potassium ($K^+$)
Note: Acetate ($C2H3O_2^-$) is also generally soluble.
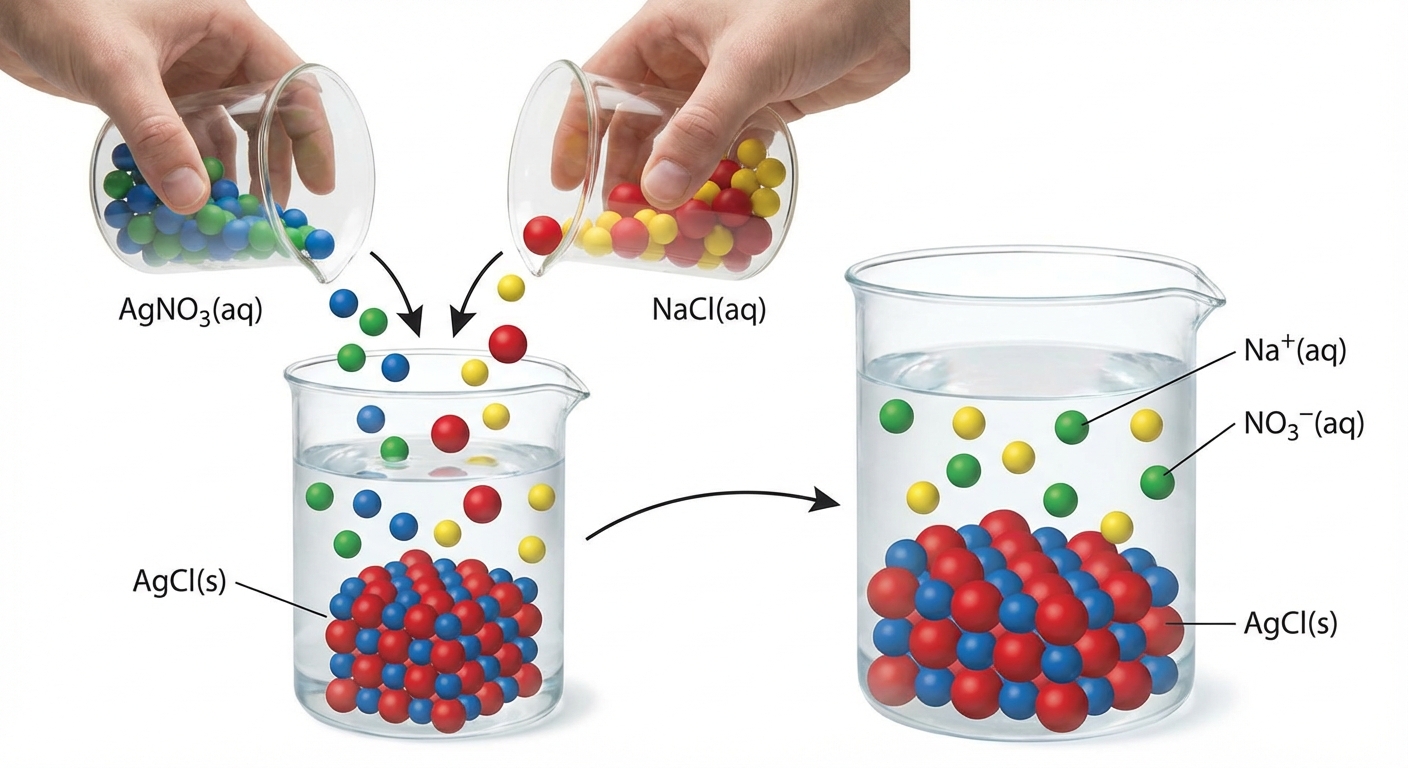
Writing Net Ionic Equations
To represent the actual chemical change, we strip away the "spectator ions" (ions that do not change state or charge).
- Molecular Equation: Shows neutral formulas.
- Complete Ionic Equation: Dissociate all strong electrolytes (soluble salts, strong acids/bases) into ions.
- Net Ionic Equation: Cancel out species that appear identical on both sides (Spectators: $Na^+$ and $NO_3^-$).
Introduction to Acid-Base Reactions
In Unit 4, we focus specifically on the transfer of protons ($H^+$). While complex equilibrium is covered in Unit 8, you must identify Brønsted-Lowry acids and bases here.
Concepts and Definitions
- Brønsted-Lowry Acid: A proton ($H^+$) donor.
- Brønsted-Lowry Base: A proton ($H^+$) acceptor.
Water plays a unique role as an amphoteric substance—it can act as either an acid or a base depending on what it reacts with.
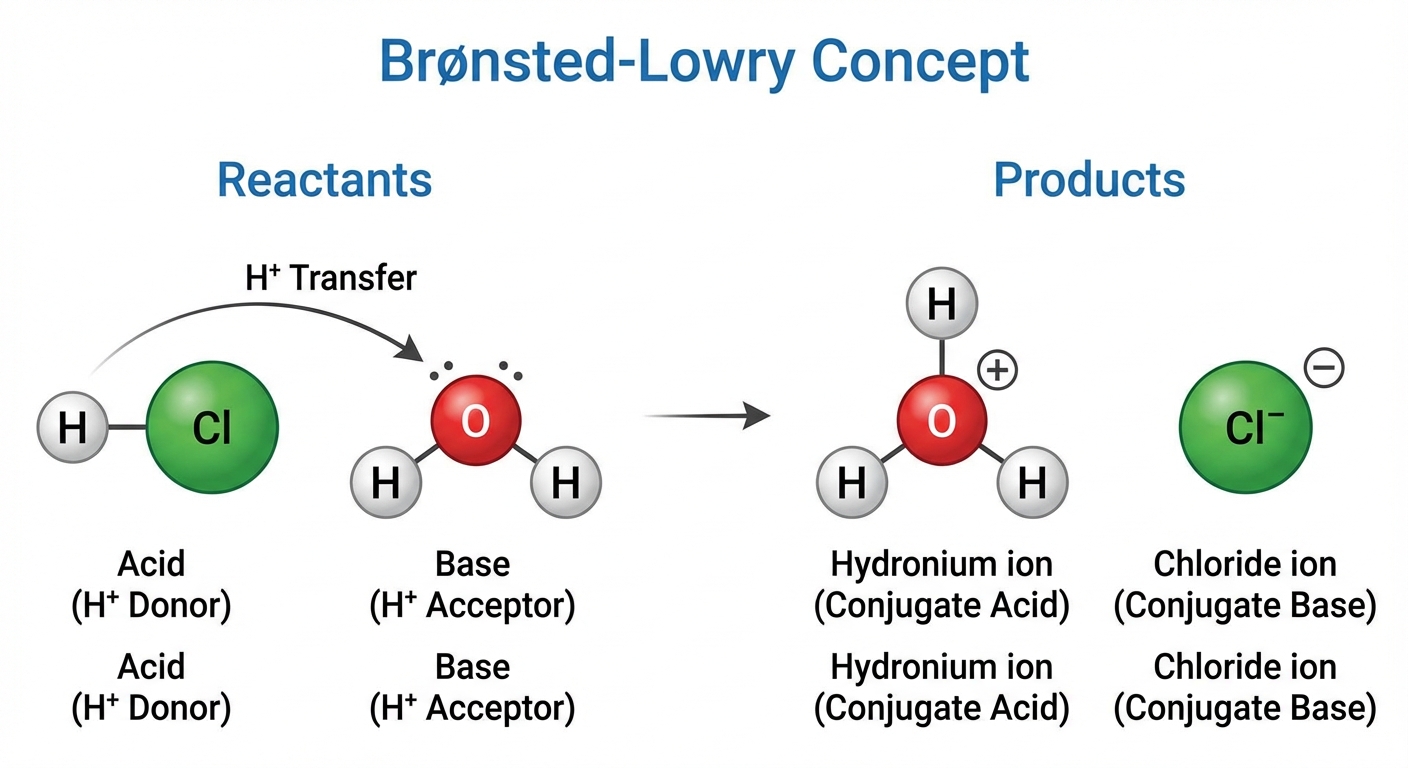
Neutralization Reactions
A specific type of double replacement where an acid reacts with a base to produce a salt (ionic compound) and water.
Net Ionic Equation for Strong Acid/Strong Base:
Since $HCl$, $NaOH$, and $NaCl$ are strong electrolytes, they dissociate. The net result is the formation of water:
Gas-Forming Acid-Base Reactions
Some neutralization reactions produce an unstable intermediate (like carbonic acid, $H2CO3$) that immediately decomposes into a gas.
- Carbonates/Bicarbonates + Acid $\rightarrow$ Salt + $CO2$ + $H2O$
Oxidation-Reduction (Redox) Reactions
Redox reactions are characterized by the transfer of electrons between species, changing the oxidation states of the elements involved.
Mnemonics for Electron Transfer
- OIL RIG: Oxidation Is Loss (of electrons); Reduction Is Gain (of electrons).
- LEO the lion says GER: Lose Electrons = Oxidation; Gain Electrons = Reduction.
Determining Oxidation Numbers
To identify if a reaction is redox, assign oxidation numbers to each atom. If the numbers change from reactants to products, it is redox.
Common Rules (in hierarchical order):
- Pure Elements: Always 0 (e.g., $Na$, $O_2$, $C$).
- Monatomic Ions: Equal to their charge (e.g., $Na^+ = +1$, $Cl^- = -1$).
- Fluorine: Always -1 in compounds.
- Oxygen: Usually -2 (Except in peroxides like $H2O2$, where it is -1).
- Hydrogen: +1 with nonmetals; -1 with metals (hydrides).
- Sum Rule: The sum of oxidation numbers in a neutral compound is 0; in a polyatomic ion, it equals the ion's charge.
Analysis Example
- Reactants: $Fe$ is 0 (element); $Cl$ is 0 (element).
- Products: $Fe$ is +3; $Cl$ is -1.
- $Fe$ changed from 0 $\rightarrow$ +3. It lost electrons (Oxidized).
- $Cl$ changed from 0 $\rightarrow$ -1. It gained electrons (Reduced).
Half-Reactions
We can separate the oxidation and reduction processes to visualize electron flow:
- Oxidation Half:
- Reduction Half:
(Note: To balance the full equation, you must equalize the electrons transferred).
Common Mistakes & Pitfalls
- Confusing Charge with Oxidation Number: While often the same value, oxidation numbers are a bookkeeping method for electrons, while charge is a real physical property. Write charges as $2+$ and oxidation numbers as $+2$.
- Spectator Ions in Net Ionic Equations: Students often forget to remove spectators or identify substances that don't dissociate (solids, liquids, gases, and weak acids) as ions. Only strong electrolytes break apart.
- Combustion Products: A common error is writing $H2$ instead of $H2O$ as a product of hydrocarbon combustion.
- Redox Agents: The "Oxidizing Agent" is the substance getting reduced, and the "Reducing Agent" is the substance getting oxidized. This inverse naming often trips students up.