AP Chemistry Unit 7: Advanced Equilibrium Concepts
Introduction to Le Chatelier's Principle
Le Chatelier's Principle is a qualitative tool used to predict how a system at equilibrium responds to external changes. The principle states that if a dynamic equilibrium is disturbed by changing the conditions, the position of equilibrium moves to counteract the change.
Types of Stress
Concentration Changes:
- Adding Reactant/Product: The system shifts away from the added component to consume it.
- Removing Reactant/Product: The system shifts toward the removed component to replace it.
- Note: Adding a pure solid or liquid does not shift equilibrium because their concentrations (density) remain constant.
Pressure/Volume Changes (Gaseous Systems Only):
- Increasing Pressure (Decreasing Volume): The system shifts toward the side with fewer moles of gas to reduce pressure.
- Decreasing Pressure (Increasing Volume): The system shifts toward the side with more moles of gas to restore pressure.
- Note: If moles of gas are equal on both sides ($\Delta n_{gas} = 0$), pressure changes cause no shift. Adding an inert gas (like Argon) at constant volume increases total pressure but does not change partial pressures of reacting species, so no shift occurs.
Temperature Changes:
- Temperature is the only stress that changes the value of the equilibrium constant ($K$).
- Exothermic Reaction ($H < 0$): Treat heat as a product. Increasing $T$ adds product, shifting equilibrium left ($K$ decreases).
- Endothermic Reaction ($H > 0$): Treat heat as a reactant. Increasing $T$ adds reactant, shifting equilibrium right ($K$ increases).
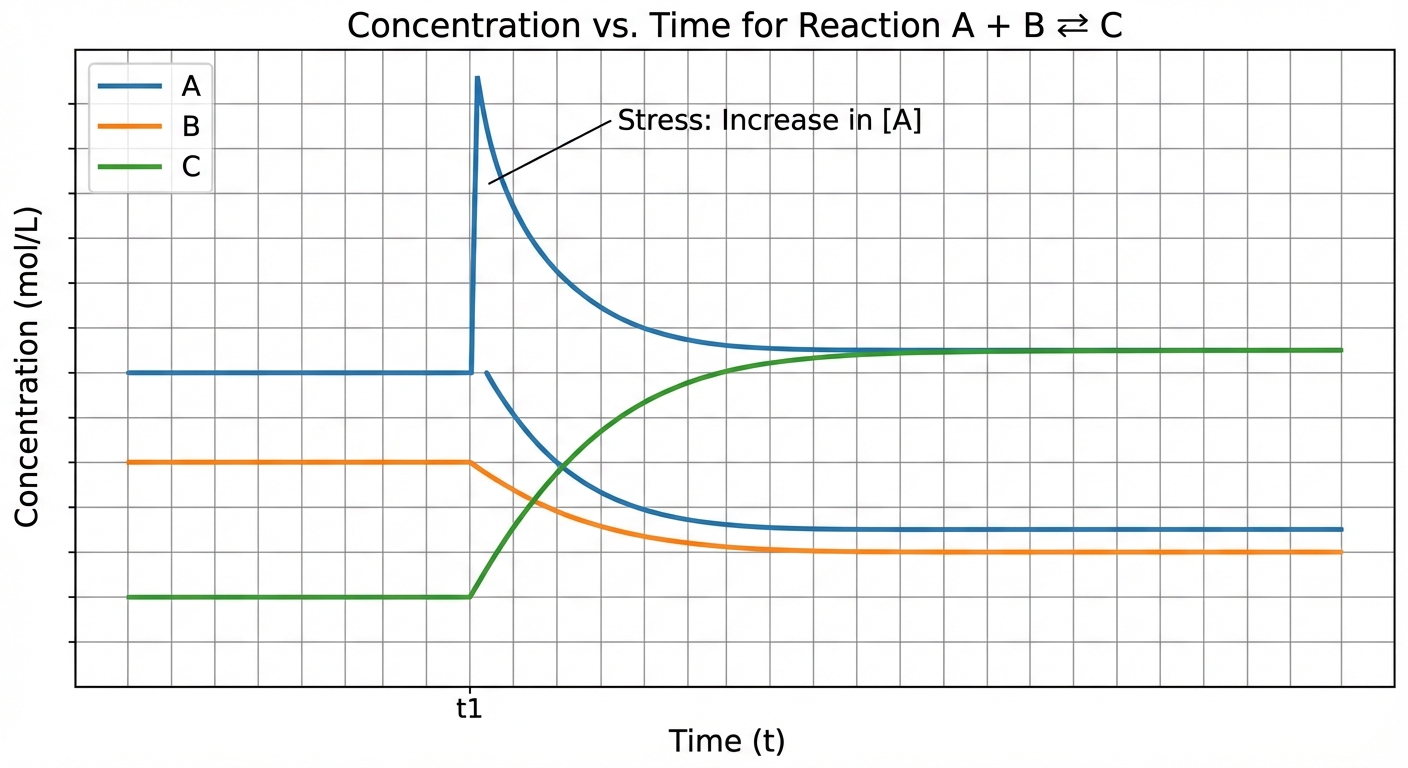
Reaction Quotient and Le Chatelier's Principle
While Le Chatelier's Principle is qualitative, the Reaction Quotient ($Q$) provides the mathematical justification for these shifts. $Q$ is calculated using the same expression as $K$, but often with non-equilibrium concentrations immediately after a stress is applied.
Comparing $Q$ and $K$
To determine the direction of the shift, compare the instantaneous $Q$ to the known $K$:
- $Q < K$: The ratio of products to reactants is too small. The forward reaction rate exceeds the reverse rate. The system shifts RIGHT (towards products) until equilibrium is re-established.
- $Q > K$: The ratio of products to reactants is too high. The reverse reaction rate exceeds the forward rate. The system shifts LEFT (towards reactants).
- $Q = K$: The system is at equilibrium.
Example: Dilution of Aqueous Solutions
For the reaction:
If water is added to dilute the solution, all concentrations decrease. Because the denominator ($[Fe^{3+}][SCN^-]$) has a higher order than the numerator, $Q$ becomes greater than $K$ (mathematically dependent on stoichiometry), causing a shift.
Introduction to Solubility Equilibria
Specifically for ionic compounds that satisfy the definition of "insoluble" or "slightly soluble," an equilibrium exists between the solid salt and its dissolved ions.
The Solubility Product Constant ($K_{sp}$)
For a generic salt $Mx Ay$ dissolving:
The equilibrium expression is:
- Important: Solids are omitted from the expression. $K_{sp}$ is a constant value at a specific temperature.
Molar Solubility ($s$)
Molar Solubility ($s$) is the number of moles of the solid that dissolve per liter of solution to reach saturation. It is distinct from $K_{sp}$.
Common Stoichiometries:
1:1 Ratio (e.g., $AgCl$):
1:2 Ratio (e.g., $PbCl2$):
1:3 Ratio (e.g., $Al(OH)3$):
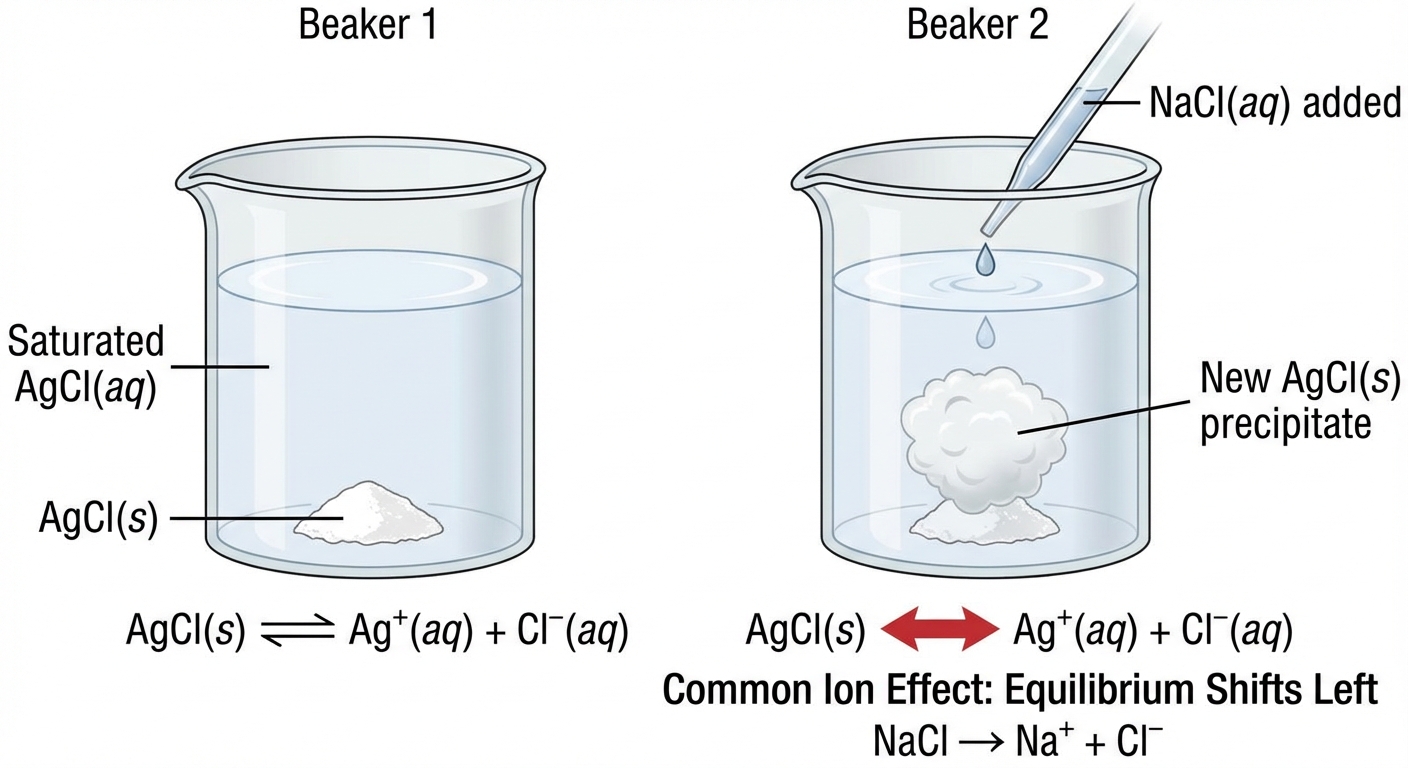
Common-Ion Effect
The Common-Ion Effect describes the decrease in solubility of an ionic precipitate when a soluble compound containing one of the same ions is added to the solution.
Conceptual Explanation
Consider a saturated solution of Lead(II) Chromate:
If we add $Na2CrO4$ (a soluble salt) to this mixture, the concentration of chromate ions $[CrO4^{2-}]$ increases drastically. According to Le Chatelier's Principle, the system tries to consume the excess chromate by shifting left. This results in more solid $PbCrO4$ precipitating out of solution.
Calculation Strategy
- Set up an ICE table.
- The initial concentration of the common ion is not zero (it equals the concentration of the added soluble salt).
- Assume $x$ (the amount of solid that dissolves) is very small relative to the added ion concentration, allowing for the approximation: $(0.10 + x) \approx 0.10$.
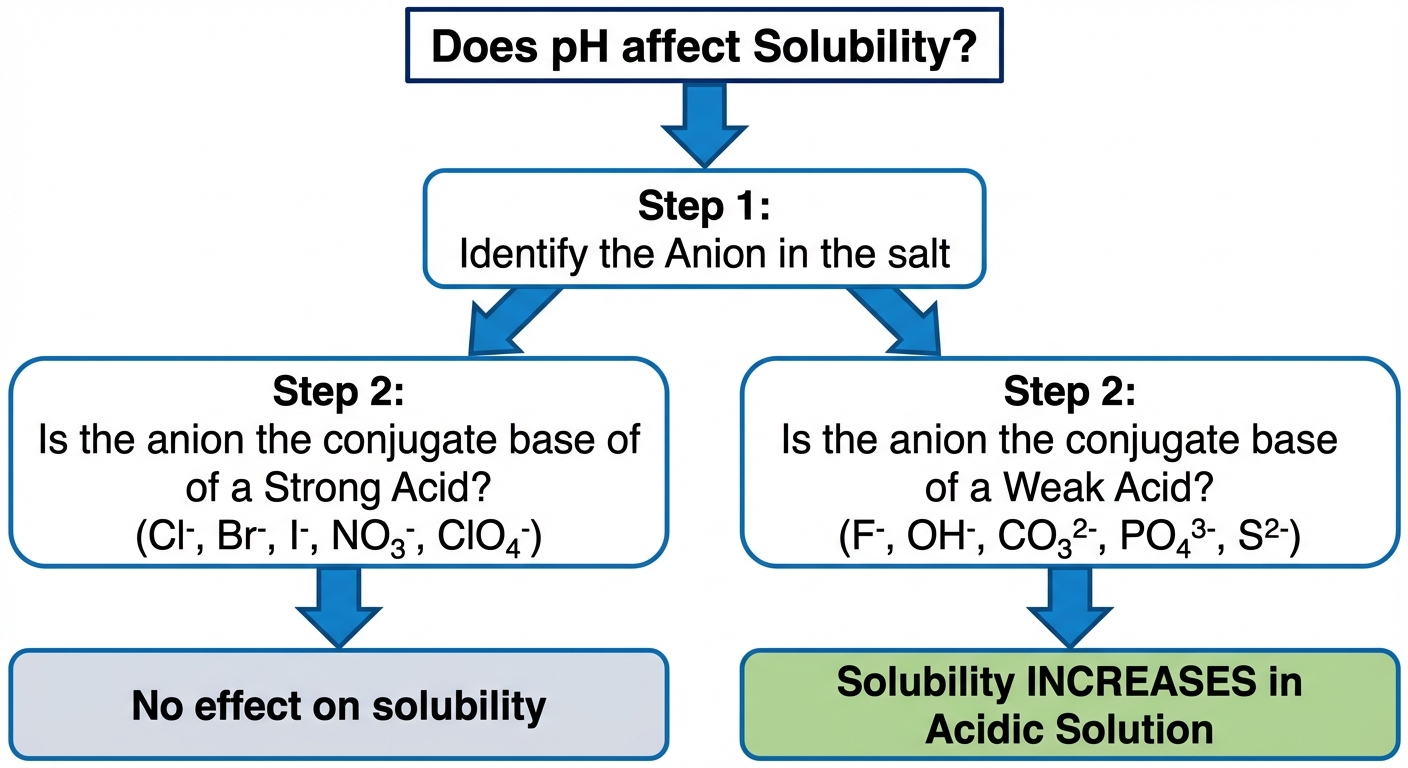
pH and Solubility
The solubility of ionic compounds containing basic anions or acidic cations is heavily dependent on pH. This is a specific application of Le Chatelier coupled with acid-base chemistry.
Salts with Basic Anions
If the anion of the salt is the conjugate base of a weak acid, decreasing pH (adding acid/$H^+$) increases solubility.
Example: Magnesium Hydroxide
- Add Acid ($H^+$): The $H^+$ reacts with $OH^-$ to form water: $H^+ + OH^- \rightarrow H_2O$.
- Effect: This effectively removes the product $OH^-$ from the solubility equilibrium. By Le Chatelier's Principle, the system shifts right to replenish the $OH^-$.
- Result: More solid $Mg(OH)_2$ dissolves.
Rule of Thumb:
- If Anion is from a Weak Acid (e.g., $F^-$, $CO_3^{2-}$, $OH^-$): Solubility increases in acidic solution.
- If Anion is from a Strong Acid (e.g., $Cl^-$, $Br^-$, $NO_3^-$): Solubility is unaffected by pH (because these ions are negligible bases and do not accept protons).
Free Energy of Dissolution
The dissolution of a salt is governed by thermodynamics. At equilibrium (saturation), the Gibbs Free Energy change is zero (\Delta G = 0). Under standard conditions, we relate free energy to equilibrium constants.
The Relationship
- $R$: 8.314 J/mol\u00b7K (Universal Gas Constant)
- $T$: Temperature in Kelvin
Enthalpy and Entropy Factors
- Entropy (\Delta S_{soln}): usually positive ($>0$) because ions dispersed in solution have more disorder than a crystal lattice structure.
- Enthalpy (\Delta H_{soln}): Can be endothermic or exothermic. Breaking the lattice requires energy (endothermic), while hydration of ions releases energy (exothermic).
If dissolution is endothermic (\Delta H > 0): heat is a reactant. Increasing $T$ shifts equilibrium right, increasing $K_{sp}$. This is why most solids become more soluble in hot water.
Common Mistakes & Pitfalls
- Including Solids/Liquids in $Q$ or $K$: Remember that pure substances have an activity of 1. Never put $[AgCl(s)]$ in the denominator.
- Confusing Solubility ($s$) with $K{sp}$: $s$ is a molar concentration (M), while $K{sp}$ is a unitless equilibrium constant. You often must convert between them.
- The "2x squared" Error: When solving for $s$ in $PbCl2$, students often write $K{sp} = [Pb][Cl]^2$ and plug in $(s)(s)^2$. It should be $(s)(2s)^2 = 4s^3$ because the concentration of Chloride is twice the solubility.
- Catalysts and Equilibrium: A catalyst speeds up both the forward and reverse reactions equally. It helps reach equilibrium faster but does not change the position of equilibrium or the value of $K$.
- Inert Gases: Adding Helium to a gas equilibrium at constant volume increases total pressure but does not shift equilibrium because partial pressures of reactants/products remain unchanged.