Unit 9 Study Guide: Electrochemistry and Thermodynamics
Galvanic (Voltaic) and Electrolytic Cells
Electrochemistry is the study of the interchange of chemical and electrical energy. This exchange occurs through Redox (oxidation-reduction) reactions. The two main types of electrochemical cells—Galvanic and Electrolytic—differ primarily in terms of spontaneity.
Basics of Redox and Electrodes
Before analyzing specific cells, you must master the fundamental definitions governing electron movement.
- Oxidation: Loss of electrons (oxidation number increases). Occurs at the Anode.
- Reduction: Gain of electrons (oxidation number decreases). Occurs at the Cathode.
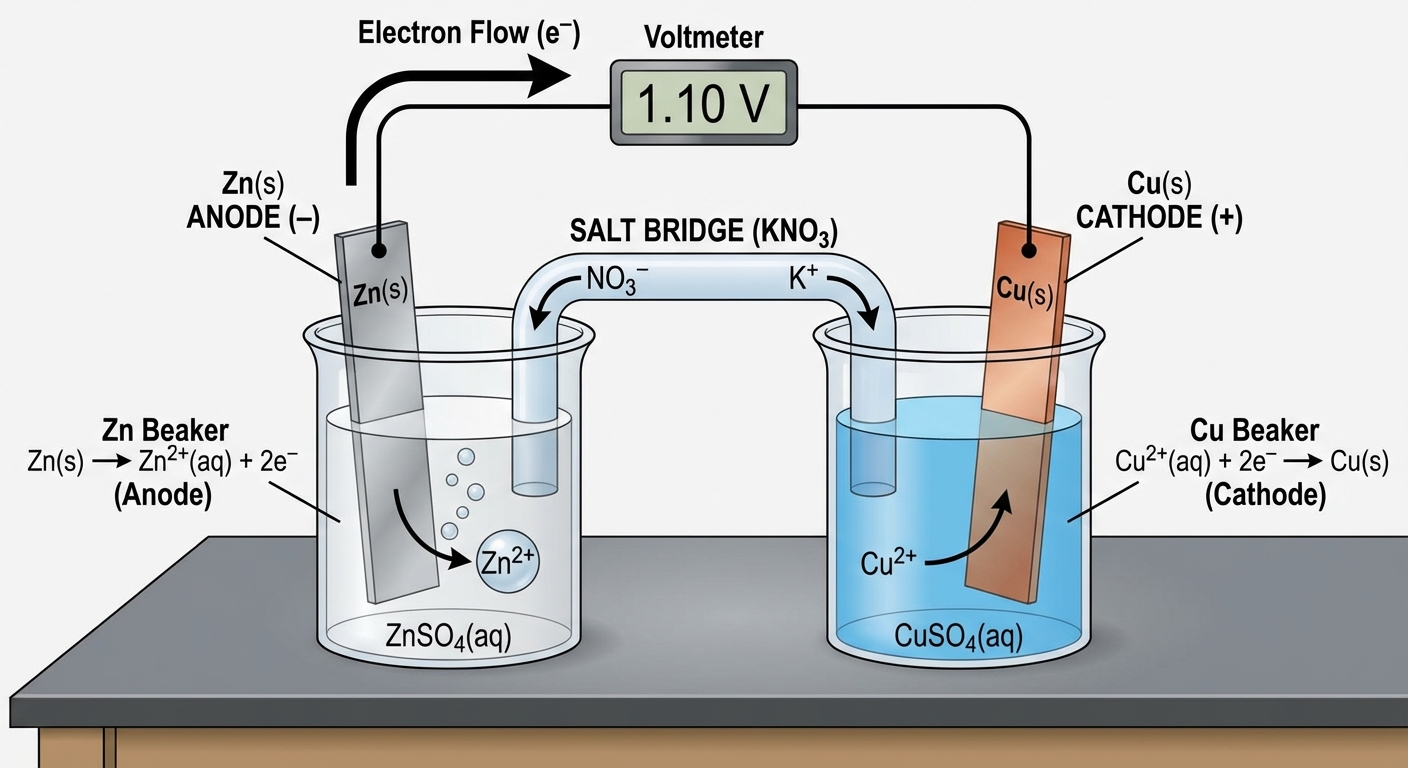
The Galvanic (Voltaic) Cell
A Galvanic cell creates electricity using a thermodynamically favorable (spontaneous) redox reaction ($ΔG < 0$). This is the chemistry behind a standard battery.
Key Components:
- Anode (Oxidation): The electrode where metal atoms lose electrons to become ions. Over time, the anode loses mass.
- Cathode (Reduction): The electrode where ions in solution gain electrons to become solid metal. Over time, the cathode gains mass (plating).
- Salt Bridge: A pathway (often a U-tube containing an electrolyte like $KNO_3$) that allows ions to flow to balance charge. Without this, charge buildup would stop the reaction immediately.
- Anions flow toward the Anode.
- Cations flow toward the Cathode.
- External Wire: Electrons flow through the wire from the Anode to the Cathode.
The Electrolytic Cell
An Electrolytic cell requires an external power source (like a battery or DC power supply) to force a thermodynamically unfavorable (non-spontaneous) reaction to occur ($ΔG > 0$).
Key Differences:
- Energy is consumed, not produced.
- The polarity of the electrodes changes (see table below), though the definition of oxidation/reduction location remains the same.
Comparison Table
| Feature | Galvanic (Voltaic) Cell | Electrolytic Cell |
|---|---|---|
| Thermodynamics | Spontaneous ($ΔG < 0$) | Non-Spontaneous ($ΔG > 0$) |
| Cell Potential ($E_{cell}$) | Positive ($+$) | Negative ($ - $) |
| Anode Sign | Negative ($-$) | Positive ($+$) |
| Cathode Sign | Positive ($+$) | Negative ($-$) |
| Electron Flow | Anode $\to$ Cathode | Anode $\to$ Cathode |
Mnemonics
- OIL RIG: Oxidation Is Loss, Reduction Is Gain.
- AN OX: Anode = Oxidation.
- RED CAT: Reduction = Cathode.
- FAT CAT: Electrons flow From Anode To CAThode.
Cell Potential and Free Energy
Standard cell potential ($E^\circ_{cell}$) defines the voltage capacity of a cell under standard conditions (1 M concentrations, 1 atm pressure, $25^\circ\text{C}$).
Calculating Standard Cell Potential
The AP Chemistry exam provides a table of Standard Reduction Potentials. To find the total cell voltage:
Alternatively, you can reverse the sign of the oxidation half-reaction and add it to the reduction half-reaction:
Critical Rules:
- The half-reaction with the more positive reduction potential will stay as a reduction (Cathode).
- The half-reaction with the more negative (or less positive) potential will be forced to reverse and become oxidation (Anode).
- Intensive Property: If you multiply a half-reaction by a coefficient to balance moles of electrons, DO NOT multiply the voltage ($E^\circ$). Voltage is independent of the amount of matter.
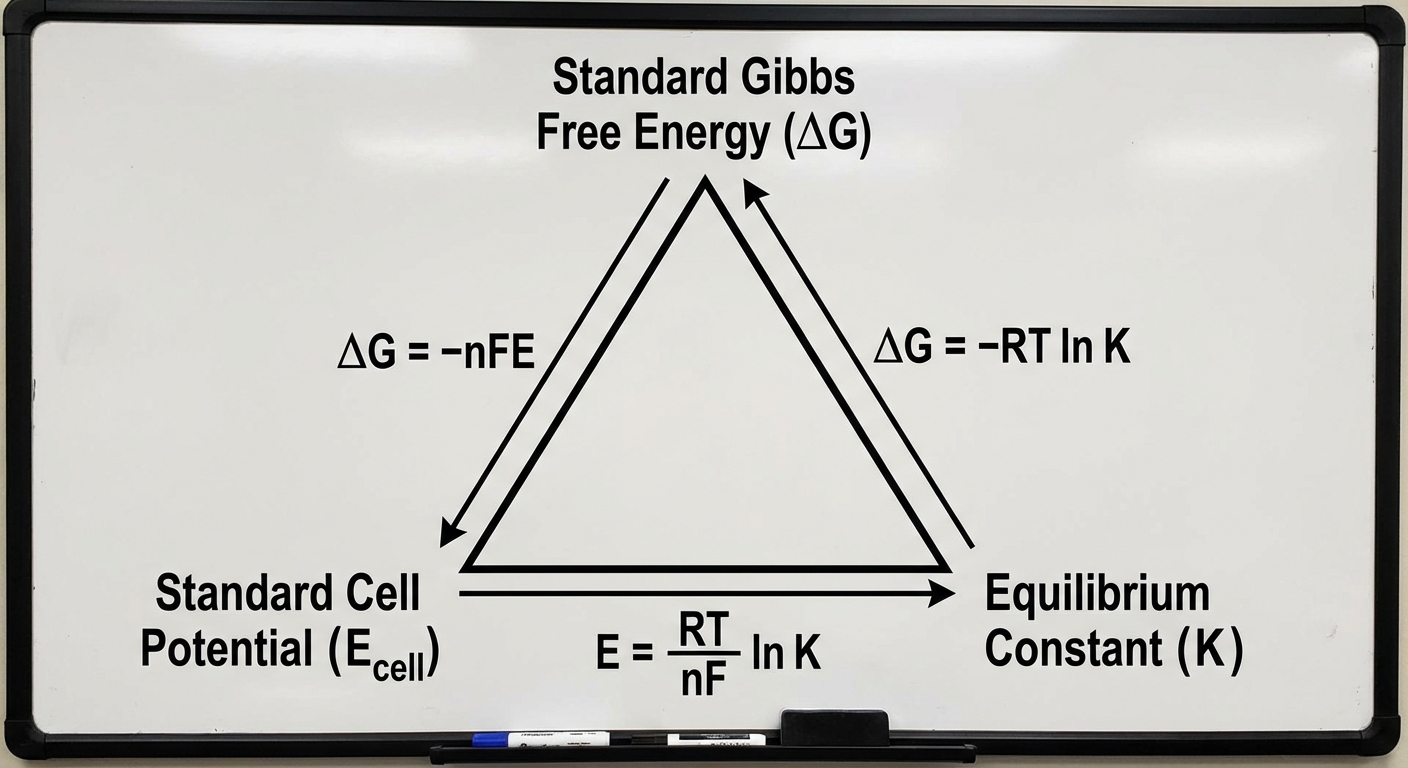
Connection to Free Energy and Equilibrium
Electrochemistry is inextricably linked to thermodynamics. The "Big Triangle" of chemistry connects Gibbs Free Energy ($ΔG^\circ$), the Equilibrium Constant ($K$), and Cell Potential ($E^\circ$).
The relationship between Free Energy and Potential is governed by:
- $n$ = moles of electrons transferred in the balanced redox equation.
- $F$ = Faraday's Constant ($96,485\ \text{C/mol } e^-$).
- $E^\circ$ = Standard cell potential (Volts, or $J/C$).
Interpreting the Signs:
| Condition | $\Delta G^\circ$ | $E^\circ_{cell}$ | $K$ | Conclusion |
|---|---|---|---|---|
| Favored | Negative ($<0$) | Positive ($>0$) | $> 1$ | Product Favored |
| Unfavored | Positive ($>0$) | Negative ($<0$) | $< 1$ | Reactant Favored |
| Equilibrium | $0$ | $0$ | $1$ | Dead Battery |
Cell Potential Under Nonstandard Conditions
Standard conditions correspond to solutions at $1.0\ M$ and gases at $1\ atm$. However, as a battery runs, reactants are consumed and products are made, changing the concentrations. This changes the voltage.
Qualitative Analysis (Le Chatelier's Principle)
To determine if the actual voltage ($E{cell}$) is higher or lower than standard ($E^\circ{cell}$), look at the reaction quotient, $Q$.
- If $Q < 1$: Highly concentrated reactants, low concentration products. The system drives "Right" vigorously. $E{cell} > E^\circ{cell}$.
- If $Q > 1$: Low reactants, high products. The driving force decreases. $E{cell} < E^\circ{cell}$.
- If $Q = K$: The system is at equilibrium. $E_{cell} = 0$.
The Nernst Equation
This equation quantifies the relationship between concentration and cell potential. At $298K$:
Often simplified for calculations at $25^\circ\text{C}$ to:
Concentration Cells
A Concentration Cell uses the same metal and solution for both anode and cathode (e.g., $Ni|Ni^{2+}$), but at different concentrations.
- $E^\circ_{cell} = 0\text{V}$ (since anode and cathode materials are identical).
- Current flows only because nature seeks to equalize the concentrations (increase entropy).
- Electrons flow from the Dilute side (Anode) to the Concentrated side (Cathode) until concentrations are equal.
Electrolysis and Faraday's Law
In electrolysis, specific stoichiometry applies to the electrons flowing through the circuit. We treat electrons as a reagent in the reaction.
Electrolysis of Aqueous Solutions
When electrolyzing aqueous solutions, water can compete with the solute to be oxidized or reduced. You must check the reduction potentials:
- Cathode: Compare the reduction potential of the metal cation vs. the reduction of water ($2H2O + 2e^- \to H2 + 2OH^-$). The more positive $E^\circ$ wins.
- Anode: Compare the oxidation of the anion vs. the oxidation of water ($2H2O \to O2 + 4H^+ + 4e^-$). The more positive oxidation potential (or less positive reduction potential) wins.
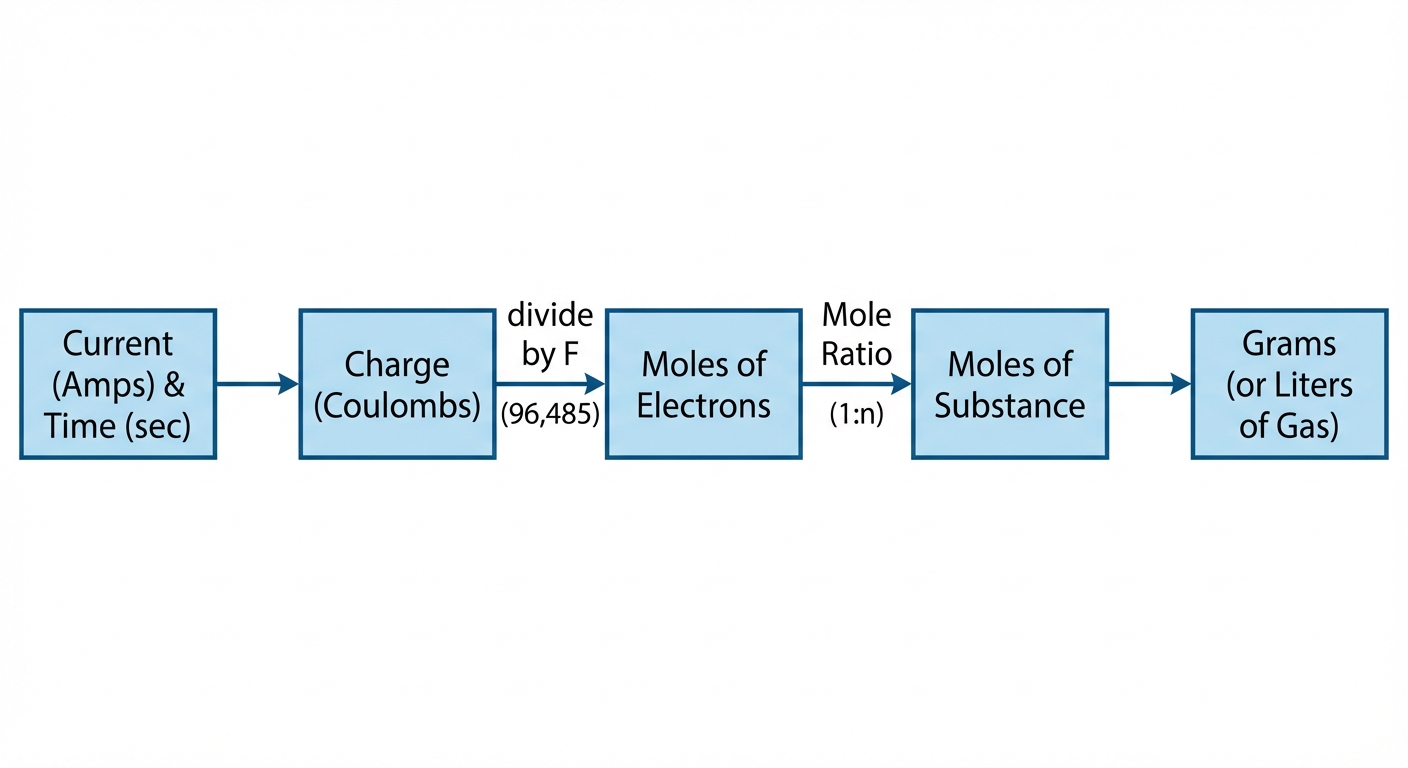
Faraday's Law Calculations (Current Stoichiometry)
Common AP questions involve calculating how much metal is plated (grams) given a current and time.
The Formula:
Where:
- $I$ = Current in Amperes (Amps or $A$).
- $q$ = Charge in Coulombs ($C$).
- $t$ = Time in seconds ($s$).
Dimensional Analysis Strategy:
Do not memorize a derived formula. Follow the "train tracks" of dimensionality:
- Current $\times$ Time $\rightarrow$ Charge ($Amps \times Seconds = Coulombs$)
- Charge $\div$ Faraday $\rightarrow$ Moles of Electrons ($Coulombs \div 96,485 = mol\ e^-$)
- Mole Ratio $\rightarrow$ Moles of Metal ($mol\ e^- \times \frac{1\ mol\ metal}{n\ mol\ e^-}$)
- Molar Mass $\rightarrow$ Grams of Metal
Example Calculation:
How many grams of Copper ($Cu$) are deposited by passing $5.00$ Amps through a $Cu(NO3)2$ solution for $20.0$ minutes?
- Convert time: $20.0 \text{ min} \times 60 = 1200 \text{ s}$.
- Calc Coulombs: $5.00 \text{ A} \times 1200 \text{ s} = 6000 \text{ C}$.
- Calc mol $e^-$: $6000 \text{ C} / 96485 \text{ C/mol} = 0.0622 \text{ mol } e^-$.
- Stoichiometry ($Cu^{2+} + 2e^- \to Cu$): $0.0622 \text{ mol } e^- \times \frac{1 \text{ mol } Cu}{2 \text{ mol } e^-} = 0.0311 \text{ mol } Cu$.
- Mass: $0.0311 \text{ mol } \times 63.55 \text{ g/mol} = 1.98 \text{ g } Cu$.
Common Mistakes and Pitfalls
- Stoichiometry and Voltage: Students often multiply the $E^\circ$ value by the coefficient when balancing the overall redox reaction. Never do this. Voltage is intensive; $2\text{Zn}$ has the same potential difference as $1\text{Zn}$.
- Negative Free Energy units: When using $\Delta G^\circ = -nFE^\circ$, the result is in Joules, not Kilojoules. $\Delta G$ is usually reported in $kJ/mol$, so generally, you must divide your result by 1000.
- Salt Bridge Function: Students forget the mechanism. Remember: Cations to the Cathode, Anions to the Anode. Without this flow to neutralize charge buildup, the cell potential drops to zero instantly.
- Signs in Electrolytic Cells: In Galvanic cells, the Anode is $(-)$. In Electrolytic cells, the Anode is attached to the $(+)$ terminal of the power source. However, Anode is ALWAYS Oxidation regardless of the cell type.
- Standard Conditions: Not realizing that if a question mentions concentrations other than $1.0 M$, you cannot simply add values from the standard table to find the actual voltage; you must qualitatively or quantitatively apply the Nernst relationship.