Unit 1 Study Guide: The Chemistry of Heredity
Nucleic Acid Structure
Nucleic acids are one of the four essential macromolecules of life, functioning primarily to store, transmit, and express hereditary information. While proteins do the work of the cell, nucleic acids provide the instructions.
The Monomer: Nucleotides
Just as beads are strung together to make a necklace, nucleic acids (the polymers) are built from repeating subunits called nucleotides (the monomers).
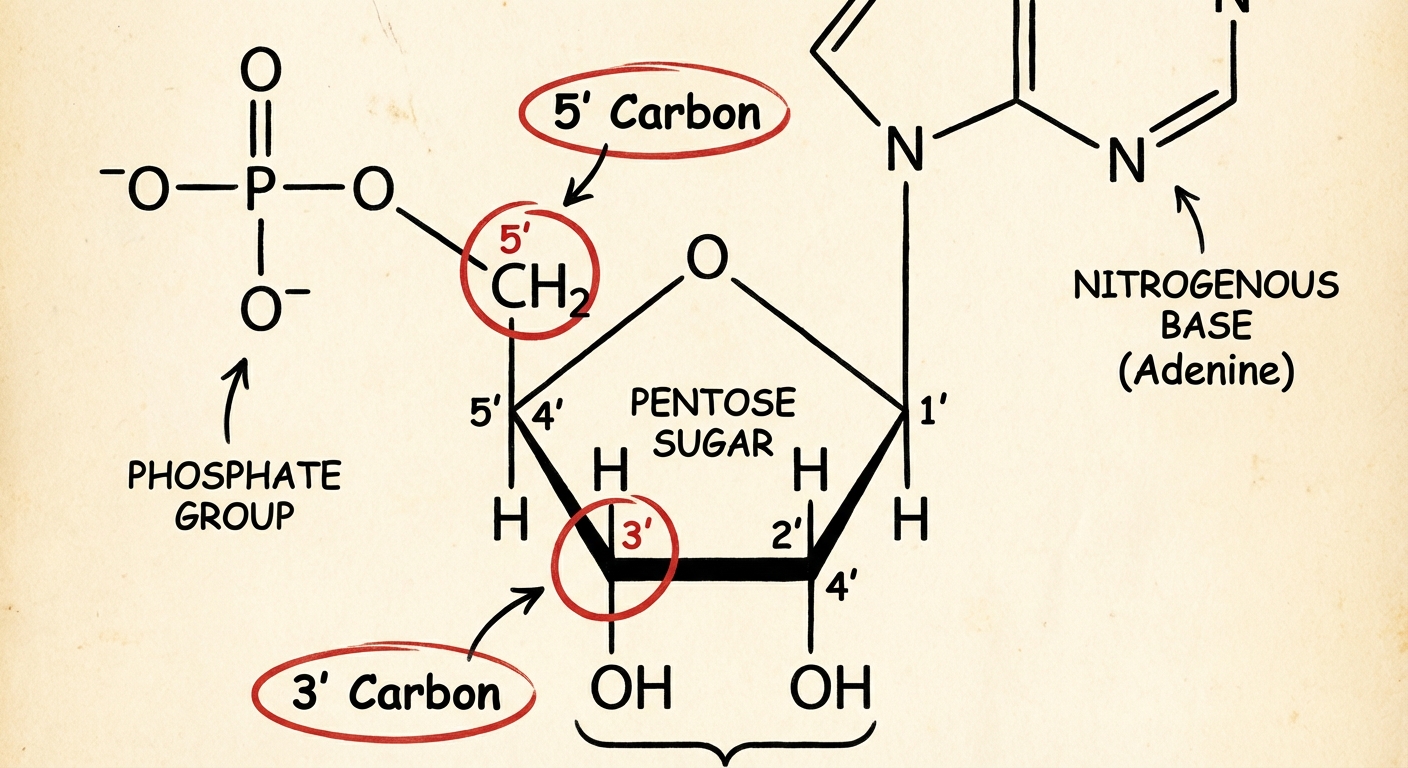
Every nucleotide consists of three specific components covalently bonded together:
- A Five-Carbon (Pentose) Sugar: This forms the central structure.
- A Phosphate Group: Attached to the $5'$ carbon of the sugar. This component is negatively charged, giving DNA its acidic character.
- A Nitrogenous Base: Attached to the $1'$ carbon of the sugar. This is the variable part of the monomer that carries the genetic code.
Nitrogenous Bases
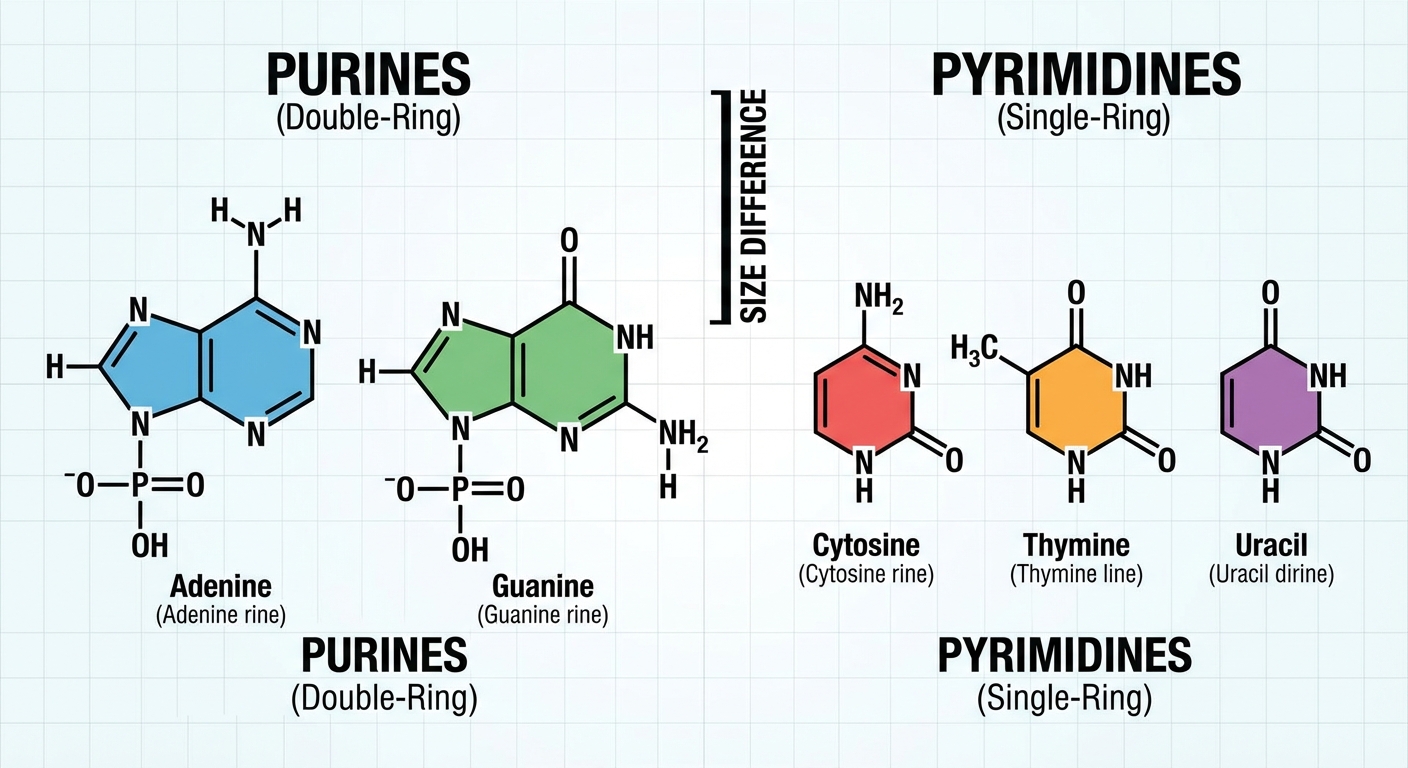
The nitrogenous bases function like letters in an alphabet. There are five specific bases used in biology, categorized into two structural groups:
- Purines: These have a double-ring structure.
- Adenine (A)
- Guanine (G)
- Pyrimidines: These have a single-ring structure.
- Cytosine (C)
- Thymine (T) — found only in DNA
- Uracil (U) — found only in RNA
Mnemonics for Bases
- Purines: "Pure As Gold" (Purines are A and G).
- Pyrimidines: "CUT the Pie (Py)" (Pyrimidines are C, U, and T).
Polymerization and Directionality
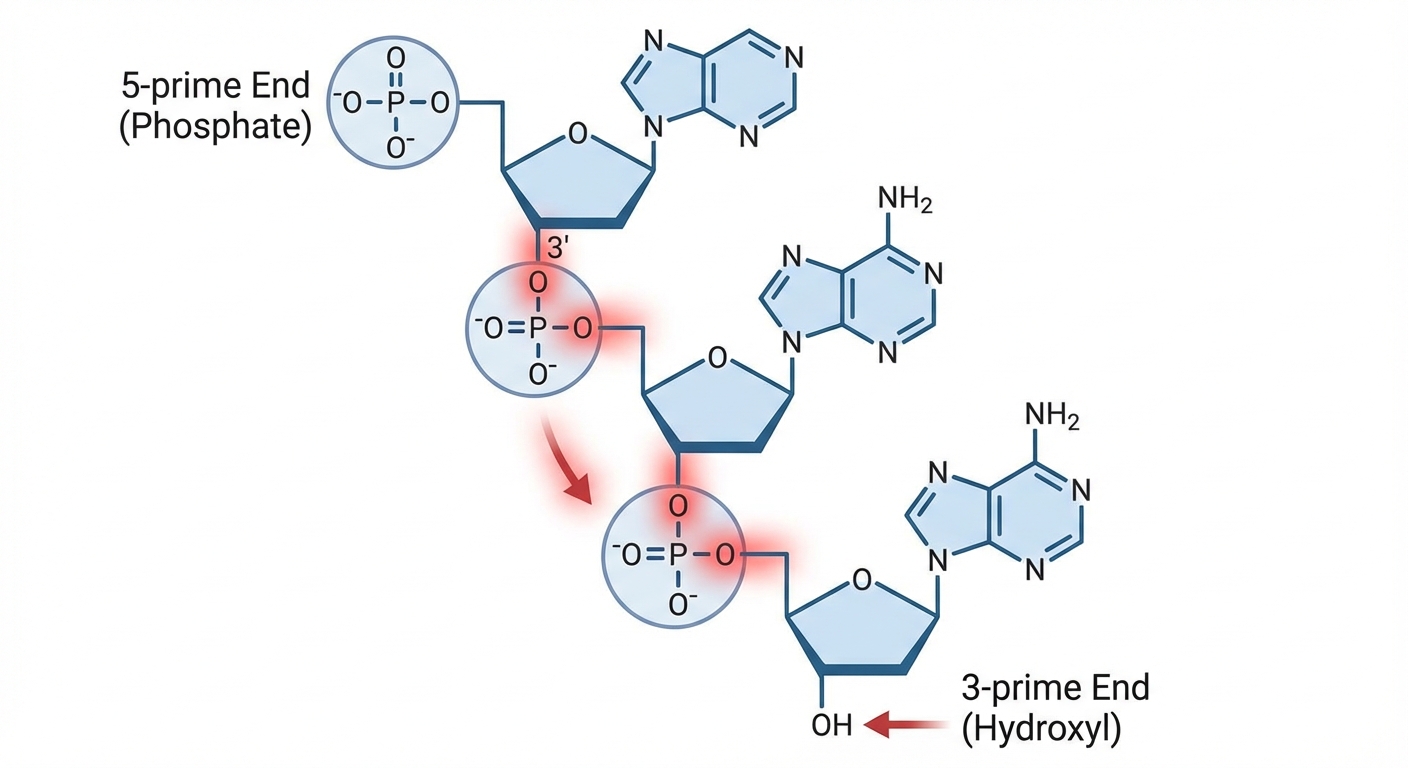
Nucleotides connect via dehydration synthesis to form a polynucleotide chain. The bond formed between the phosphate group of one nucleotide and the sugar of the next is called a phosphodiester bond.
This bonding creates a "Sugar-Phosphate Backbone" that is structurally very strong. The sequence of nitrogenous bases protrudes from this backbone.
Directionality is Crucial:
Biologists define the ends of a nucleic acid strand based on the carbon numbers of the sugar:
- $5'$ End: Terminates with a Phosphate group.
- $3'$ End: Terminates with a Hydroxyl (-OH) group.
DNA and RNA synthesis always occurs in a specific direction (adding new bases to the $3'$ end). You must be able to identify these ends on a diagram.
DNA (Deoxyribonucleic Acid)
DNA acts as the master blueprint for biological organisms. In eukaryotic cells, it is housed in the nucleus.
Structure of DNA
DNA is a double helix, meaning it consists of two strands twisted around each other. Key structural features include:
- Antiparallel Strands: The two strands run in opposite directions. One strand runs $5' \rightarrow 3'$, while the complementary strand runs $3' \rightarrow 5'$.
- Deoxyribose Sugar: The pentose sugar in DNA lacks an oxygen atom at the $2'$ carbon position (hence "deoxy").
- Base Pairing Rules: The strands are held together by hydrogen bonds between bases. A purine always pairs with a pyrimidine:
- Adenine (A) pairs with Thymine (T).
- Guanine (G) pairs with Cytosine (C).
Hydrogen Bonding Strength
Not all base pairs are equal in strength:
- A—T pairs are held by 2 hydrogen bonds.
- G—C pairs are held by 3 hydrogen bonds.
Application: A DNA segment with a high percentage of G-C pairs is more stable and requires higher temperatures to denature (separate) than a segment rich in A-T pairs.
RNA (Ribonucleic Acid)
RNA performs various roles, primarily translating the genetic code from DNA into proteins.
Structure of RNA
While chemically similar to DNA, RNA has three distinct differences:
- Ribose Sugar: The sugar has a hydroxyl (-OH) group at the $2'$ carbon (DNA has only a hydrogen there).
- Uracil instead of Thymine: RNA uses Uracil (U) to pair with Adenine. It does not contain Thymine.
- Single-Stranded: RNA generally exists as a single polynucleotide strand, though it can fold back on itself to form complex 3D shapes (like tRNA).
Comparison Table: DNA vs. RNA
| Feature | DNA | RNA |
|---|---|---|
| Sugar | Deoxyribose | Ribose |
| Strands | Double (Double Helix) | Single |
| Bases | A, C, G, T | A, C, G, U |
| Stability | Highly Stable (Long-term storage) | Less Stable (Short-term transmission) |
| Location | Nucleus (in Eukaryotes) | Nucleus, Cytoplasm, Ribosome |
Common Mistakes & Exam Pitfalls
Confusing the Directionality:
- Mistake: Thinking strands run the same way.
- Correction: DNA strands are antiparallel. If the left strand is $5'$ up and $3'$ down, the right strand is $3'$ up and $5'$ down.
Bond Type Confusion:
- Mistake: Thinking the backbone is held by hydrogen bonds.
- Correction: The backbone (sugar-phosphate) is held by strong covalent (phosphodiester) bonds. The bases are connected across the center by weak hydrogen bonds. This allows enzyme access (unzipping) without breaking the strand itself.
Identifying the Ends:
- Mistake: Looking for the base to determine direction.
- Correction: Look at the Backbone. The Phosphate is the $5'$ end. The empty corner of the sugar (hydroxyl, no phosphate attached) is the $3'$ end.
Uracil vs. Thymine:
- Mistake: Seeing a "T" in a sequence and calling it RNA.
- Correction: If you see a T, it MUST be DNA. If you see a U, it MUST be RNA.