AP Chemistry Unit 3: Interactions of Light and Matter
Spectroscopy and the Electromagnetic Spectrum
Spectroscopy is the study of how matter interacts with electromagnetic radiation. In AP Chemistry, understanding which type of radiation causes which specific change in a molecule is critical for multiple-choice questions. These interactions are quantized, meaning molecules only absorb specific packets of energy.
Types of Radiation and Molecular Motion
The College Board AP exam emphasizes three specific regions of the electromagnetic spectrum. You must memorize the relationship between the region of radiation and the molecular transition it induces.
| Radiation Type | Relative Energy | Effect on Matter | Key Application |
|---|---|---|---|
| Ultraviolet / Visible (UV/Vis) | High | Electronic Transitions (electrons jump to higher energy levels) | Determining concentration (Beer’s Law), analyzing electronic structure |
| Infrared (IR) | Medium | Molecular Vibration (bonds stretch, bend, or wiggle) | Identifying types of bonds and functional groups inside a molecule |
| Microwave | Low | Molecular Rotation (the entire molecule spins) | Determining molecular geometry and bond lengths |
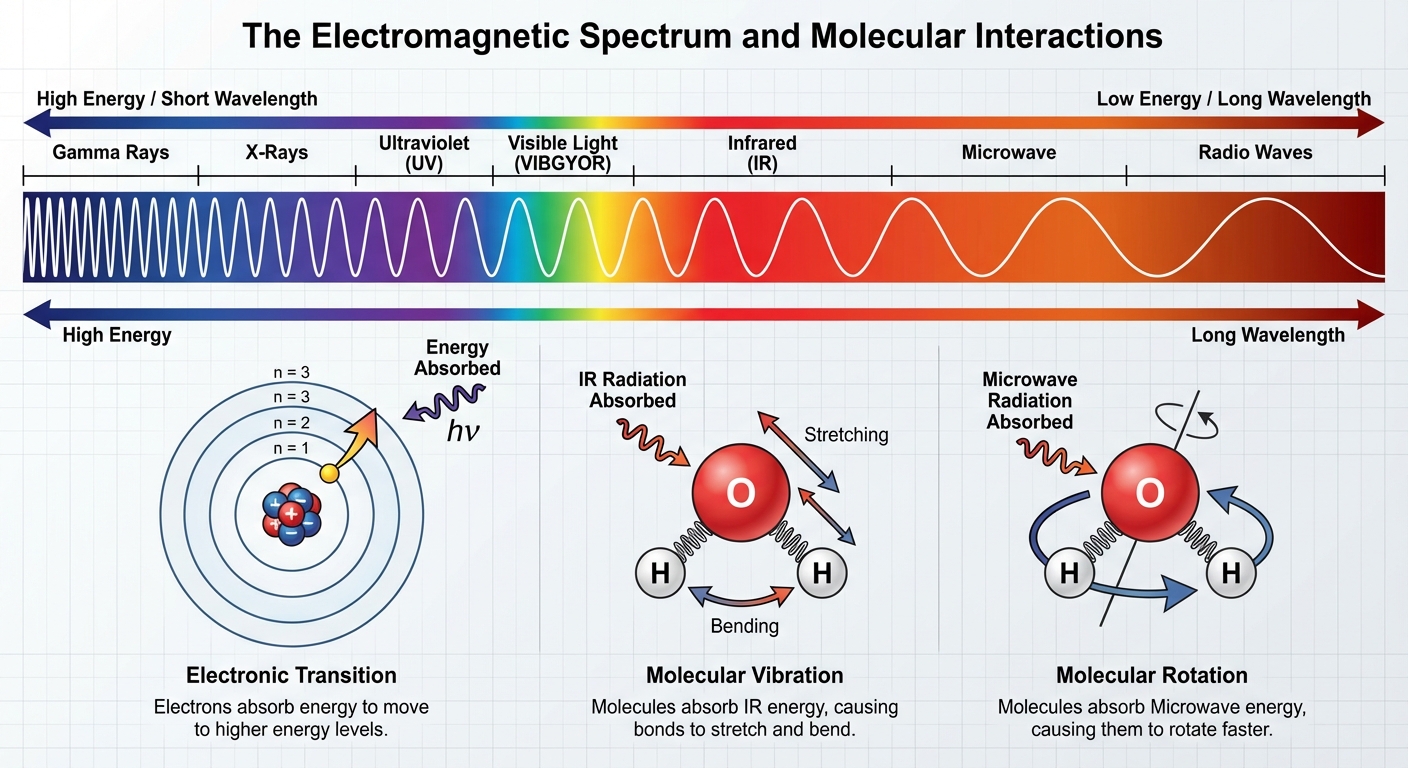
Why This Matters
Different molecules have different energy gaps between their rotational, vibrational, and electronic states. Because these gaps are unique, the wavelengths of light a substance absorbs serve as a "fingerprint" to identify the substance or its concentration.
The Photoelectric Effect & Energy Quantization
To understand spectroscopy, you must treat light as a particle (photon) that carries a specific amount of energy. The Photoelectric Effect demonstrates that light interacts with matter in discrete packets.
Fundamental Formulas
There are two governing equations you will use frequently. These constants are provided on the AP Reference Sheet, but the relationships must be understood.
Relationship between Wavelength and Frequency:
- $c$: Speed of light ($2.998 \times 10^8 \text{ m/s}$)
- $\lambda$ (lambda): Wavelength (usually in meters)
- $\nu$ (nu): Frequency (in Hertz, $\text{Hz}$ or $\text{s}^{-1}$)
Energy of a Photon:
- $E$: Energy of a single photon (Joules, $\text{J}$)
- $h$: Planck's constant ($6.626 \times 10^{-34} \text{ J·s}$)
Key Conceptual Relationships
- Energy and Frequency are distinctively proportional (High $\nu$ = High $E$).
- Energy and Wavelength are inversely proportional (Long $\lambda$ = Low $E$).
- Amplitude (intensity/brightness) relates to the number of photons, not the energy of an individual photon.
Example Problem
A laser emits light with a wavelength of $532 \text{ nm}$. Calculate the energy of one photon.
Solution:
- Convert to meters: $532 \text{ nm} \times \frac{1 \text{ m}}{10^9 \text{ nm}} = 5.32 \times 10^{-7} \text{ m}$
- Apply formula: $E = \frac{hc}{\lambda}$
- Compute:
E = \frac{(6.626 \times 10^{-34} \text{ J·s})(2.998 \times 10^8 \text{ m/s})}{5.32 \times 10^{-7} \text{ m}}
The Beer-Lambert Law
For solutions involving colored compounds, UV/Vis Spectroscopy is used to determine concentration. This relies on the Beer-Lambert Law (often just called Beer's Law).
The Concept
The more concentrated a solution is, the more light it absorbs. The relationship is linear.
The Formula:
- $A$: Absorbance (no units). Note: This is not Transmittance ($T$). If $A=0$, light passes through perfectly. High $A$ means light is blocked.
- $\epsilon$ (epsilon): Molar Absorbtivity ($M^{-1}cm^{-1}$). A constant characteristic of the specific chemical at a specific wavelength.
- $b$: Path Length (cm). The width of the cuvette (sample holder). Usually $1.0 \text{ cm}$.
- $c$: Concentration (Molarity, $M$ or $\text{mol/L}$).
Spectrophotometry Lab Techniques
This is one of the most common lab setups questioned on the AP Exam.
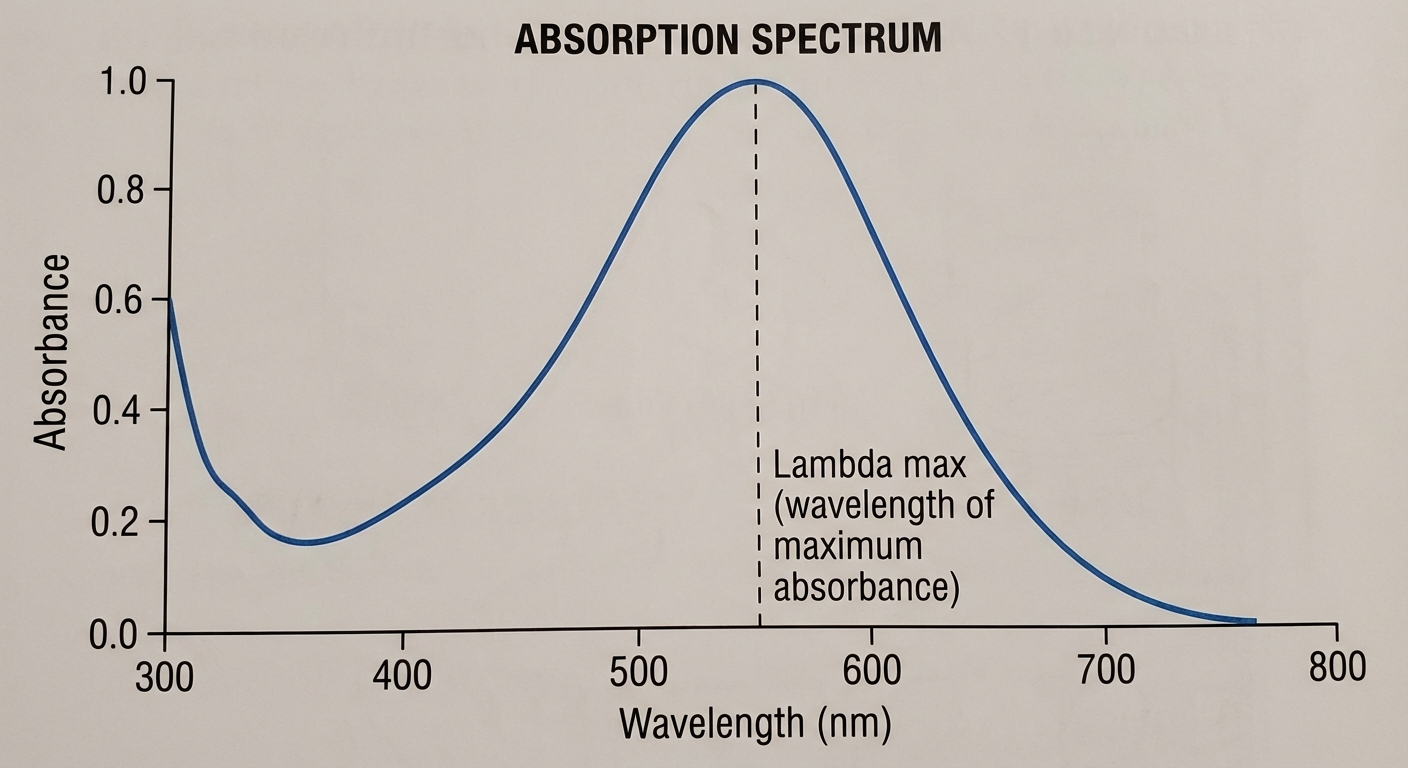
- Selection of Wavelength ($\lambda_{max}$):
You must set the spectrophotometer to the wavelength where the chemical absorbs the most light (the peak of the absorption spectrum). This provides the highest sensitivity.
- Note: If a solution looks Red, it transmits red light and absorbs its complementary color (Green). You would measure absorbance at the green wavelength.
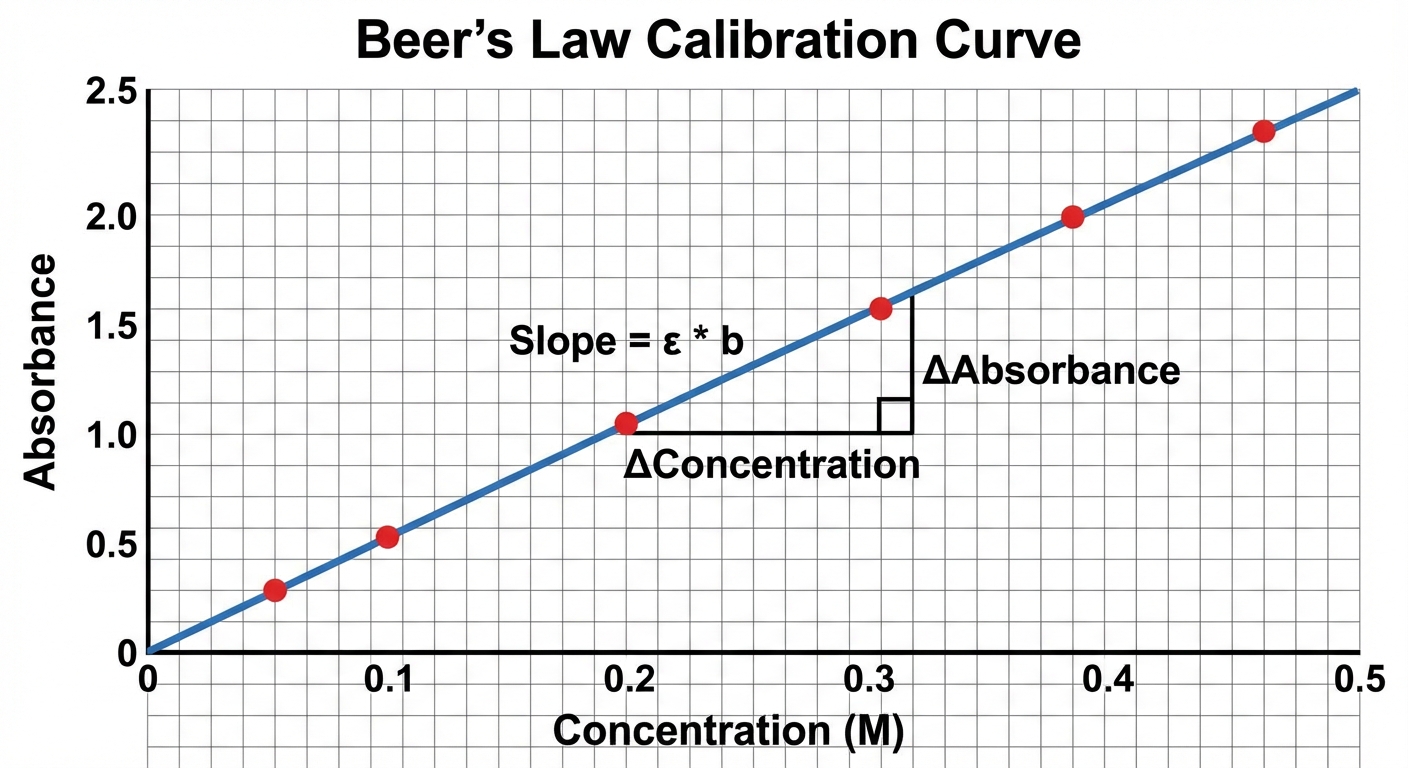
- The Calibration Curve:
To find an unknown concentration, you create a standard curve by plotting Absorbance vs. Concentration for standard solutions of known molarity. The slope of the line of best fit equals $\epsilon b$.
Sources of Error (Common Exam Questions)
- Fingerprints on the Cuvette: Oils from fingers absorb or scatter light. This records a seemingly higher absorbance, leading to a calculated concentration that is too high.
- Rinsing with Water (Dilution Error): If you rinse the cuvette with distilled water but don't dry it or rinse it with the solution to be tested, remaining water dilutes the sample. Concentration decreases $\rightarrow$ Absorbance decreases $\rightarrow$ Result is too low.
- Improper Zeroing (Blanking): You must calibrate the machine with a "blank" (solvent only) to subtract the absorbance of the glass and solvent.
Common Mistakes & Pitfalls
- Confusing Microwave and UV/Vis:
- Mistake: Thinking microwaves break bonds.
- Correction: Microwaves only rotate molecules. UV/Vis moves electrons (and can break bonds at high enough energy).
- Unit Conversion Failures:
- Mistake: Plugging nanometers ($nm$) directly into formulas requiring meters ($m$).
- Correction: Always assume $c$ is in $m/s$, so $\lambda$ must be in meters ($1 \text{ nm} = 10^{-9} \text{ m}$).
- Absorbance vs. Transmittance:
- Mistake: Thinking high Transmittance means high Concentration.
- Correction: They are opposites. $A = -\log(T)$. High concentration = High Absorbance = Low Transmittance.
- The Path Length Variable:
- Mistake: Ignoring variable $b$ when comparing different cuvetttes.
- Correction: If you use a wider cuvette ($2 \text{ cm}$ instead of $1 \text{ cm}$), the light travels through more molecules. Absorbance doubles, even if concentration stays the same.
Summary of Key Relationships
- Energy & Frequency: Direct ($E \propto \nu$)
- Energy & Wavelength: Inverse ($E \propto 1/\lambda$)
- Absorbance & Concentration: Direct ($A \propto c$) (Beer's Law)
- Microwave: Rotation
- Infrared: Vibration
- UV/Visible: Electronic Transition