AP Chemistry Study Guide: Periodic Trends and Ionic Properties
The Theoretical Foundation: Coulomb’s Law and Zeff
Before memorizing trends, you must understand the underlying physics that dictates atomic behavior. In AP Chemistry, stating "because it's on the right side of the table" is never a sufficient explanation. You must explain in terms of nuclear attraction and electron repulsion.
Coulomb’s Law
This is the governing equation for atomic structure. It describes the force of attraction or repulsion between charged particles.
- $Q1$ and $Q2$: The magnitudes of the charges (e.g., the positive nucleus and the negative electron).
- $r$: The distance between the charged particles.
The Rule: The force of attraction increases as the magnitude of charge increases and decreases significantly as the distance increases.
Effective Nuclear Charge ($Z_{eff}$)
The Effective Nuclear Charge is the net positive charge experienced by a valence electron. It is less than the actual nuclear charge ($Z$) because inner (core) electrons shield the valence electrons from the full pull of the nucleus.
- $Z$: Number of protons (Atomic Number).
- $S$: Shielding constant (approx. number of core electrons).
| Trend Direction | Change in $Z_{eff}$ | Reason |
|---|---|---|
| **Across a Period ($ | ||
| ightarrow$)** | Increases | Protons increase, but core electrons (shielding) remain constant. |
| Down a Group ($ iny \downarrow$) | Constant (roughly) | Protons increase, but shielding electrons increase proportionally. |
Periodic Trends
1. Atomic Radius
Definition: Half the distance between the nuclei of two identical atoms bonded together.
- Trend Across a Period: Decreases.
- Why? Higher $Z_{eff}$ pulls the electron cloud tighter toward the nucleus. There are no new shells added to shield this pull.
- Trend Down a Group: Increases.
- Why? New principal energy levels (shells) are added ($n=1, n=2, n=3…$). This increases the distance ($r$) significantly, overpowering the increase in nuclear charge.
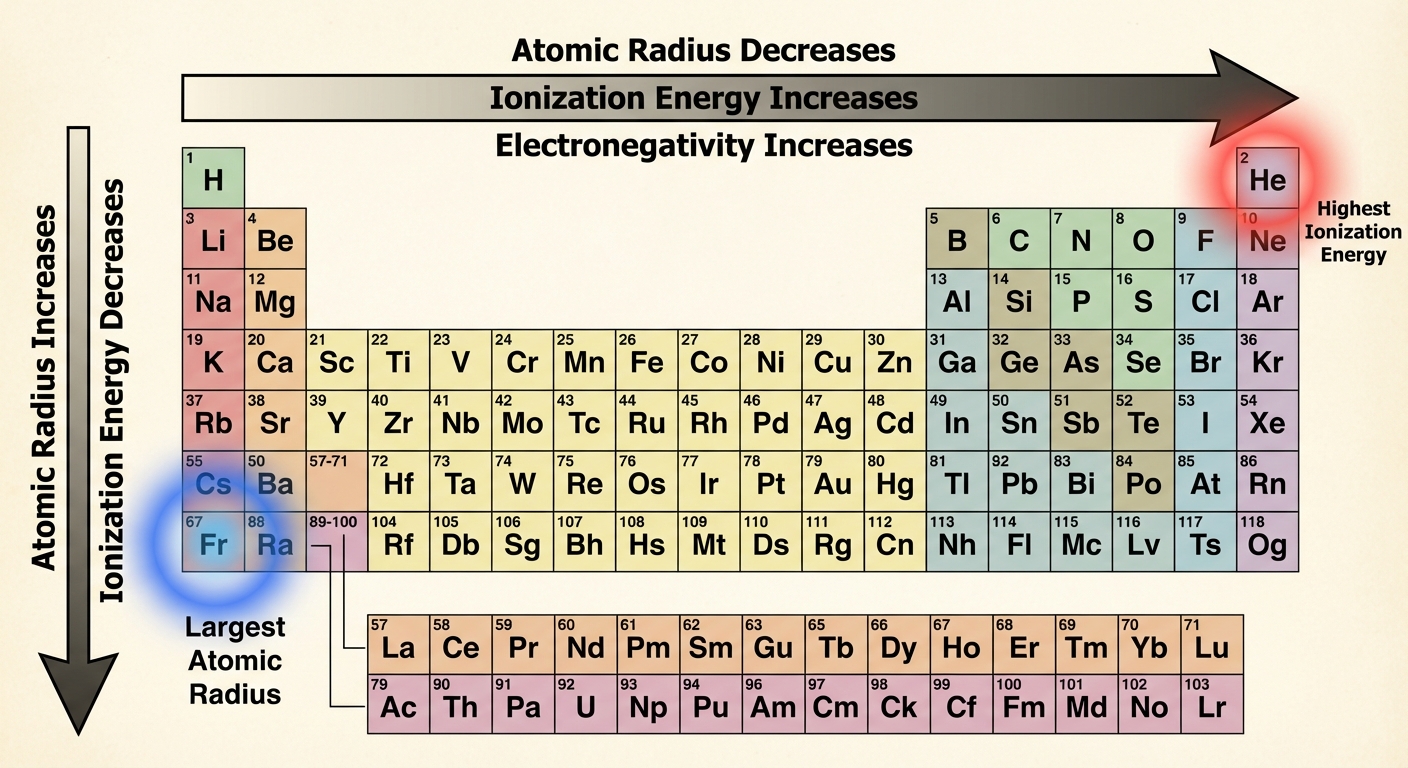
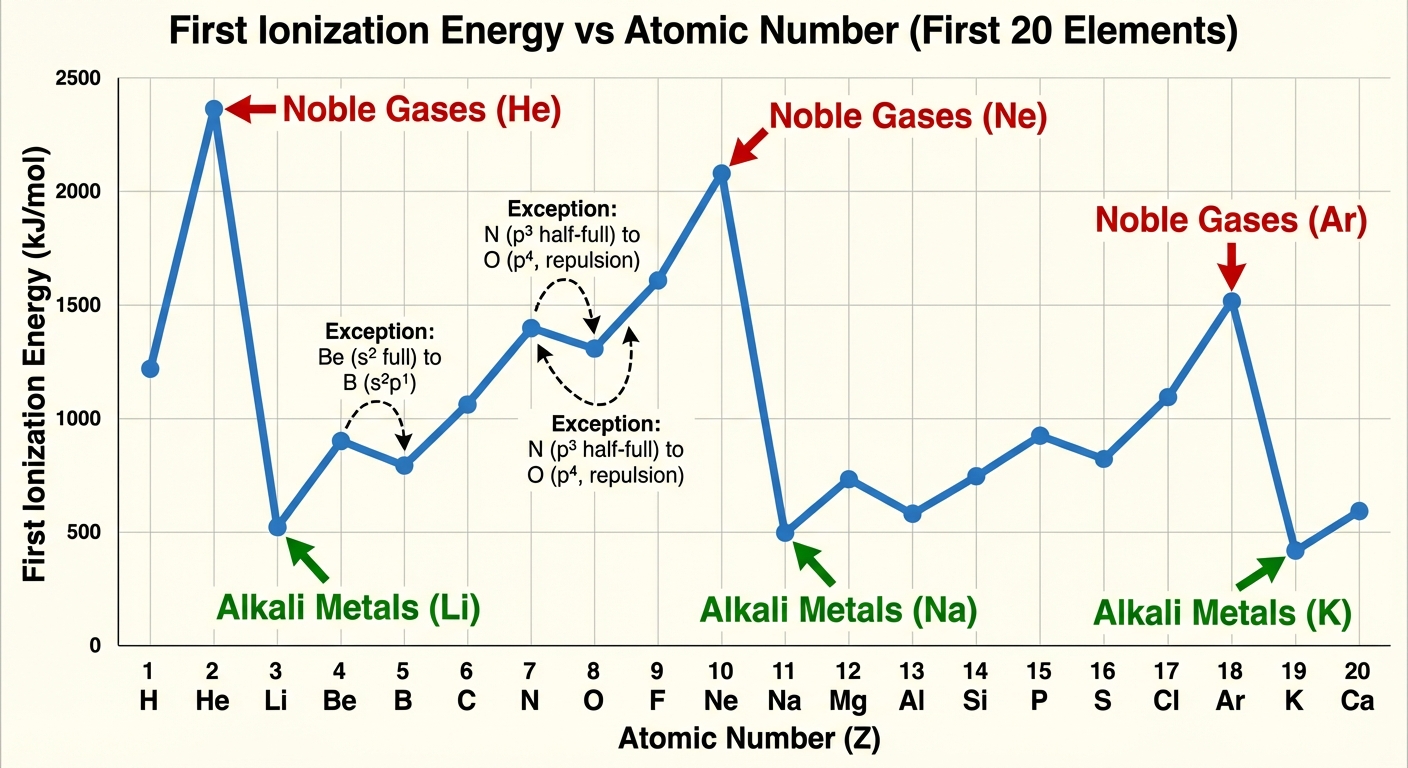
2. Ionization Energy (IE)
Definition: The energy required to remove the outermost electron from a gaseous atom in its ground state.
- Trend Across a Period: Increases.
- Why? Higher $Z_{eff}$ and smaller radius ($r$) mean the nucleus holds valence electrons more tightly. It requires more energy to overcome this Coulombic attraction.
- Trend Down a Group: Decreases.
- Why? Valence electrons are in higher energy levels further from the nucleus. Larger $r$ means weaker attraction according to Coulomb's Law.
Crucial Exceptions to the IE Trend
AP Chemistry frequently tests these two discontinuities:
- Group 2 to Group 13 (e.g., Be vs. B): Boron has a lower IE than Beryllium.
- Reason: Boron's electron is removing from the 2p subshell, which is higher energy (and slightly further away) than Be's 2s subshell. It is easier to remove.
- Group 15 to Group 16 (e.g., N vs. O): Oxygen has a lower IE than Nitrogen.
- Reason: Nitrogen has a half-filled p-subshell ($2p^3$). Oxygen has $2p^4$, meaning the fourth electron forms the first pair in a p-orbital. The electron-electron repulsion within that orbital makes it easier to remove that specific electron.
3. Electronegativity
Definition: The ability of an atom's nucleus to attract shared electrons in a chemical bond.
- Trend: Increases across a period, decreases down a group.
- The King: Fluorine (F) is the most electronegative element (4.0). Noble gases generally do not have electronegativity values because they rarely bond.
4. Electron Affinity
Definition: The energy change that occurs when an electron is added to a gaseous atom.
- Trend: Generally becomes more exothermic (more negative energy value) across a period.
- Nuance: Halogens define this trend (they release massive energy to complete their octet). Noble gases have positive electron affinity (it requires energy to force an electron into a new, higher energy level).
Ion Formation and Ionic Radius
Valence Electrons & Charges
Atoms gain or lose electrons to achieve a stable noble gas configuration (isoelectronic).
| Group | Valence Electrons | Typical Ion Charge | Example |
|---|---|---|---|
| Alkali Metals (1) | 1 | $+1$ | $Na^+$ |
| Alkaline Earth (2) | 2 | $+2$ | $Mg^{2+}$ |
| Halogens (17) | 7 | $-1$ | $Cl^-$ |
| Chalkogens (16) | 6 | $-2$ | $O^{2-}$ |
Ionic Radius Trends
Cations are SMALLER than their parent atoms.
- Why? You lose the outermost valence shell. Furthermore, the remaining electrons experience a higher effective hold because protons > electrons.
- Example: $Ca > Ca^{2+}$
Anions are LARGER than their parent atoms.
- Why? You acquire electrons into the same shell. This increases electron-electron repulsion, forcing the electron cloud to expand outward.
- Example: $F^- > F$
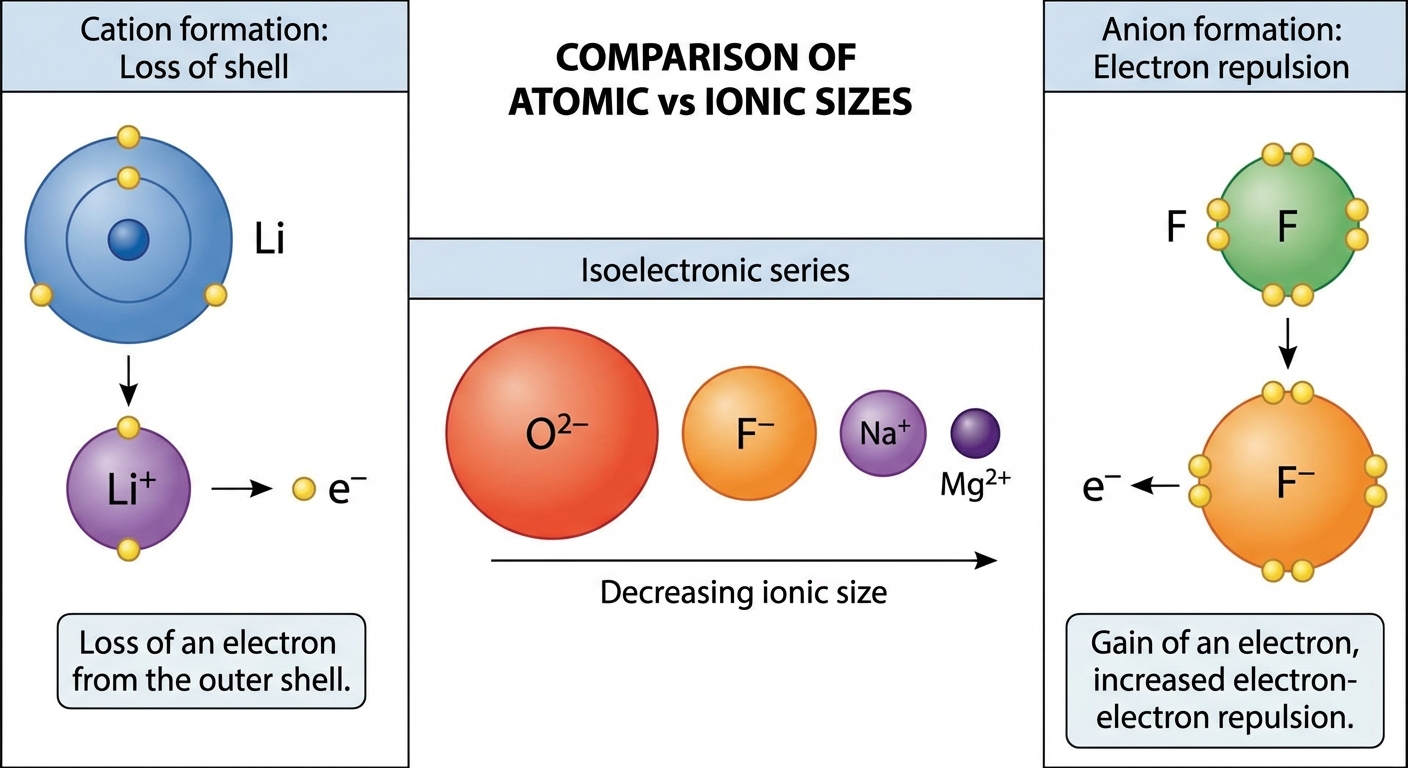
Isoelectronic Series
An Isoelectronic Series is a group of ions/atoms that have the same electron configuration (same number of electrons).
Example: $O^{2-}$, $F^-$, $Ne$, $Na^+$, $Mg^{2+}$ (all have 10 electrons).
The Rule: As the number of protons (atomic number) increases, the radius decreases.
- $Mg^{2+}$ is the smallest (12 protons pulling on 10 electrons).
- $O^{2-}$ is the largest (8 protons pulling on 10 electrons).
Ionic Compounds and Lattice Energy
Definition
Ionic compounds form crystal lattices held together by electrostatic attraction between cations and anions.
Lattice Energy
This is the energy required to completely separate one mole of a solid ionic compound into its gaseous ions. It is a measure of bond strength.
Using Coulomb's Law ($F \propto \frac{Q1 Q2}{r^2}$):
Charge Magnitude ($Q$) is the dominant factor.
- Higher charges = Stronger attraction = Higher Lattice Energy.
- Example: $MgO$ ($+2/-2$) has much higher lattice energy than $NaCl$ ($+1/-1$).
Ionic Radius ($r$) is the secondary factor.
- Smaller ions = Smaller distance ($r$) = Stronger attraction = Higher Lattice Energy.
- Example: $NaCl$ has higher lattice energy than $KBr$ because Na/Cl are smaller than K/Br.
Common Mistakes & Pitfalls
"It wants to be happy/full."
- STOP. Atoms do not have feelings. Never write that an atom "wants" an electron. Instead, write "The system is more stable due to the release of energy" or "High $Z_{eff}$ exerts a strong pull on electrons."
Confusing Electronegativity and Electron Affinity.
- Electronegativity refers to bonded atoms. Electron Affinity refers to a single gaseous atom gaining an electron.
Forgetting Electron Repulsion in Anions.
- When explaining why $Cl^-$ is larger than $Cl$, don't just say "it gained an electron." You must mention that increased electron-electron repulsion causes the cloud to expand.
Misidentifying Trends in Isoelectronic Series.
- Students often think $O^{2-}$ and $Mg^{2+}$ are the same size because they have the same electrons. Remember: Protons determine the pull. More protons = smaller ion.
Successive Ionization Energy.
- When asked to identify an element from a list of ionization energies (e.g., 700, 1400, 7700, 10500), look for the massive jump. The jump signifies removing a core electron. In this example, the jump happens after the 2nd electron, so the element is in Group 2.