AP Chemistry Unit 6: Deep Dive into Enthalpy
Introduction to Enthalpy of Reaction
Enthalpy ($H$) is a thermodynamic quantity equivalent to the total heat content of a system. In AP Chemistry, we are primarily concerned with the change in enthalpy ($\Delta H$) of a chemical reaction, which represents the heat energy exchanged between the system (the chemical reaction) and the surroundings at constant pressure.
Endothermic vs. Exothermic Processes
The sign of $\Delta H$ dictates the direction of heat flow:
- Exothermic Reaction ($\Delta H < 0$): The system releases heat to the surroundings. The products have lower potential energy than the reactants. The surroundings get hotter.
- Example: Combustion of methane, freezing water.
- Endothermic Reaction ($\Delta H > 0$): The system absorbs heat from the surroundings. The products have higher potential energy than the reactants. The surroundings get cooler.
- Example: Decomposition of calcium carbonate, melting ice.
Potential Energy Diagrams
To visualize these energy changes, chemists use potential energy diagrams (or reaction coordinate diagrams). These graphs show the potential energy of the system as reactants transition to products.
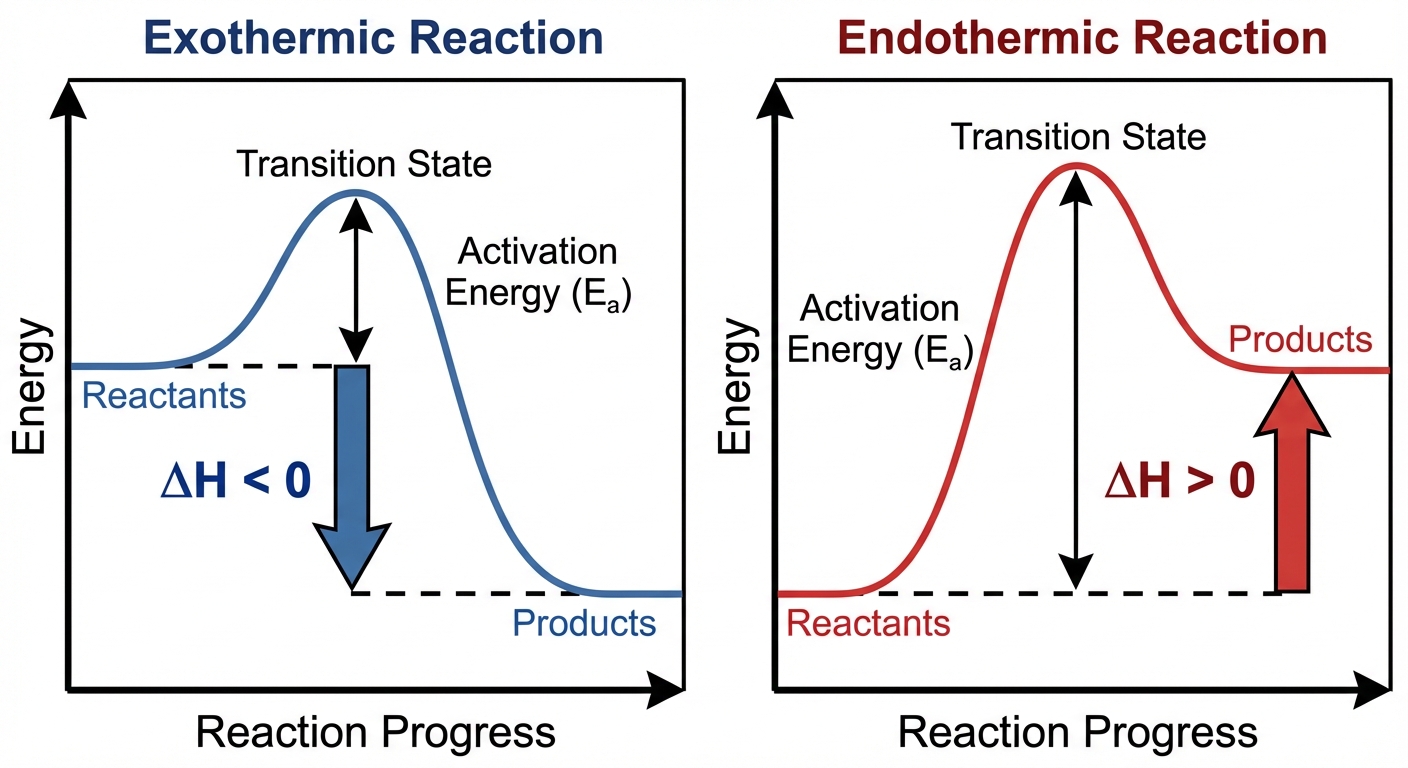
- Activation Energy ($E_a$): The energy barrier that must be overcome for the reaction to occur (from reactants to the transition state).
- $\Delta H$: The vertical difference in energy between Products and Reactants.
Stoichiometry of Enthalpy
Enthalpy change is an extensive property, meaning it depends on the amount of substance reacting. If you double the coefficients of a reaction, you must double the $\Delta H$ value.
Example:
If we only react 1 mole of $H2$, the energy released is halved: $-286 \text{ kJ}$.
Bond Enthalpies
Chemical reactions involve the rearrangement of electrons and atoms. This process can be broken down into two theoretical steps: breaking bonds in reactants and forming new bonds in products.
The Golden Rule of Bond Energy
- Breaking Bonds is Endothermic: It always requires energy input to pull atoms apart. (Energy is absorbed, $+$)
- Forming Bonds is Exothermic: Energy is always released when atoms stabilize by sharing electrons. (Energy is released, $-$)
Mnemonic: "Reactants minus Products" (This is rare! Most thermodynamic formulas are Products minus Reactants. Remember: You break reactants and form products.)
Worked Example
Calculate the approximate $\Delta H$ for the chlorination of methane:
Given Bond Energies (kJ/mol):
- C-H: 413
- Cl-Cl: 242
- C-Cl: 328
- H-Cl: 431
Step 1: Identify Bonds Broken (Reactants)
- 1 $\times$ C-H bond ($413$)
- 1 $\times$ Cl-Cl bond ($242$)
- Total Energy Absorbed = $413 + 242 = 655 \text{ kJ/mol}$
Step 2: Identify Bonds Formed (Products)
- 1 $\times$ C-Cl bond ($328$)
- 1 $\times$ H-Cl bond ($431$)
- Total Energy Released = $328 + 431 = 759 \text{ kJ/mol}$
Step 3: Calculate $\Delta H$
Enthalpy of Formation ($\Delta H_f^\circ$)
The Standard Enthalpy of Formation ($\Delta H_f^\circ$) is the enthalpy change when 1 mole of a compound is formed from its elements in their standard states (25°C, 1 atm).
Key Rules
- The $\Delta Hf^\circ$ of any element in its standard state is ZERO. (e.g., $O2(g)$, $Fe(s)$, $Br2(l)$, $H2(g)$ all have $\Delta H_f^\circ = 0$).
- Pay attention to states of matter (e.g., $H2O(l)$ has a different formation enthalpy than $H2O(g)$).
The Summation Formula
To find the enthalpy of any reaction using formation data, use the standard thermodynamic equation:
Where $n$ is the stoichiometric coefficient from the balanced equation.
Memory Aid: "Products minus Reactants" (The standard way).
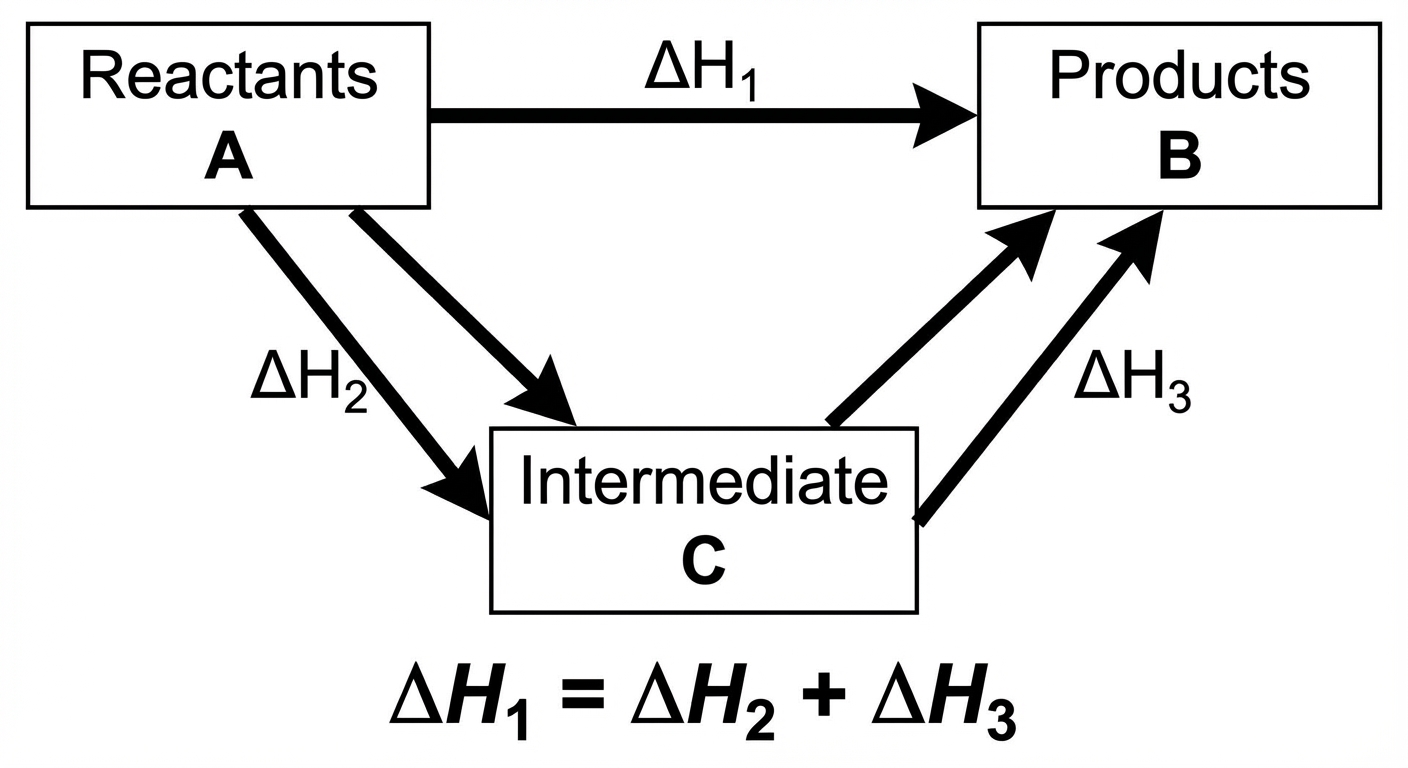
Hess's Law
Enthalpy is a state function, meaning the change in enthalpy is independent of the pathway taken. Whether a reaction occurs in one step or five steps, the total $\Delta H$ is the same.
Rules for Manipulating Equations
When solving Hess's Law puzzles, you manipulate a series of given intermediate reactions to sum up to a target reaction.
- Reversing a Reaction: If you flip the reactants and products, you must flip the sign of $\Delta H$.
- $A \rightarrow B$ ($\Delta H = 50$) $\implies$ $B \rightarrow A$ ($\Delta H = -50$)
- Multiplying Coefficients: If you multiply the reaction coefficients by a factor $n$, you must multiply $\Delta H$ by $n$.
- $A \rightarrow B$ ($\Delta H = 50$) $\implies$ $2A \rightarrow 2B$ ($\Delta H = 100$)
- Adding Reactions: When you sum chemical equations, you sum their $\Delta H$ values.
Worked Example
Target Reaction: $C(s) + 2H2(g) \rightarrow CH4(g)$
Given:
- $C(s) + O2(g) \rightarrow CO2(g) \quad \Delta H = -393.5 \text{ kJ}$
- $H2(g) + \frac{1}{2}O2(g) \rightarrow H_2O(l) \quad \Delta H = -285.8 \text{ kJ}$
- $CH4(g) + 2O2(g) \rightarrow CO2(g) + 2H2O(l) \quad \Delta H = -890.8 \text{ kJ}$
Strategy:
- Keep Eq 1 as is (matches $C(s)$).
- Multiply Eq 2 by 2 (matches $2H_2(g)$). $\Delta H$ becomes $2 \times (-285.8) = -571.6$.
- Reverse Eq 3 (moves $CH_4$ to product side). $\Delta H$ becomes $+890.8$.
Summing:
Common Mistakes & Pitfalls
- Bond Energy Formula Inversion: Students often apply "products minus reactants" to Bond Energies. Don't! Bond energy is "Broken (Reactant) minus Formed (Product)".
- Forgetting Coefficients: When using $\Delta H_f^\circ$, students often look up the value but forget to multiply by the stoichiometric coefficient from the balanced equation.
- Standard States: Assuming all elements have zero formation enthalpy without checking the phase. Iodine is $I2(s)$, not $I2(g)$. Bromine is $Br_2(l)$.
- Sign Errors: Losing track of negative signs when subtracting a negative formation enthalpy (e.g., $10 - (-5)$ becomes $10+5$).
- State of Water: Confusing liquid water and water vapor in combustion reactions. They have different $\Delta H_f^\circ$ values because of the heat of vaporization.