Unit 2: Molecular and Ionic Compound Structure and Properties - Bonding Patterns
Chemical Bonding Fundamentals
This section of AP Chemistry explores how atoms interact to form molecules and ionic lattices. Understanding the electrostatic nature of these bonds and the energy changes associated with their formation is critical for predicting the physical properties of substances.
Types of Chemical Bonds
Chemical bonds are attractive forces that hold atoms together. The primary driver for bond formation is the minimization of potential energy. Atoms bond to achieve a more stable electron configuration, typically resembling a noble gas (the Octet Rule).
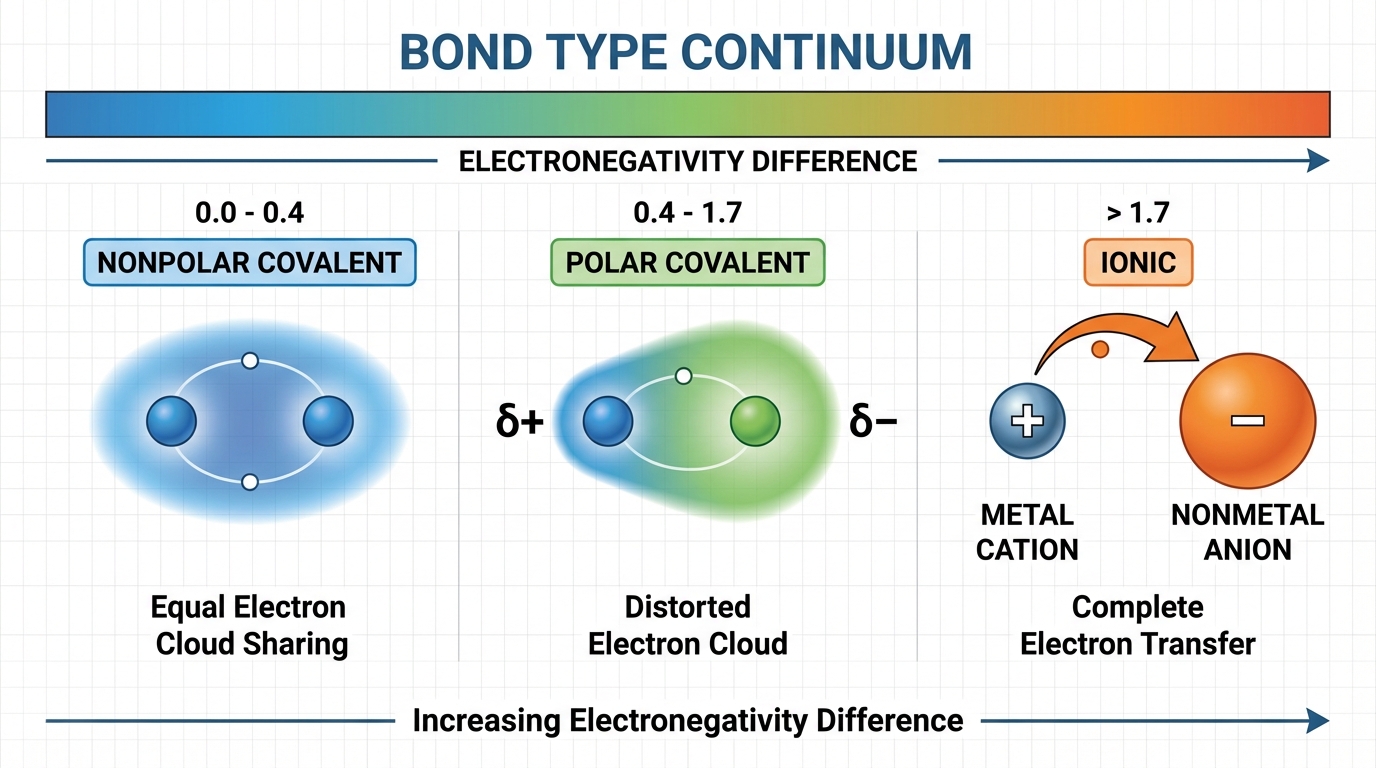
The Bonding Continuum
While we categorize bonds into distinct types, bonding is effectively a spectrum based on the difference in electronegativity ($\Delta EN$) between the interacting atoms.
Ionic Bonding
- Definition: The electrostatic attraction between cations (positive) and anions (negative) resulting from a transfer of electrons.
- Typical Partners: Metal + Nonmetal.
- Nature: High $\Delta EN$ (typically $> 1.7$). The nonmetal pulls the electron completely away from the metal.
Covalent Bonding
- Definition: The sharing of valence electrons between nuclei.
- Typical Partners: Nonmetal + Nonmetal.
- Nonpolar Covalent: Electrons shared equally ($\Delta EN \approx 0$). Example: $Cl2$, $H2$.
- Polar Covalent: Electrons shared unequally due to different electronegativities ($\Delta EN$ between $0.4$ and $1.7$). This creates a dipole (partial positive $\delta+$ and partial negative $\delta-$ ends).
Metallic Bonding
- Definition: Positively charged metal cores (nuclei + core electrons) surrounded by a "sea" of delocalized valence electrons.
- Typical Partners: Metal + Metal.
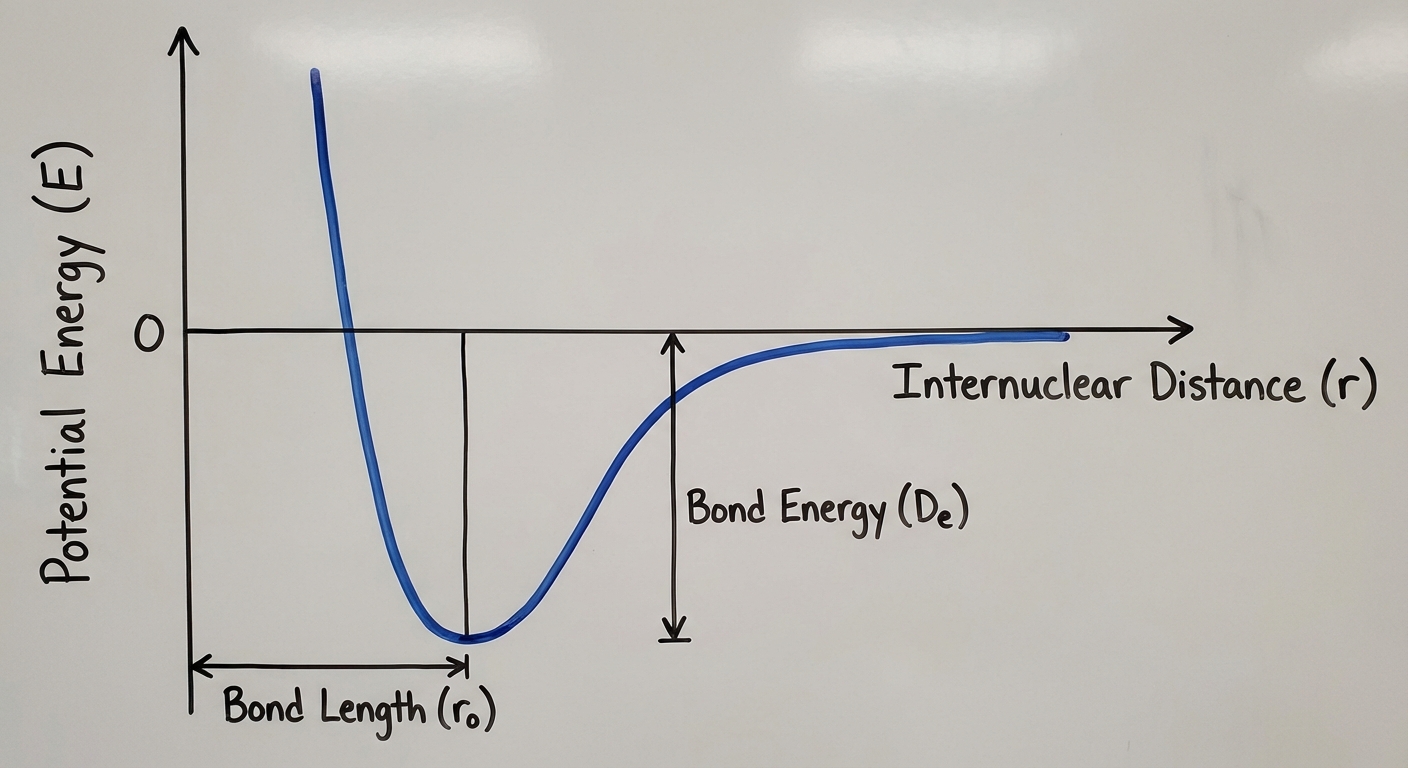
Intramolecular Force and Potential Energy
To understand why covalent bonds form at specific distances, we analyze the Potential Energy vs. Internuclear Distance graph. This is a crucial graph for the AP exam.
Forces at Play
As two atoms approach each other, three electrostatic forces interact:
- Attraction: Nucleus A $\leftrightarrow$ Electron cloud B (and vice versa).
- Repulsion: Nucleus A $\leftrightarrow$ Nucleus B.
- Repulsion: Electron cloud A $\leftrightarrow$ Electron cloud B.
The Potential Energy Curve
The graph plots Potential Energy (y-axis) against the distance between nuclei (x-axis).
- Far Apart (Right side): Potential Energy $\approx 0$. Atoms do not sense each other.
- Approaching: As atoms move closer, attractive forces dominate. Energy decreases (system stabilizes).
- The Minimum (The Well): This is the Bond Length. At this specific distance, attractive and repulsive forces are balanced, and potential energy is at its lowest point.
- Too Close (Left side): If atoms form closer than the bond length, nuclear repulsion dominates. Potential energy rockets upward (destabilizing).
Bond Energy and Length
- Bond Energy: The energy required to break the bond (the depth of the "well" on the graph). A deeper well means a stronger, more stable bond.
- Bond Length: The x-coordinate of the minimum energy point.
- Relationship: Generally, as bond order increases (Single $\rightarrow$ Double $\rightarrow$ Triple), bond length decreases and bond energy increases.
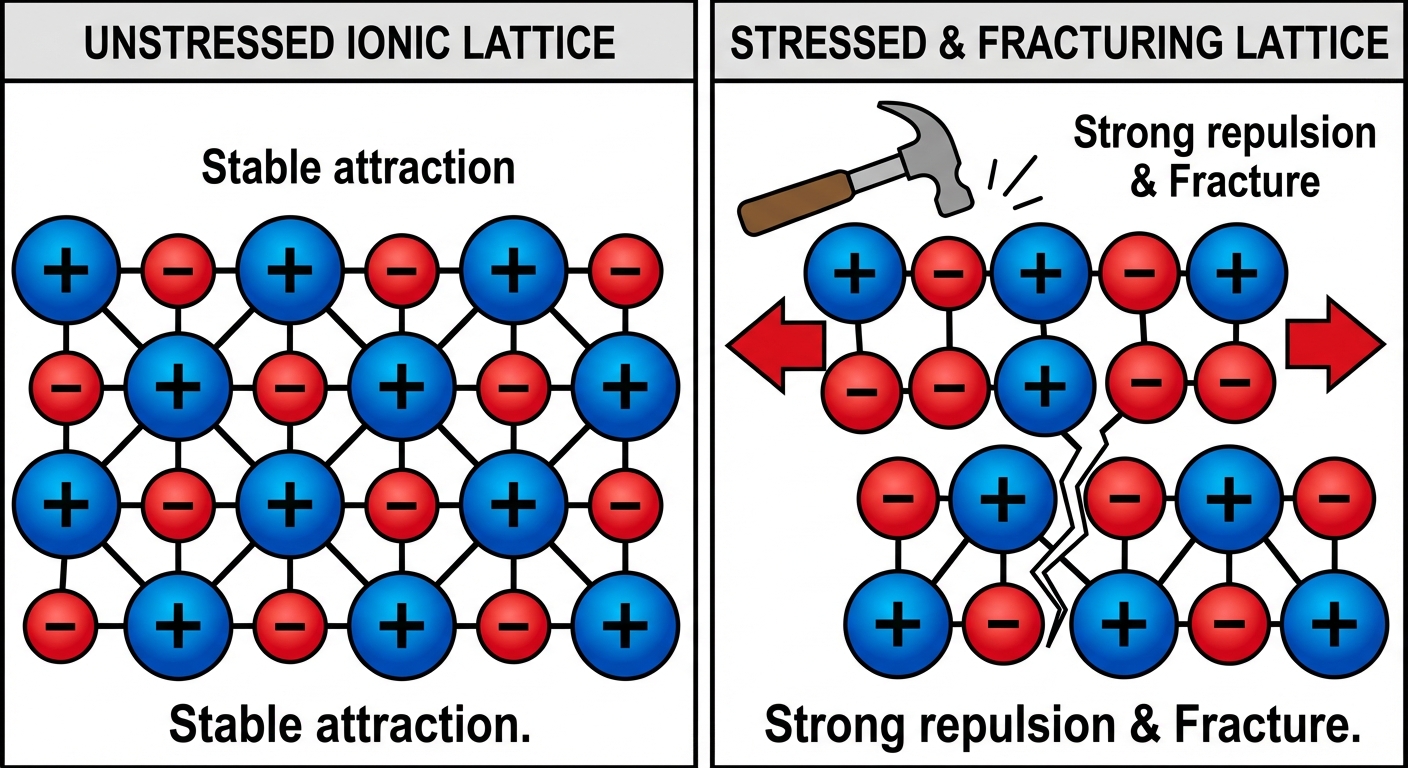
Structure of Ionic Solids
Ionic compounds do not exist as discrete molecules (like $H_2O$); they form huge, continuous 3D arrays called Crystal Lattices.
Energetics: Coulomb's Law
The strength of the interaction between ions in the lattice is governed by Coulomb's Law:
Where:
- $q1, q2$ are the charges of the ions.
- $r$ is the distance between the centers of the ions (sum of ionic radii).
Key Trend:
- Charge Magnitude ($q$) dominates: An ion pair with charges $+2/-2$ (e.g., $MgO$) has a significantly higher lattice energy (and Melting Point) than a pair with $+1/-1$ (e.g., $NaCl$).
- Ionic Radius ($r$): Smaller ions can get closer together. Smaller $r$ leads to stronger attraction. ($LiF$ has a higher lattice energy than $KBr$).
Properties of Ionic Solids
- High Melting/Boiling Points: Breaking the lattice requires overcoming strong electrostatic forces.
- Brittleness: If stress is applied to the lattice, layers shift. Ions of like charge align ($+$ next to $+$), causing massive repulsion that shatters the crystal.
- Conductivity:
- Solid: Does not conduct (ions are locked in place).
- Molten (Liquid) or Aqueous: Does conduct (ions are mobile and free to carry charge).
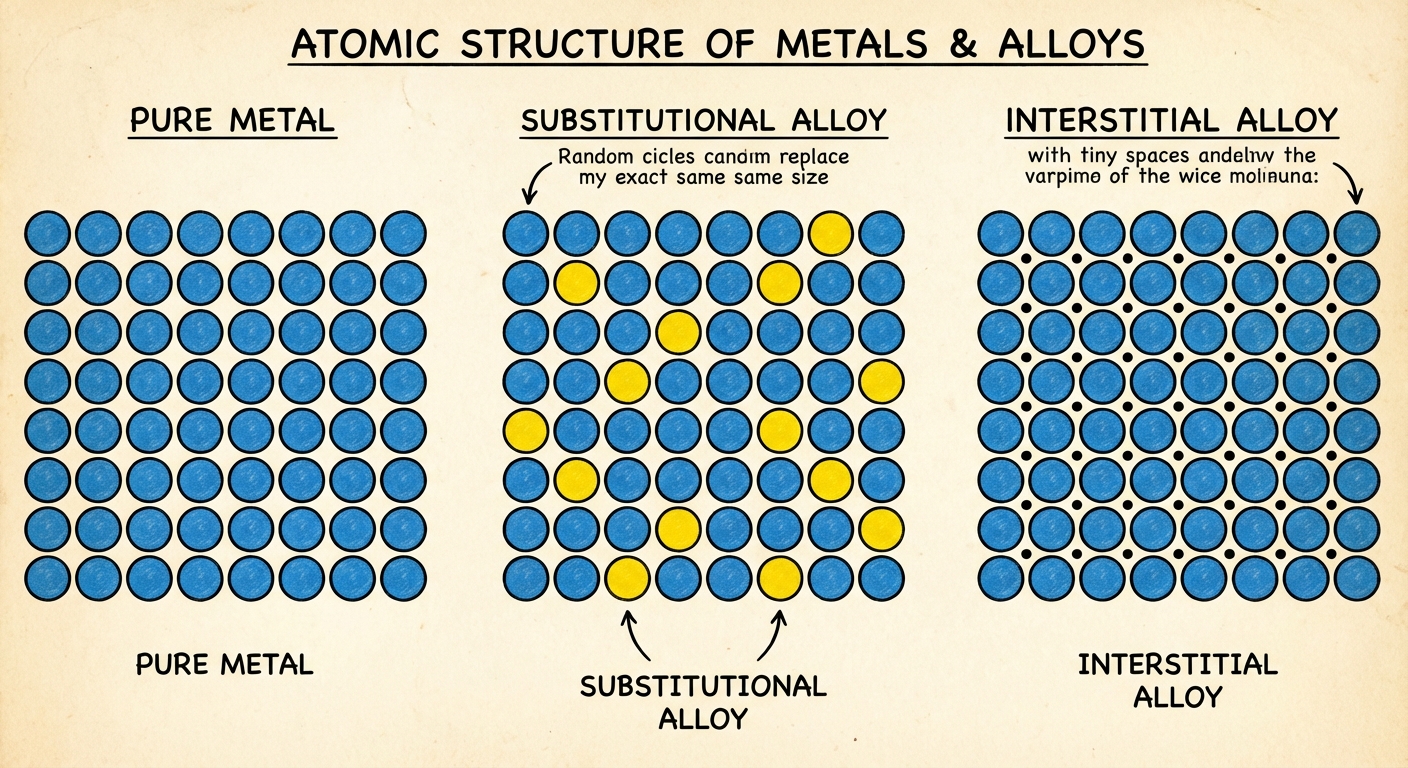
Structure of Metals and Alloys
The Electron Sea Model
Metals are modeled as an array of positive cations immersed in a "sea" of delocalized valence electrons. These electrons are not bound to any specific atom but flow freely throughout the solid.
Properties Explained:
- Conductivity: Delocalized electrons are free to move and carry current.
- Malleability/Ductility: Unlike ionic solids, metals are non-directional. If atoms shift positions, the "sea" adjusts immediately, maintaining the bond without breaking.
Alloys
An alloy is a mixture involving at least one metal. Modifying a pure metal with other elements creates an alloy with altered properties.
1. Substitutional Alloys
- Structure: Atoms of the host metal are replaced by other metal atoms of similar size (radius).
- Example: Brass (Copper and Zinc). Copper and Zinc have similar atomic radii.
2. Interstitial Alloys
- Structure: Small atoms fill the "holes" (interstices) between the larger host metal atoms.
- Example: Steel (Iron and Carbon). Carbon is much smaller than Iron and fits in the gaps.
- Impact: Interstitial atoms tend to make the lattice more rigid and less malleable because the presence of the small atoms prevents the metal layers from sliding over each other easily.
Common Mistakes & Pitfalls
- Ionic vs. Covalent Identification: Students often assume any Bond involves a metal is Ionic. Be careful with high oxidation state transition metals (like $Mn$ in $MnO_4^-$) or metalloids; look at the properties (MP, conductivity) provided in the question, not just the periodic table location.
- Conductivity Confusion: A frequent exam trap asks if solid NaCl conducts electricity. It does not. It must be molten or dissolved. Conversely, metals conduct in both solid and liquid states.
- Lattice Energy Logic: When comparing MP of ionic salts, check Charge ($q$) FIRST, then Radius ($r$). Doubling the charge (x2) has a quadrupling effect on force compared to radius changes.
- Bond Energy vs. Lattice Energy:
- Bond Energy refers to breaking covalent bonds within a molecule.
- Lattice Energy refers to separating ions in an ionic solid.
- Justifying Bond Polarity: Do not simply say "it is polar." You must reference the difference in electronegativity or the unequal sharing of electrons.