Comprehensive Guide to AP Chemistry Unit 7: Equilibrium
Unit 7: Equilibrium
Introduction to Dynamic Equilibrium
The Nature of Reversible Reactions
Most reactions introduced early in chemistry are treated as irreversible (going to completion). However, in equilibrium systems, reactions are reversible. Reactants form products (forward reaction), and products simultaneously react to reform reactants (reverse reaction).
Consider the Haber Process for the synthesis of ammonia:
The double arrow ($\rightleftharpoons$) indicates a reversible process.
Defining Equilibrium
Dynamic Equilibrium is achieved when two conditions are met:
- The rates of the forward and reverse reactions are equal.
- The concentrations (or partial pressures) of observable species differ but remain constant over time.
Important: Equilibrium does not mean the amount of reactants equals the amount of products. It implies that the ratio of their concentrations is constant.
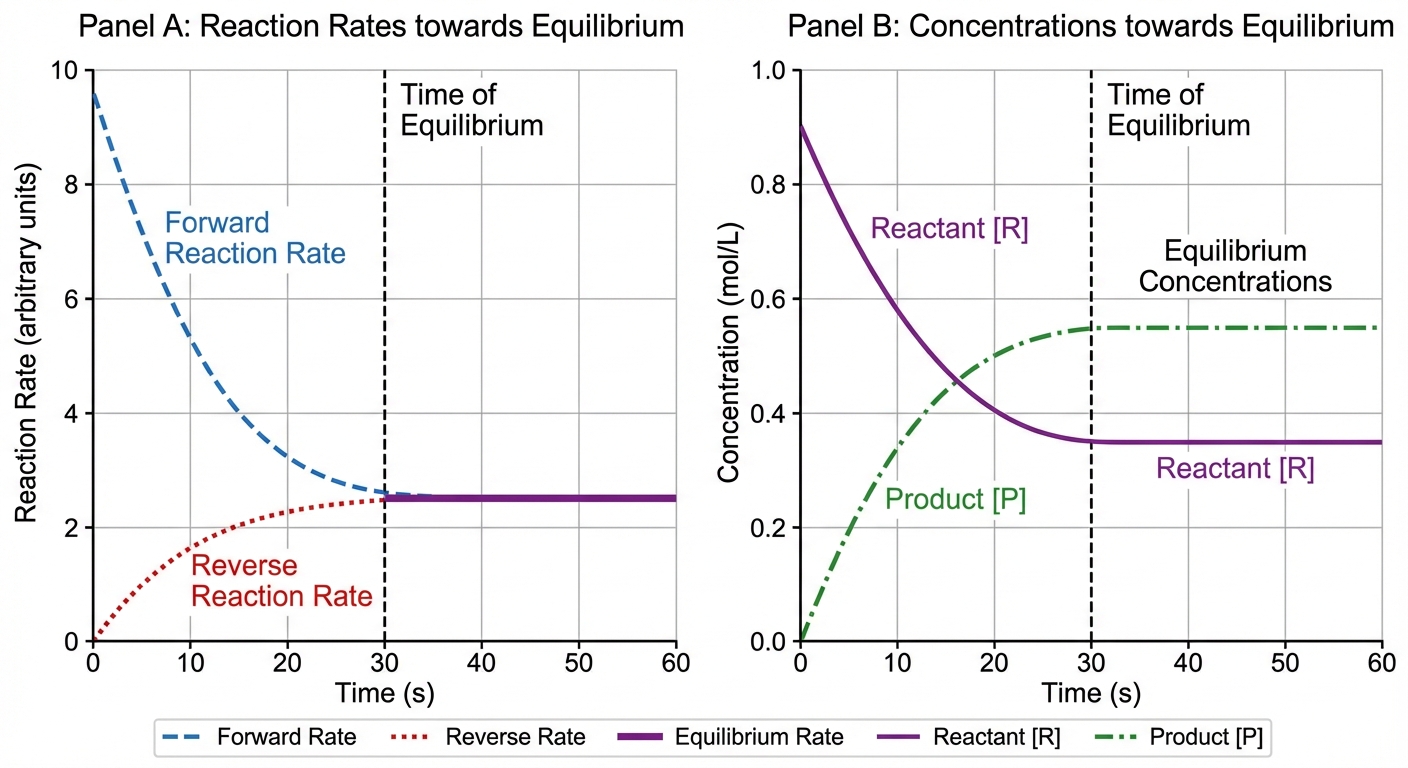
- Rate vs. Time Graph: Forward rate decreases, reverse rate increases, until they meet at a non-zero horizontal line.
- Concentration vs. Time Graph: Slopes flatten out. The lines do not have to meet, but they must become horizontal (slope = 0).
The Equilibrium Constant ($K$) and Reaction Quotient ($Q$)
The Law of Mass Action
The Law of Mass Action describes the quantitative relationship between reactants and products at equilibrium. For a general reaction:
The equilibrium expression is:
Rules for Writing Expressions:
- Pure Solids (s) and Liquids (l) are omitted. Their concentrations (densities) do not change significantly. Only gases (g) and aqueous solutions (aq) are included.
- It is always Products over Reactants.
- Coefficients become exponents.
- $K$ is temperature-dependent.
Types of Calculation Constants
| Constant | Use Case | Formula |
|---|---|---|
| $K_c$ | Molar Concentration | Uses standard brackets $[M]$ |
| $K_p$ | Partial Pressure (gases) | Uses parentheses with P: $\frac{(PC)^c (PD)^d}{(PA)^a (PB)^b}$ |
| $K_{sp}$ | Solubility Product | For insoluble salts dissociating |
| $Ka, Kb$ | Acids and Bases | Acid/Base dissociation (Unit 8) |
| $K_w$ | Water | Autoionization of constant ($1.0 \times 10^{-14}$ at 25°C) |
Converting $Kp$ and $Kc$
For gases, concentration and pressure are related. The conversion formula is:
- $R = 0.08206 \text{ L atm} / \text{mol K}$
- $T = \text{Temperature in Kelvin}$
- $\Delta n = (\text{moles of gaseous product}) - (\text{moles of gaseous reactant})$
Interpreting the Magnitude of $K$
- $K \gg 1$ (e.g., $10^5$): Equilibrium favors products. The reaction goes essentially to completion.
- $K \ll 1$ (e.g., $10^{-5}$): Equilibrium favors reactants. Very little product forms.
- $K \approx 1$: Significant amounts of both reactants and products exist.
The Reaction Quotient ($Q$)
The reaction quotient $Q$ follows the exact same formula as $K$, but it is calculated using concentrations at any specific instant, not necessarily at equilibrium.
Comparing $Q$ to $K$ tells us the direction the reaction must shift to reach equilibrium:
- $Q < K$: The ratio of products is too low. The reaction proceeds FORWARD (right) to form more products.
- $Q > K$: The ratio of products is too high. The reaction proceeds IN REVERSE (left) to form more reactants.
- $Q = K$: The system is at equilibrium.
Manipulating the Equilibrium Constant
Relationships derived from Hess's Law logic apply to $K$, but the mathematical operations differ.
- Reversing a Reaction: Take the reciprocal.
- Multiplying Coefficients by Factor $n$: Raise $K$ to the power of $n$.
- Adding Reactions: Multiply the $K$ values.
Calculating Equilibrium: The RICE Table
To solve problems where you have initial values and need equilibrium values (or $K$), use a RICE table.
- Reaction: Balanced chemical equation.
- Initial: Starting concentrations/pressures.
- Change: Changes in terms of '$x$', utilizing stoichiometric ratios.
- Equilibrium: The expression ($I + C$) to plug into the $K$ equation.
Example Problem:
At a certain temperature, $K_c = 4.0$ for $2A(g) \rightleftharpoons B(g)$. If 1.0 M of A is placed in a flask, what is the equilibrium concentration of B?
| $2A$ | $\rightleftharpoons$ | $B$ | |
|---|---|---|---|
| I | 1.0 | 0 | |
| C | $-2x$ | $+x$ | |
| E | $1.0 - 2x$ | $x$ |
Take the square root of both sides:
(Note: usually we solve for $x$ via approximations or quadratics. If $K$ is very small, we can assume $1.0 - 2x \approx 1.0$ to simplify).
Le Châtelier’s Principle
Le Châtelier’s Principle states that if a stress (change in conditions) is applied to a system at equilibrium, the system will shift to counteract the stress and establish a new equilibrium.
1. Change in Concentration
- Add Reactant: System shifts Right (consume reactant, make product).
- Remove Product: System shifts Right (replenish product).
- Add Product: System shifts Left.
Example: In $N2 + 3H2 \rightleftharpoons 2NH3$, adding $N2$ drives collisions up, increasing the forward rate temporarily until a new equilibrium is reached.
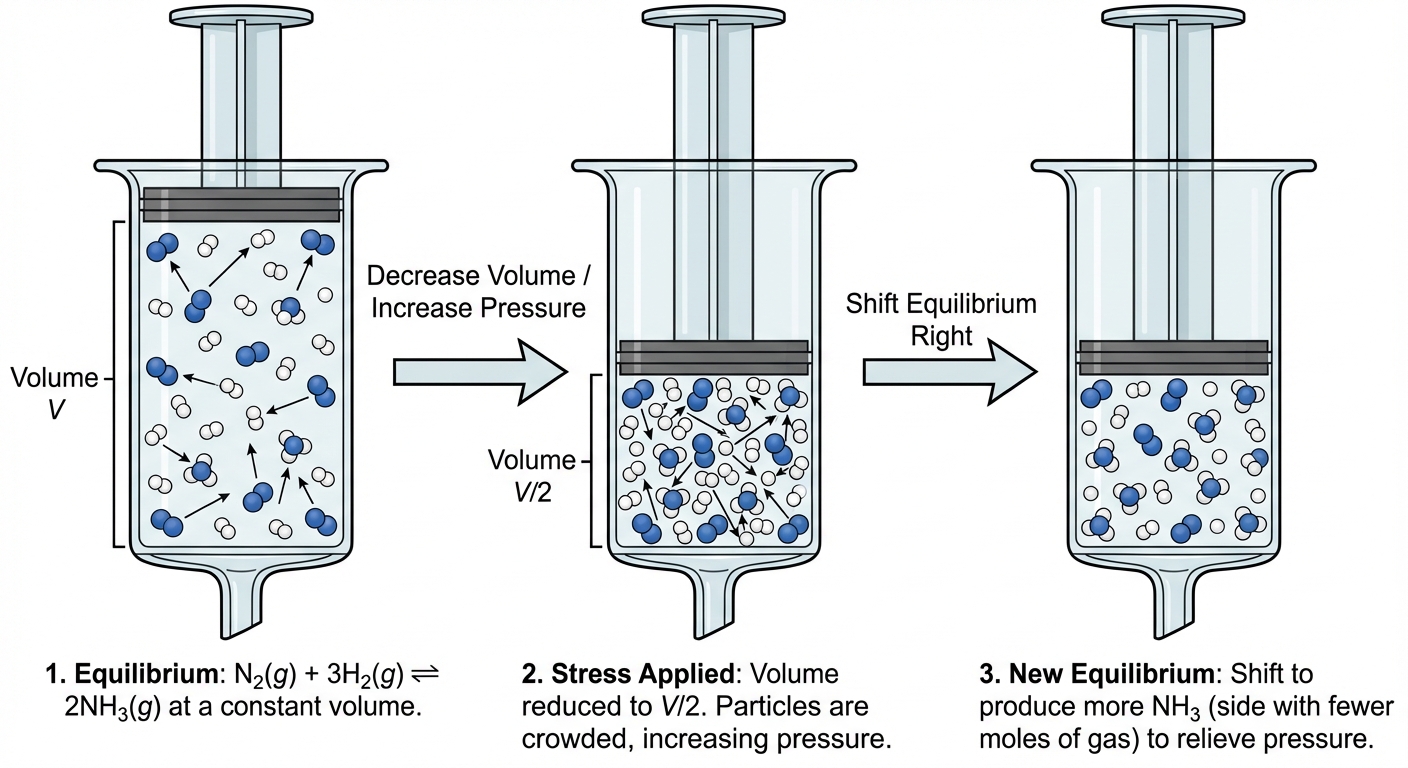
2. Change in Pressure / Volume (Gases Only)
Changes in volume affect partial pressures. This depends on the number of gas moles ($n_{gas}$).
- Increase Pressure (Decrease Volume): Reaction shifts toward the side with FEWER moles of gas to reduce pressure.
- Decrease Pressure (Increase Volume): Reaction shifts toward the side with MORE moles of gas to restore pressure.
Visual Aid: Imagine squeezing a container. The system wants to occupy less space, so it combines molecules.
Special Cases:
- If $\Delta n_{gas} = 0$ (equal moles on both sides), pressure changes cause no shift.
- Adding an Inert Gas (e.g., Helium):
- At Constant Volume: Total pressure rises, but partial pressures of reacting species stay the same. NO SHIFT.
- At Constant Pressure: Volume must increase to accommodate He. Partial pressures of reactants decrease. Shifts to side with more moles.
3. Change in Temperature
Treat heat as a reagent.
- Exothermic (${Delta H < 0$): $Reactants \rightleftharpoons Products + Heat$.
- Increasing T adds "product" $\rightarrow$ Shifts Left ($K$ decreases).
- Endothermic (${Delta H > 0$): $Heat + Reactants \rightleftharpoons Products$.
- Increasing T adds "reactant" $\rightarrow$ Shifts Right ($K$ increases).
Note: Temperature change is the ONLY stress that changes the numerical value of $K$.
4. Dilution (Aqueous Systems)
For $Fe^{3+}(aq) + SCN^-(aq) \rightleftharpoons FeSCN^{2+}(aq)$:
- Adding water lowers the concentration of ALL species.
- Calculate $Q$. Since reactant side usually has more aqueous particles (sum of exponents is higher), the denominator decreases faster than the numerator. $Q$ becomes $> K$.
- Rule of Thumb: Diluting shifts equilibrium toward the side with more aqueous particles.
Solubility Equilibria ($K_{sp}$)
Solubility Product Constant ($K_{sp}$) describes salts that are slightly soluble. Even "insoluble" salts dissociate slightly.
Reaction: $AgCl(s) \rightleftharpoons Ag^+(aq) + Cl^-(aq)$
Expression: $K_{sp} = [Ag^+][Cl^-]$
Molar Solubility ($s$)
- $s$ (Molar Solubility) is the moles of solid that dissolve per liter (M).
- $K_{sp}$ is a constant; $s$ can change based on conditions (pH, common ions).
Converting $s$ to $K{sp}$:
If $PbCl2$ has molar solubility $s$:
Determine ions at equilibrium: $[Pb^{2+}] = s$, $[Cl^-] = 2s$.
The Common Ion Effect
The solubility of a salt is decreased if it is dissolved in a solution that already contains one of its ions.
Example: Dissolving $AgCl$ in $0.1M NaCl$.
- Initial $[Cl^-]$ is 0.1 M (from NaCl).
- This pushes equilibrium $AgCl(s) \rightleftharpoons Ag^+ + Cl^-$ to the Left.
- Result: Less $AgCl$ dissolves compared to pure water.
pH Effects on Solubility
If the anion in the salt is a weak base (e.g., $F^-, OH^-, CO_3^{2-}$), solubility increases in acidic solutions.
- Mechanism: $Mg(OH)_2(s) \rightleftharpoons Mg^{2+} + 2OH^-$. Adding $H^+$ removes $OH^-$ (forming water). Using Le Chatelier's, the system shifts Right to replenish $OH^-$, dissolving more solid.
- If the anion is a neutral conjugate (e.g., $Cl^-, Br^-$), pH has little effect.
Precipitation Conditions ($Q$ vs $K_{sp}$)
When mixing two solutions, calculate $Q_{sp}$ of the potential precipitate.
- $Q > K{sp}$: Solution is supersaturated. Precipitate forms until $Q = K{sp}$.
- $Q < K_{sp}$: Solution is unsaturated. No solid forms.
Common Mistakes & Pitfalls
- Solids/Liquids in $K$: Students often accidentally include water ($l$) or solids ($s$) in the equilibrium expression. Correction: Only use (aq) and (g).
- Inert Gas Shift: Assuming adding Neon shifts the reaction because "pressure went up." Correction: If volume is constant, partial pressures didn't change. No shift.
- Temperature & K: Thinking $K$ is constant when T changes. Correction: T is the only thing that changes the value of $K$.
- Mistaking $s$ for $K{sp}$: Molar solubility ($s$) is a concentration ($M$). $K{sp}$ is the product of concentrations.
- RICE Tables: Forgetting the stoichiometry in the 'C' row. If the reaction is $2A$, the change is $-2x$, not $-x$.
- Q Calculation: Using moles instead of concentration (M) or P (atm) inside the expression. Always convert to Molarity first!