Unit 3: Cellular Energetics — Metabolic Pathways & Survival
Introduction to Cellular Respiration
Life requires a constant input of energy. Cellular Respiration is the set of catabolic pathways that break down organic molecules (like glucose) to produce ATP (Adenosine Triphosphate), the "energy currency" of the cell. While photosynthesis captures energy, respiration releases it.
In the context of AP Biology, you must understand the flow of electrons, the establishment of proton gradients, and how these processes relate to evolutionary fitness.
The Overall Equation
Though respiration involves many steps, the summary equation for the breakdown of glucose is:
This reaction is exergonic (releases energy, $\Delta G < 0$). The process relies heavily on REDOX reactions (Reduction-Oxidation):
- Oxidation: The loss of electrons (Glucose is oxidized to $CO_2$).
- Reduction: The gain of electrons (Oxygen is reduced to $H_2O$).
Mnemonic: OIL RIG (Oxidation Is Loss, Reduction Is Gain).
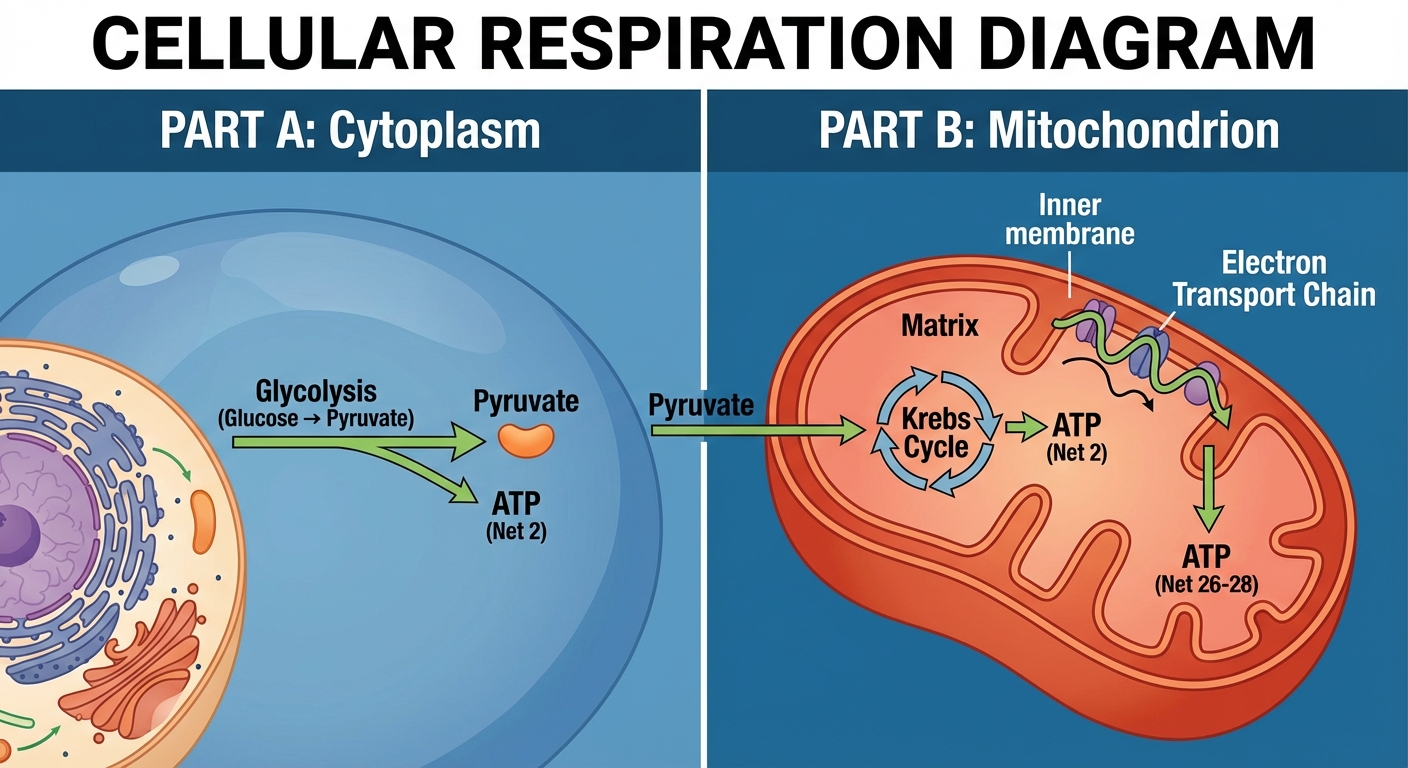
Glycolysis: The Evolutionary Anchor
Glycolysis is the starting point of cellular respiration. It is considered one of the most ancient metabolic pathways because it occurs in the cytosol (cytoplasm) and does not require oxygen.
Key Features
- Location: Cytosol/Cytoplasm.
- Oxygen Requirement: None (Anaerobic).
- Process: A 6-carbon Glucose is split into two 3-carbon molecules of Pyruvate.
The Investment and Payoff
- Energy Investment Phase: The cell uses 2 ATP to phosphorylate glucose compounds, making them unstable and reactive.
- Energy Payoff Phase: Bonds are broken, and energy is harvested. 4 ATP are produced via Substrate-Level Phosphorylation.
Net Yield per Glucose molecule:
- 2 ATP ($4 \text{ produced} - 2 \text{ invested}$)
- 2 NADH (Electrons transferred to the carrier $NAD^+$)
- 2 Pyruvate
Pyruvate Oxidation & The Krebs Cycle
If oxygen is present, pyruvate enters the mitochondrion. This connects the cytosolic breakdown to the mitochondrial engine.
1. Pyruvate Oxidation (The Link Reaction)
Pyruvate is transported from the cytosol into the mitochondrial matrix.
- Each Pyruvate loses a carbon (released as $CO_2$).
- Electrons reduce $NAD^+$ to NADH.
- Coenzyme A attaches to the remaining 2-carbon fragment, forming Acetyl-CoA.
2. The Krebs Cycle (Citric Acid Cycle)
Occuring in the mitochondrial matrix, this cycle completes the breakdown of glucose. Acetyl-CoA combines with Oxaloacetate (4-carbon) to form Citrate (6-carbon), which is then catabolized back to Oxaloacetate.
Per Glucose Molecule (2 turns of the cycle), we yield:
- $4 CO_2$ (Waste product)
- 6 NADH (Electron Carrier)
- 2 FADH$_2$ (Electron Carrier)
- 2 ATP (via Substrate-Level Phosphorylation)
Crucial Note: At this point, most of the energy extracted from glucose is stored in the electron carriers (NADH and FADH$_2$), not in ATP directly.
Oxidative Phosphorylation: The Payoff
This is where the vast majority of ATP is generated. It occurs on the Inner Mitochondrial Membrane (cristae) in Eukaryotes (or the plasma membrane in Prokaryotes).
The Electron Transport Chain (ETC)
The ETC is a series of proteins embedded in the membrane.
- Electron Drop-off: NADH and FADH$_2$ donate electrons to the chain.
- Proton Pumping: As electrons move down the chain (toward higher electronegativity), free energy is released. This energy is used by proteins to pump $H^+$ ions (protons) from the matrix into the intermembrane space.
- The Gradient: This creates a high concentration of $H^+$ in the intermembrane space—an electrochemical gradient known as the Proton Motive Force.
Chemiosmosis & ATP Synthase
The $H^+$ ions cannot diffuse back through the phospholipid bilayer. They must pass through the protein channel ATP Synthase.
- As $H^+$ flows down its gradient (back into the matrix), it spins a rotor in ATP Synthase.
- This mechanical energy catalyzes the addition of a phosphate to ADP: $ADP + P_i \rightarrow ATP$.
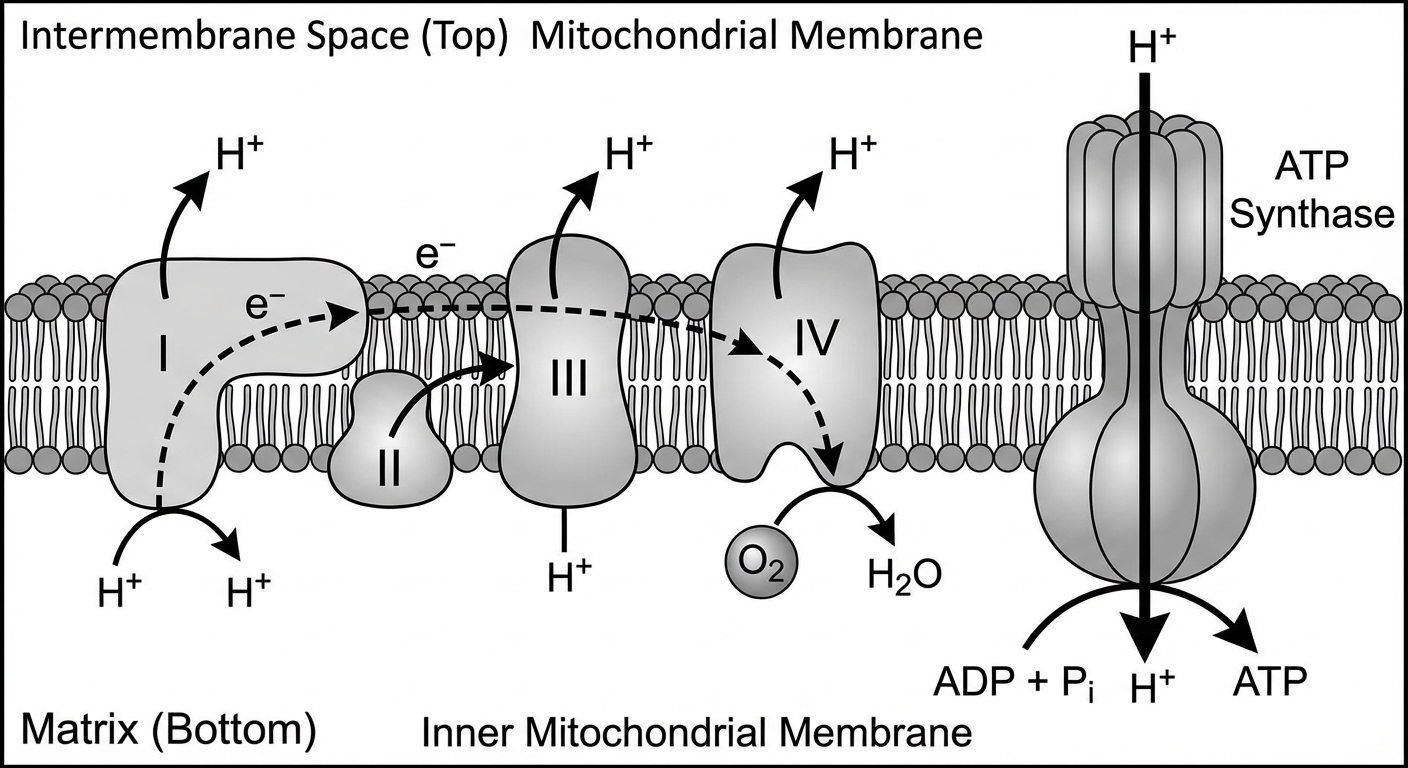
The Role of Oxygen
Oxygen is the final electron acceptor.
- It sits at the end of the chain, having the highest electronegativity.
- It captures the spent electrons and combines with protons ($H^+$) to form water: $\frac{1}{2}O2 + 2e^- + 2H^+ \rightarrow H2O$.
- Without Oxygen: The chain backs up, NADH cannot be oxidized back to $NAD^+$, mechanisms halt, and the cell must switch to fermentation.
Fermentation
When Oxygen is unavailable, the ETC stops. Fermentation allows glycolysis to continue by regenerating $NAD^+$. It occurs in the cytosol.
The Misconception
Fermentation does not produce ATP. It recycles $NAD^+$ so that Glycolysis can continue producing 2 ATP per glucose.
Types of Fermentation
| Type | Product | Example Organisms | Use |
|---|---|---|---|
| Alcohol Fermentation | Ethanol + $CO_2$ | Yeast, Bacteria | Brewing, Baking |
| Lactic Acid Fermentation | Lactate | Muscle cells, Bacteria | Muscle fatigue, Yogurt |
Fitness and Cellular Energetics
In AP Biology Unit 3, "Fitness" refers to how variation in molecular structure and metabolic efficiency impacts an organism's ability to survive and reproduce.
Molecular Variation Increases Fitness
Organisms exhibit different metabolic variations that allow survival in specific environments:
- Hemoglobin variance: Different organisms have hemoglobin with different affinities for oxygen, allowing survival in low-oxygen environments (e.g., high altitudes).
- Chlorophyll types: Different plants possess distinct chlorophyll pigments (a vs. b) that absorb different light wavelengths, increasing fitness in shaded vs. sunny environments.
Decoupling Oxidation from Phosphorylation
Usually, proton movement equates to ATP production. However, in Hibernation or Thermogenesis, these processes can be decoupled.
- Process: Proteins (like thermogenin in brown fat) allow protons to leak back into the matrix without passing through ATP Synthase.
- Result: No ATP is made, but the potential energy is released as HEAT.
- Fitness Advantage: This maintains body temperature in endotherms (mammals/birds) during cold stress, crucial for survival.
Heterotroph Isolation
Evolutionary fitness is also determined by metabolic pathways used by specific bacteria to exploit unique energy sources (e.g., chemotrophs in hydrothermal vents), reducing competition for glucose.
Common Mistakes & Pitfalls
- "Energy is created": Energy is never created; it is transformed. Glucose converts chemical potential energy into ATP and heat.
- Oxygen in the Krebs Cycle: Students often think Oxygen is used in the Krebs Cycle. It is not directly used there; it is only used at the very end of the ETC. However, without Oxygen, the Krebs cycle stops because $NAD^+$ runs out.
- Fermentation produces ATP: No! Glycolysis produces the ATP. Fermentation just clears the "waste" (NADH) so Glycolysis can run again.
- H+ Accumulation Location: Remember, in mitochondria, $H^+$ is pumped out to the intermembrane space (high concentration) and flows in to the matrix. (In chloroplasts, it's the reverse: into the thylakoid space).
- Mitochondria in Plants: Plants do have mitochondria and perform respiration. They are not just photosynthesis machines; they need to burn that sugar for energy when the sun goes down.