Mastering Chemical Equilibrium: Fundamentals and Properties
Introduction to Dynamic Equilibrium
In previous units, we often assumed reactions went to completion—meaning reactants were entirely consumed to form products. In reality, most chemical reactions are reversible. They reach a state where reactants and products coexist indefinitely.
Defining Chemical Equilibrium
Chemical Equilibrium occurs in a closed system when the rate of the forward reaction equals the rate of the reverse reaction.
It is essential to understand that equilibrium is a dynamic process, not a static one. On the macroscopic level (what you see with your eyes), properties like color, pressure, and concentration appear constant. However, on the particulate level (what is happening to atoms and molecules), the forward and reverse reactions are continuously occurring at equal speeds.
Key Conditions for Equilibrium:
- Rates are Equal: Rate${forward}$ = Rate${reverse}$
- Concentrations are Constant: The concentrations of reactants and products stop changing over time. Important: Constant does not mean equal. The concentration of products usually differs from the concentration of reactants.
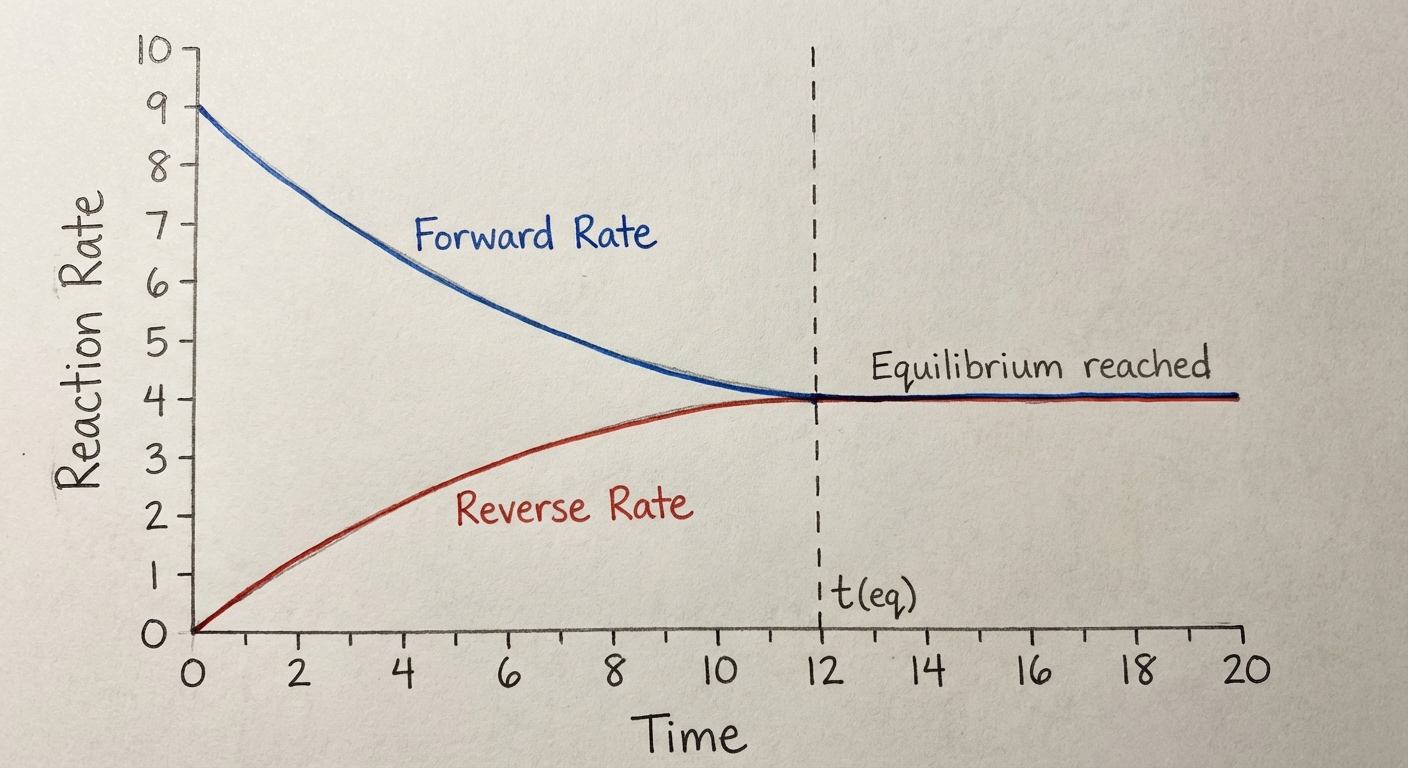
Direction of Reversible Reactions
Reversible reactions are denoted by a double arrow ($\rightleftharpoons$). Consider the generic reaction:
- Forward Reaction ($k_f$): Reactants $A$ and $B$ collide to form $C$ and $D$.
- Reverse Reaction ($k_r$): Products $C$ and $D$ collide to reform $A$ and $B$.
The Kinetics Connection
Initially, only reactants are present. Therefore, the forward rate is high, and the reverse rate is zero. As reactants are consumed, their concentration decreases, causing the forward rate to slow down. Simultaneously, as products accumulate, they collide more frequently, increasing the reverse rate. Eventually, the two rates synchronize.
Reaction Quotient and Equilibrium Constant
To quantify the balance of species at any given moment, we use the Law of Mass Action. This law relates the balanced chemical equation to the amounts of chemical species present.
The Equilibrium Constant ($K$)
The Equilibrium Constant ($K$) describes the ratio of product concentrations to reactant concentrations when the system has reached equilibrium. It is temperature-dependent.
For the reaction $aA + bB \rightleftharpoons cC + dD$, the expression is:
Where:
- $[X]$ denotes the molar concentration (Molarity) of species X.
- Superscripts are the stoichiometric coefficients from the balanced equation.
Rules for Writing $K$ Expressions
- Product over Reactants: Always placed products in the numerator and reactants in the denominator.
- States of Matter:
- Include Gases (g) and Aqueous (aq) species.
- Exclude Solids (s) and Liquids (l). Pure solids and liquids have constant densities and concentrations regardless of the amount present, so their activity is defined as 1 in the expression.
$Kc$ vs. $Kp$
While $Kc$ uses molar concentrations, gas-phase reactions are often measured using partial pressures (atm). This is denoted as $Kp$.
Notation Note: Use brackets $[ ]$ only for molarity. Use parentheses $( )$ or simply $P_{gas}$ for pressure.
To convert between the two:
- $R$ = 0.08206 L·atm/(mol·K) (Ideal Gas Constant)
- $T$ = Temperature in Kelvin
- $\Delta n$ = (moles of gaseous product) $-$ (moles of gaseous reactant)
The Reaction Quotient ($Q$)
The Reaction Quotient ($Q$) uses the exact same formula as $K$, but it is calculated using concentrations (or pressures) at any specific time, not necessarily at equilibrium.
- Formula: $Q = \frac{[C]{initial}^c[D]{initial}^d}{[A]{initial}^a[B]{initial}^b}$
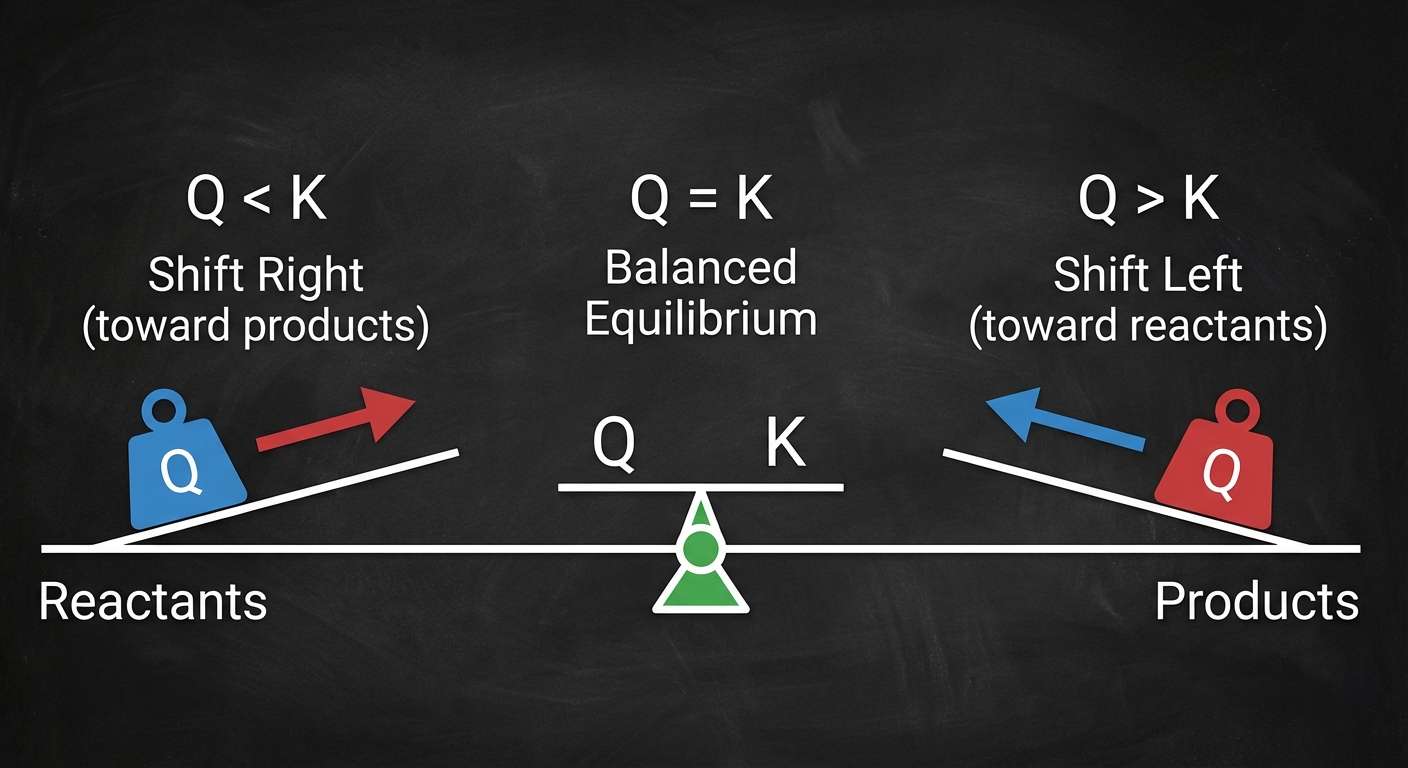
Comparing $Q$ and $K$
By comparing the calculated $Q$ to the known constant $K$, you can predict the direction the reaction must proceed to reach equilibrium.
| Condition | Meaning | Shift Required |
|---|---|---|
| $Q < K$ | Ratio of products to reactants is too small. | Shift Right (Forward): Form more products. |
| $Q = K$ | The system is at equilibrium. | No Shift: Rates are balanced. |
| $Q > K$ | Ratio of products to reactants is too large. | Shift Left (Reverse): Form more reactants. |
Calculating the Equilibrium Constant
There are two primary ways you will be asked to handle $K$ in this section: calculating it from data or manipulating it based on equation changes.
1. Manipulating $K$ (Hess's Law for Equilibrium)
If you modify the chemical equation, the value of $K$ changes in a predictable mathematical way.
| Modification to Equation | Effect on $K$ |
|---|---|
| Reverse the reaction | Take the reciprocal ($1/K$) |
| Multiply coefficients by factor $n$ | Raise $K$ to the power of $n$ ($K^n$) |
| Add two equations together | Multiply their constants ($K1 \times K2$) |
2. Calculating $K$ from Equilibrium Data
If a problem provides the equilibrium concentrations (or pressures) of all species, simply plug them into the equilibrium expression.
Worked Example:
Consider the reaction: $2NO2(g) \rightleftharpoons N2O4(g)$.
At equilibrium, $[NO2] = 0.054 M$ and $[N2O4] = 0.18 M$. Calculate $K_c$.
Step 1: Write the expression.
Step 2: Substitute values.
Step 3: Solve.
Interpreting Magnitude:
- $K \gg 1$: Equilibrium favors products (reaction goes mostly to completion).
- $K \ll 1$: Equilibrium favors reactants (reaction barely proceeds).
- $K \approx 1$: Significant amounts of both reactants and products are present.
Common Mistakes & Pitfalls
- "Equal Concentrations" Fallacy: Students simple assume equilibrium means $[Reactants] = [Products]$. This is rarely true. Always remember: Equilibrium means Rates are equal, Concentrations are constant.
- Solids and Liquids: Do not include solids formulas or liquid water (as a solvent) in your $K$ or $Q$ expressions. This will invalidate your calculation.
- Pressure Notation: If asking for $Kp$, do not use brackets $[ ]$. Use $P{gas}$ notation. AP graders are strict about notation.
- Stoichiometric Powers: A common calculator error is forgetting to square or cube a concentration if the coefficient in the balanced equation is 2 or 3.
- Reversing vs. Negating: When reversing a reaction, you take the inverse ($1/K$), not the negative ($-K$). (Do not confuse this with $\Delta H$ or $\Delta G$).
Memory Aid: "P.O.R."
When writing equilibrium expressions, remember P.O.R.:
Products Over Reactants.