AP Biology Unit 1: Foundations of Biochemistry
The Structure of Water and Hydrogen Bonding
Water is the medium of life. Most cells are surrounded by water, and cells themselves are about 70–95% water. The structure of the water molecule and the interactions between water molecules govern the chemical environment for all life forms.
Molecular Geometry and Polarity
At its core, water ($H_2O$) consists of two hydrogen atoms joined to one oxygen atom by polar covalent bonds. To understand water's behavior, you must understand electronegativity.
- Electronegativity: The measure of an atom's affinity for electrons. Oxygen is highly electronegative, while Hydrogen is less so.
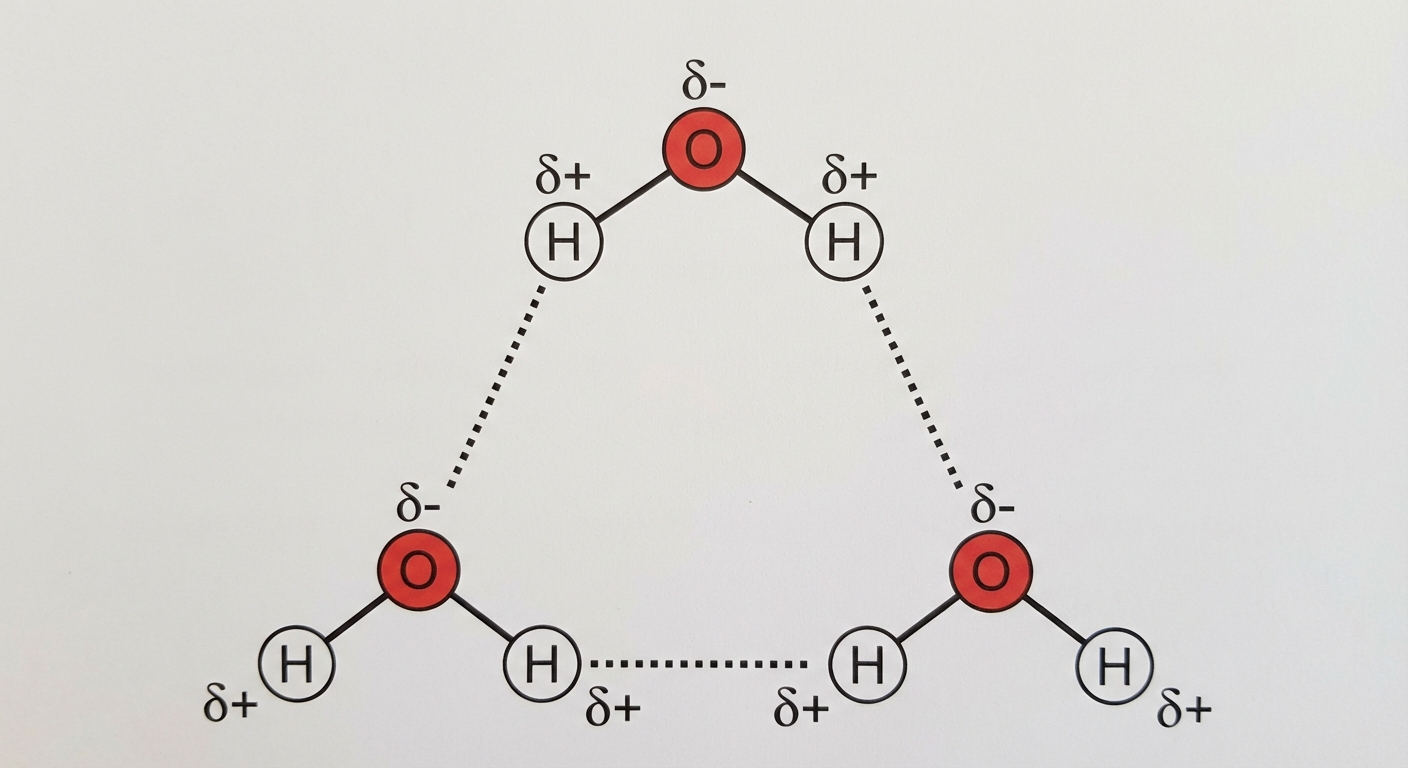
- Polarity: Because Oxygen is more electronegative, it pulls the shared electrons toward itself. This creates a partial negative charge ($\delta-$) on the Oxygen atom and a partial positive charge ($\delta+$) on the Hydrogen atoms.
Because the molecule is bent (shape matters!), the charge is unevenly distributed, making water a polar molecule.
Hydrogen Bonds
The polarity of water molecules results in weak electrical attractions between neighboring molecules. The partial positive hydrogen of one molecule is attracted to the partial negative oxygen of a nearby molecule. This interaction is called a Hydrogen Bond.
- Strength: While individual hydrogen bonds are weak (fragile), they form, break, and reform with high frequency. Collectively, they are incredibly strong and responsible for water's unique properties.
- Notation: In diagrams, covalent bonds are usually solid point-to-point lines, while hydrogen bonds are often represented as dotted or dashed lines.
Emergent Properties of Water
Due to hydrogen bonding, water exhibits four emergent properties that contribute to Earth's suitability for life.
1. Cohesion, Adhesion, and Surface Tension
- Cohesion: The attraction of molecules for other molecules of the same kind. In water, hydrogen bonds hold the substance together. This allows for phenomena like water transport in plants against gravity.
- Adhesion: The clinging of one substance to another. Water adhering to cell walls helps counter the downward pull of gravity.
- Surface Tension: A measure of how difficult it is to stretch or break the surface of a liquid. Water has an unusually high surface tension because molecules at the surface are hydrogen-bonded to one another and to the water below, but not to the air above.
Real World Example: Transpiration in plants relies on cohesion and adhesion. As water evaporates from a leaf (transpiration pull), the chain of water molecules within the xylem is pulled upward from the roots.
2. Temperature Regulation (High Specific Heat)
Water resists changing its temperature because of its high specific heat. Specific heat is the amount of heat that must be absorbed or lost for 1 g of that substance to change its temperature by 1°C.
- Mechanism: Heat is thermal energy. When water absorbs heat, much of that energy is used to break hydrogen bonds before the water molecules can begin moving faster (which raises temperature). Conversely, when water cools, hydrogen bonds form and release energy.
- Biological Relevance: This property allows large bodies of water to absorb and store huge amounts of heat from the sun with only a slight change in temperature. It also helps organisms (which are mostly water) maintain homeostasis (constant internal body temperature).
Evaporative Cooling: As a liquid evaporates, the surface of the liquid that remains behind cools down. This occurs because the "hottest" molecules form gas first. This helps prevents terrestrial organisms from overheating (e.g., sweating in humans, panting in dogs).
3. Density of Ice (Expansion upon Freezing)
Unlike most substances, water is less dense as a solid than as a liquid. This is due to the geometry of hydrogen bonds.
- Liquid Water: Hydrogen bonds constantly break and reform; molecules slide past each other.
- Solid Ice: At 0°C, molecules become locked into a crystalline lattice. The hydrogen bonds keep the molecules at "arm's length," far enough apart to make ice about 10% less dense than liquid water.
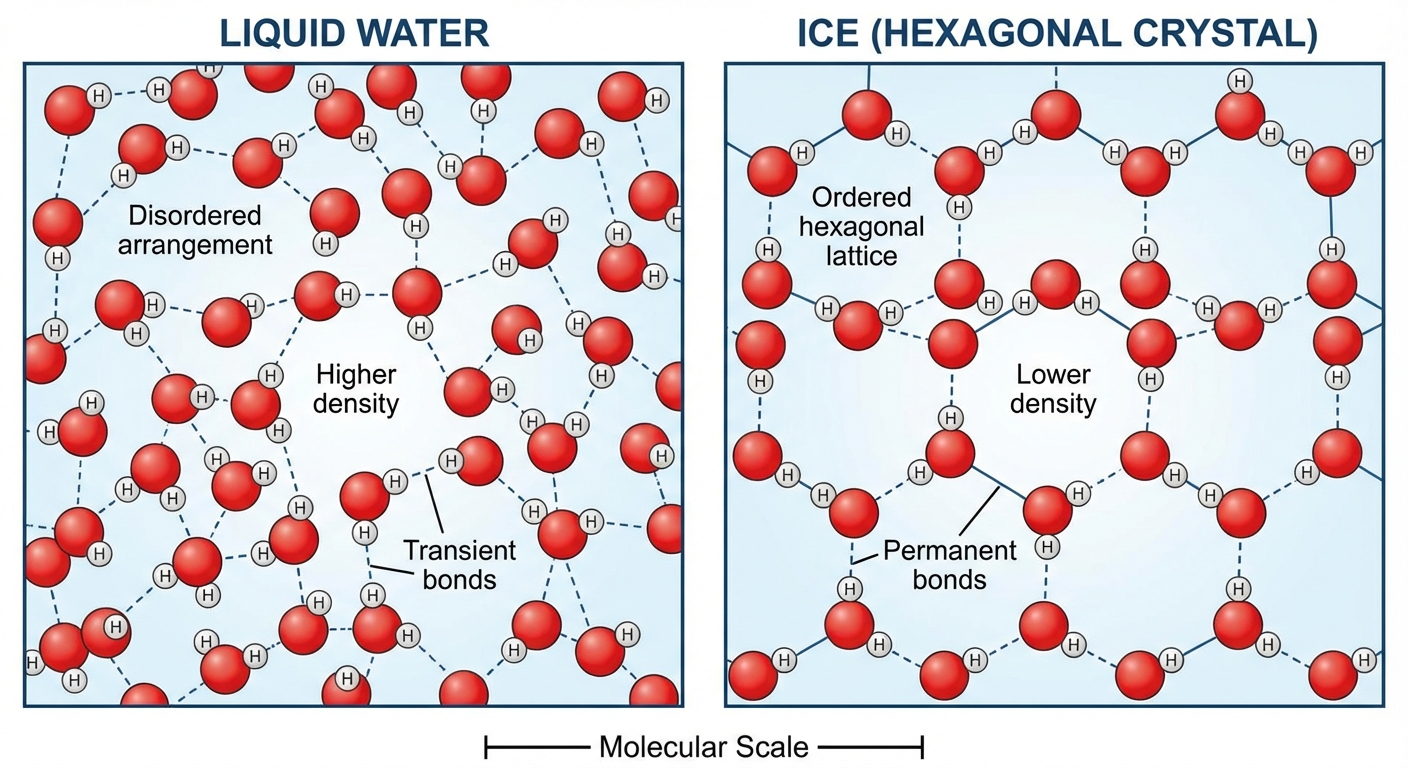
Ecological Significance: If ice sank, all bodies of water would eventually freeze solid, making life impossible on Earth. Instead, ice floats, insulating the liquid water below and preventing it from freezing, allowing aquatic life to survive winter.
4. Water as a Universal Solvent
Water is a versatile solvent because of its polarity.
- Hydration Shells: When an ionic compound (like NaCl) is dissolved in water, each ion is surrounded by a sphere of water molecules called a hydration shell. The positive hydrogens surround the anion ($Cl^-$), and negative oxygens surround the cation ($Na^+$).
- Hydrophilic: Substances that have an affinity for water (ions and polar molecules).
- Hydrophobic: Substances that repel water (nonpolar molecules like oils and lipids). These substances cannot form hydrogen bonds.
The Elements of Life
Organisms must exchange matter with the environment to grow, reproduce, and maintain organization. The Conservation of Energy law applies to living systems: atoms are not created or destroyed, only rearranged.
CHNOPS: The Major Elements
Approximately 96% of living matter consists of six elements: Carbon, Hydrogen, Nitrogen, Oxygen, Phosphorus, and Sulfur. You must know which macro-molecules utilize which elements.
| Element | Symbol | Key Biological Roles & Macromolecules |
|---|---|---|
| Carbon | C | The backbone of all organic molecules (Carbohydrates, Lipids, Proteins, Nucleic Acids). |
| Hydrogen | H | Found in all organic molecules; involved in pH regulation and energy transfer ($H^+$). |
| Nitrogen | N | Required for Proteins (amino groups) and Nucleic Acids (nitrogenous bases). |
| Oxygen | O | Found in most organic molecules; essential for cellular respiration. |
| Phosphorus | P | Required for Nucleic Acids (sugar-phosphate backbone) and Lipids (phospholipids). |
| Sulfur | S | Found in Proteins (specifically the amino acids methionine and cysteine) to form disulfide bridges. |
Carbon: The Backbone of Life
Carbon is unique in its ability to form large, complex, and diverse molecules. This is due to its electron configuration.
- Tetravalence: Carbon has 4 valence electrons. It can form 4 covalent bonds with a variety of atoms. This allows for complex molecular shapes: chains, rings, and branches.
- Bonding Versatility: Carbon can form single, double, or triple bonds. It is compatible with many different elements.
Nitrogen and Phosphorus Cycles
Living systems depend on the cycling of these elements.
- Nitrogen Source: Most nitrogen enters the biological web via bacteria that fix atmospheric nitrogen ($N_2$) into usable forms for plants. Animals acquire nitrogen by consuming plants or other animals.
- Phosphorus Source: Often derived from weathering of rocks and soil minerals. It acts as a limiting nutrient in many ecosystems.
Common Mistakes & Pitfalls
Confusing Bond Types:
- Wrong: "Hydrogen bonds hold the hydrogen and oxygen atoms together inside a water molecule."
- Right: Polar Covalent bonds are inside the molecule; Hydrogen bonds are between different water molecules.
Phase Changes:
- Wrong: "When water boils, the covalent bonds break."
- Right: When water boils, only the Hydrogen bonds break, releasing water vapor. If covalent bonds broke, you would get Hydrogen gas and Oxygen give, not steam.
Adhesion vs. Cohesion:
- Students often swap these definitions. Remember: Cohesion is with a Co-worker (same type/water-to-water). Adhesion is adding to something else (water-to-surface).
Identifying Elements:
- Be careful not to assume all macromolecules have Phosphorus. Proteins generally do not have Phosphorus, and Carbohydrates generally do not have Nitrogen.
- Mnemonic: CHO (Carbs), CHO (Lipids), CHON (Proteins), CHONP (Nucleic Acids).