Unit 8: Fundamentals of Fluids
Internal Structure and Density
Before analyzing how fluids move or exert force, we must define what a fluid is and how we quantify the amount of matter within a given space. In AP Physics 1, we treat matter not just as point masses, but as continuous distributions of mass.
Phases of Matter: Solids vs. Fluids
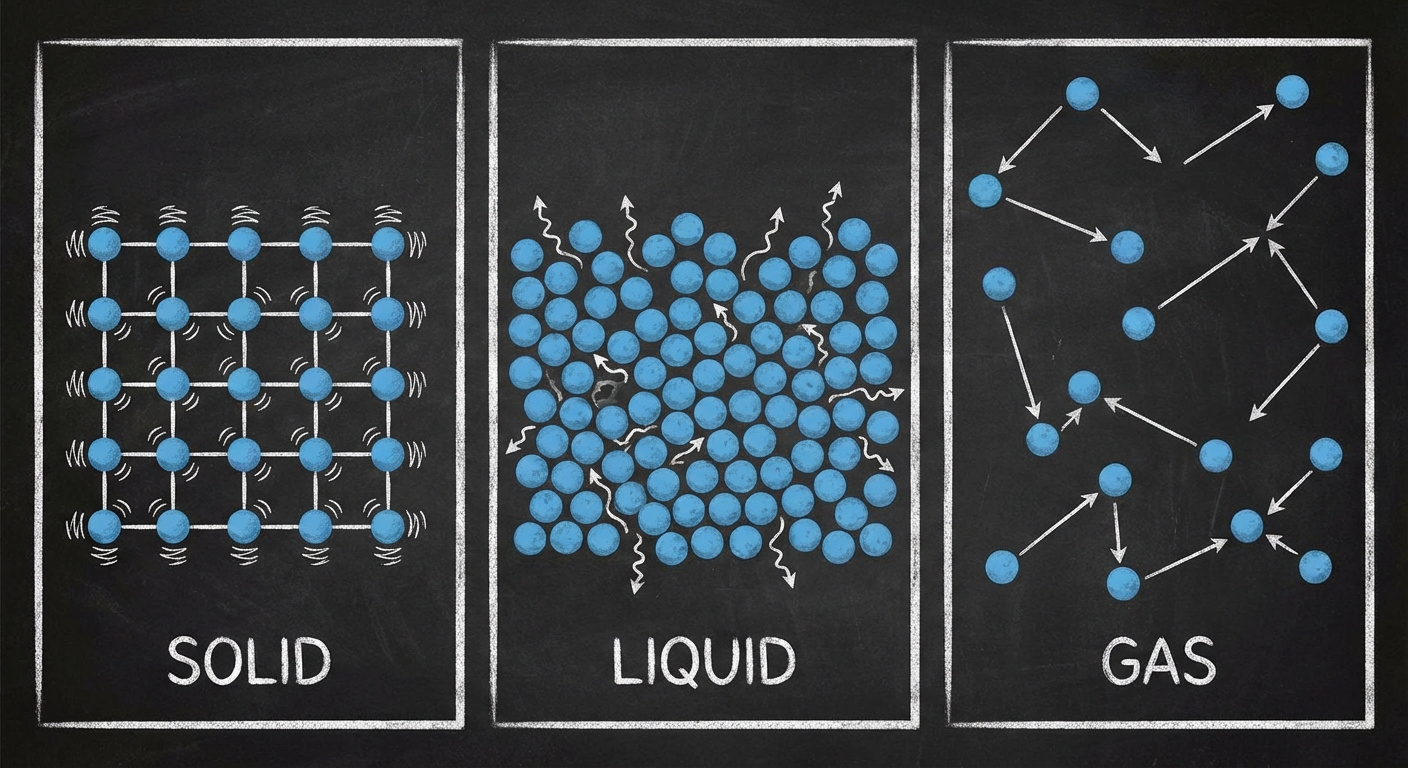
A fluid is any substance that can flow and change the shape of the volume it occupies. This category includes both liquids and gases.
- Solids: Atoms are vibrating in fixed lattice structures. They maintain a definite shape and volume. Forces between molecules are strong perpendicular to the surface.
- Liquids: Atoms or molecules are close together (like solids) but are free to slide past one another. They have a definite volume but no definite shape (they take the shape of their container).
- Gases: Molecules are far apart and move randomly at high speeds. They have neither definite shape nor volume (they expand to fill the container).
Defining Density ($
ho$)
Because fluids do not have a fixed shape, it is not convenient to talk about their total mass ($m$) in dynamic situations. Instead, we use density, defined as mass per unit volume. The symbol for density is the Greek letter rho ($
ho$).
Where:
- $\rho$ is density (kg/m³)
- $m$ is mass (kg)
- $V$ is volume (m³)
Key Properties of Density
- Scalar Quantity: Density has magnitude but no direction.
- Incompressibility: In AP Physics 1, liquids (like water or oil) are generally treated as incompressible. This means their density $\rho$ remains constant regardless of the pressure applied to them.
- Gases are Compressible: The density of a gas can change significantly if the volume of the container changes.
| Substance | Approx. Density (kg/m³) |
|---|---|
| Air (at sea level) | $1.2$ |
| Water (fresh) | $1,000$ (or $1.0 \times 10^3$) |
| Seawater | $1,025$ |
| Mercury | $13,600$ |
Note: You should memorize the density of fresh water (${1000\; \text{kg/m}^3$).
Pressure
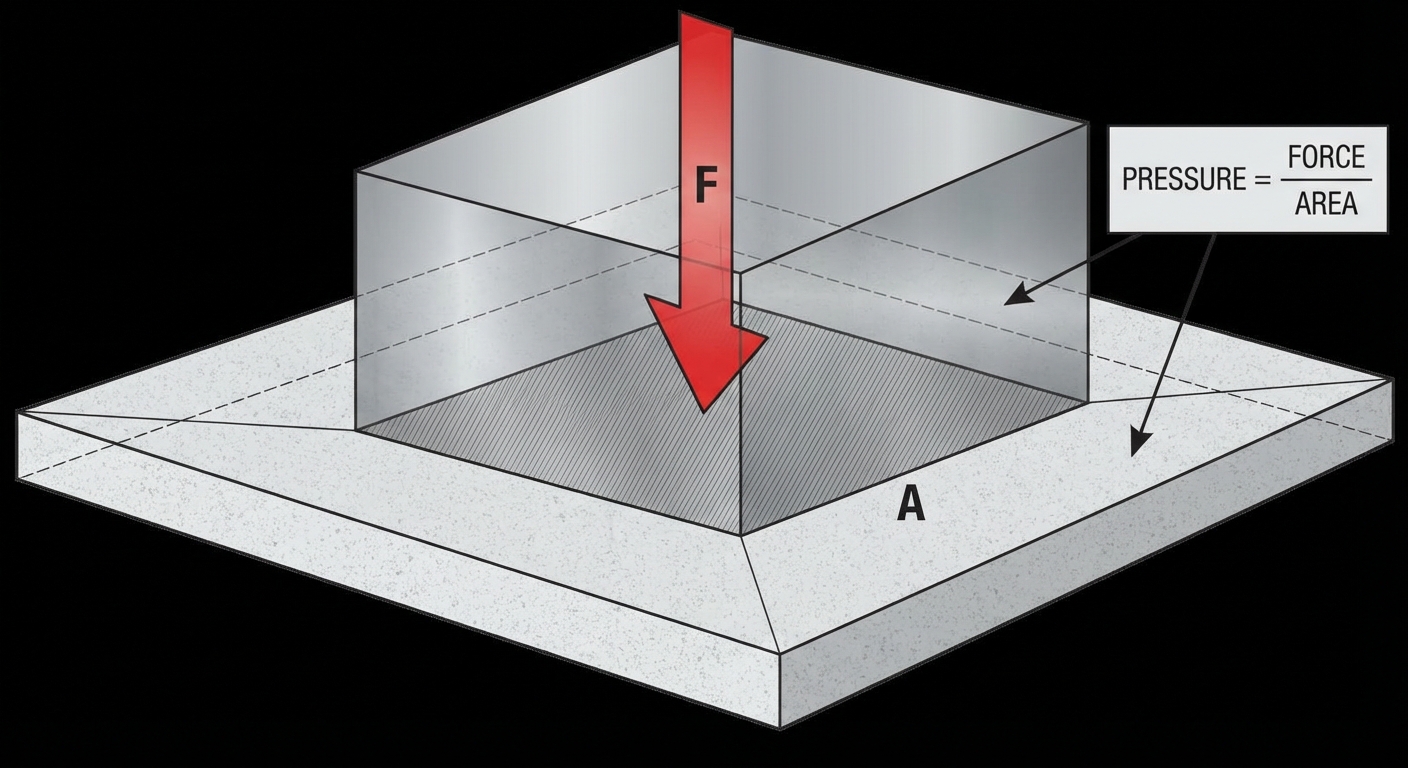
Pressure is the primary way we describe forces acting within fluids. While force describes a push or pull on a specific object, pressure describes how that force is distributed over a surface area.
Definition and Formula
Pressure ($P$) is defined as the magnitude of the force applied perpendicular to a surface, divided by the area of that surface.
Where:
- $P$ is pressure (Pascals, Pa)
- $F_{\perp}$ is the component of force perpendicular to the surface (Newtons, N)
- $A$ is the area of contact (m²)
Units
The SI unit for pressure is the Pascal (Pa).
Common alternative units you might see (but should convert to Pascals for calculation):
- Atmosphere (atm): $1 \; \text{atm} \approx 1.01 \times 10^5 \; \text{Pa}$ (standard atmospheric pressure at sea level)
The Microscopic Origin of Pressure
Why do fluids exert pressure? It comes down to kinetic theory and internal structure.
- Fluids consist of trillions of particles moving in random directions.
- These particles collide with the walls of their container or any object submerged in the fluid.
- Each collision imparts a tiny impulse ($J = \Delta p$) to the wall.
- The collective frequency and strength of these billions of collisions result in a macroscopic average force.
- Because the collisions are random, the net force is always directed perpendicular (normal) to the surface.
Key Concept: Pressure is a scalar quantity, but it generates a force that has direction (always pushing into the surface).
Pressure in Static Fluids (Hydrostatic Pressure)
When a fluid is at rest (static), the pressure is not uniform throughout the fluid; it depends on depth. This is due to the weight of the fluid column pressing down from above.
The Relationship Between Pressure and Depth
Imagine a vertical column of water. The water at the bottom must support the weight of all the water above it. Therefore, pressure increases as you go deeper.
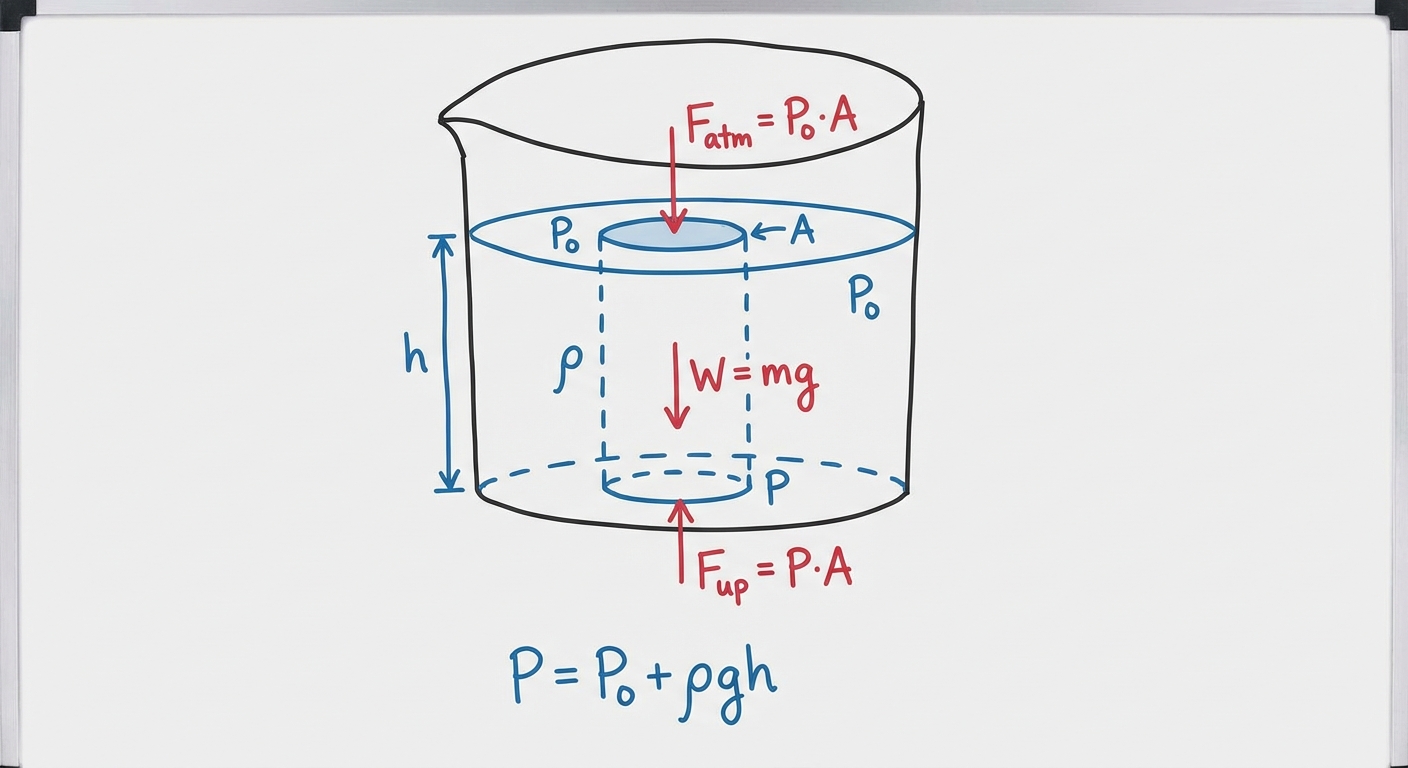
This relationship is linear and is given by the hydrostatic pressure equation:
Where:
- $P$ is the absolute pressure at depth $h$
- $P0$ is the external pressure at the surface (usually Atmospheric Pressure, $P{atm}$)
- $\rho$ is the density of the fluid
- $g$ is the acceleration due to gravity ($9.8 \; \text{m/s}^2$)
- $h$ is the depth below the surface (positive downwards)
Absolute vs. Gauge Pressure
It is vital to distinguish between total pressure and the pressure added by the fluid alone.
Gauge Pressure ($P{gauge}$): This measures the pressure difference between the fluid and the atmosphere. It ignores the weight of the air above the liquid. Examples: Tire pressure gauges, blood pressure cuffs.
Absolute Pressure ($P{total}$ or $P{abs}$): This is the true total pressure acting at a point, including the atmospheric pressure pushing on the surface of the fluid.
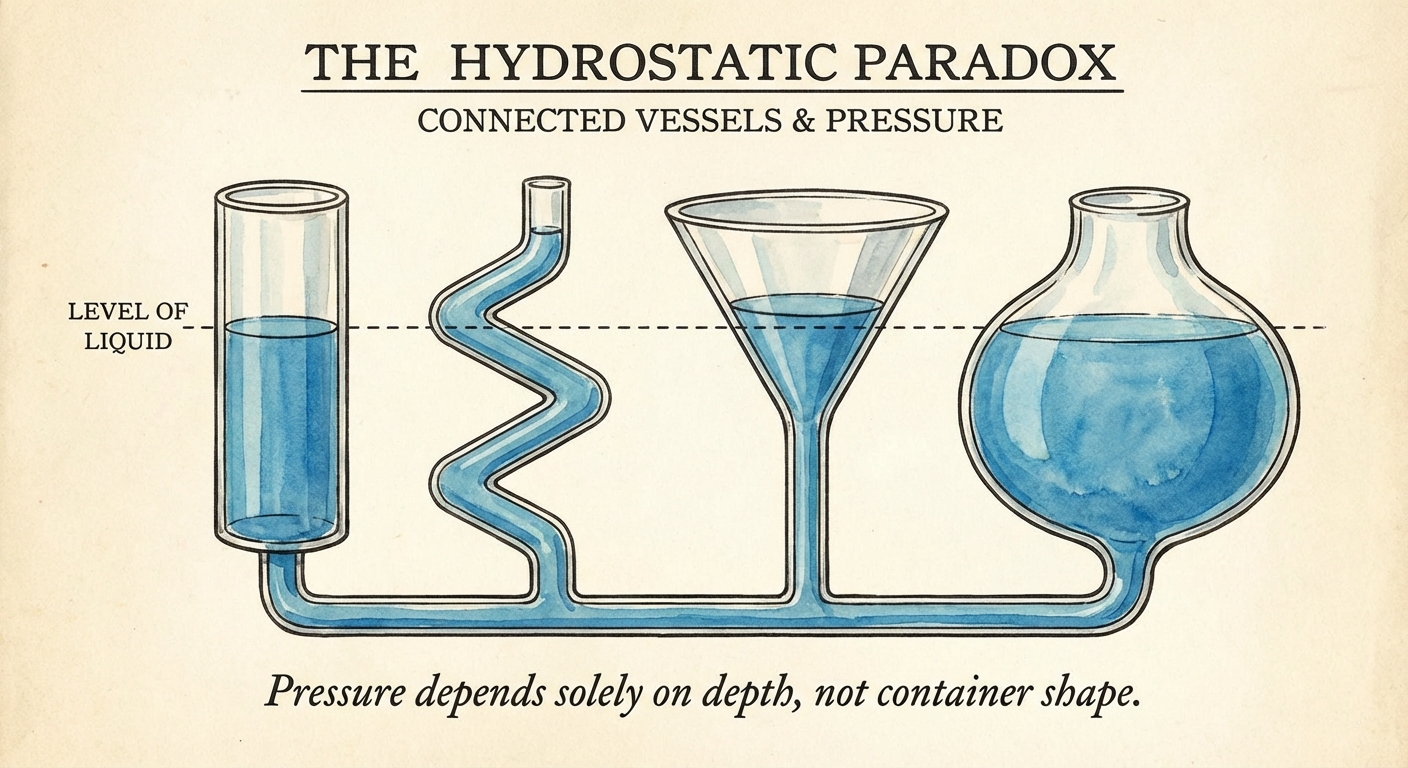
The Shape of the Container (Hydrostatic Paradox)
A common counter-intuitive concept in AP Physics 1 is that the pressure at a specific depth depends only on the depth ($h$), the fluid density ($\rho$), and gravity ($g$).
It does NOT depend on:
- The shape of the container.
- The total volume of water in the container.
- The surface area of the liquid at the top.
If you have a thin straw and a wide pool connected at the bottom, the water level will rise to the same height in both, and the pressure at the bottom will be identical if the depth is the same.
Worked Example: The Swimming Pool
Problem:
A diver is swimming in a freshwater lake ($\rho = 1000 \; \text{kg/m}^3$) at a depth of $5.0$ meters. If atmospheric pressure is $1.01 \times 10^5 \; \text{Pa}$, calculate:
- The gauge pressure on the diver.
- The absolute pressure on the diver.
Solution:
Step 1: Calculate Gauge Pressure
Step 2: Calculate Absolute Pressure
Common Mistakes & Pitfalls
Confusing Depth ($h$) with Altitude:
In the formula $P = P_0 + \rho g h$, $h$ is defined as depth below the surface. Do not measure from the bottom of the container up. As typically defined in this context, $h=0$ at the surface and increases as you go down.Neglecting Atmospheric Pressure:
If a question asks for the "total pressure" or "absolute pressure," you MUST add $P{atm}$ (usually $10^5$ Pa). If it asks for the "pressure exerted by the water" or "gauge pressure," you exclude $P{atm}$.Assuming Pressure has a Direction:
Pressure is a scalar. It does not go "down." However, the force resulting from pressure acts perpendicular to the surface of the object. On the side of a submerged cube, the force acts horizontally inward. On the bottom, it acts vertically upward.Mixing Units:
Ensure density is in $\text{kg/m}^3$. A common trap is providing density in $\text{g/cm}^3$ (e.g., $1.0 \; \text{g/cm}^3$). You must convert this to $1000 \; \text{kg/m}^3$ before calculating.