Unit 3 Study Guide: Cellular Energetics and Photosynthesis
Fundamentals of Cellular Energy
Biological systems are governed by the laws of chemistry and physics. Before understanding how plants make food, you must understand how energy moves through living systems.
Laws of Thermodynamics
- First Law (Conservation of Energy): Energy cannot be created or destroyed, only transformed. The total energy of the universe is constant. Plants do not "create" energy; they transform light energy into chemical energy.
- Second Law (Entropy): Every energy transfer increases the entropy (disorder) of the universe. During energy transfer, some energy is lost as heat. Organisms fight entropy by constantly inputting energy.
Metabolic Pathways
Metabolism is the sum of an organism's chemical reactions.
- Catabolic Pathways: Break down complex molecules into simpler compounds. They release energy.
- Example: Cellular Respiration.
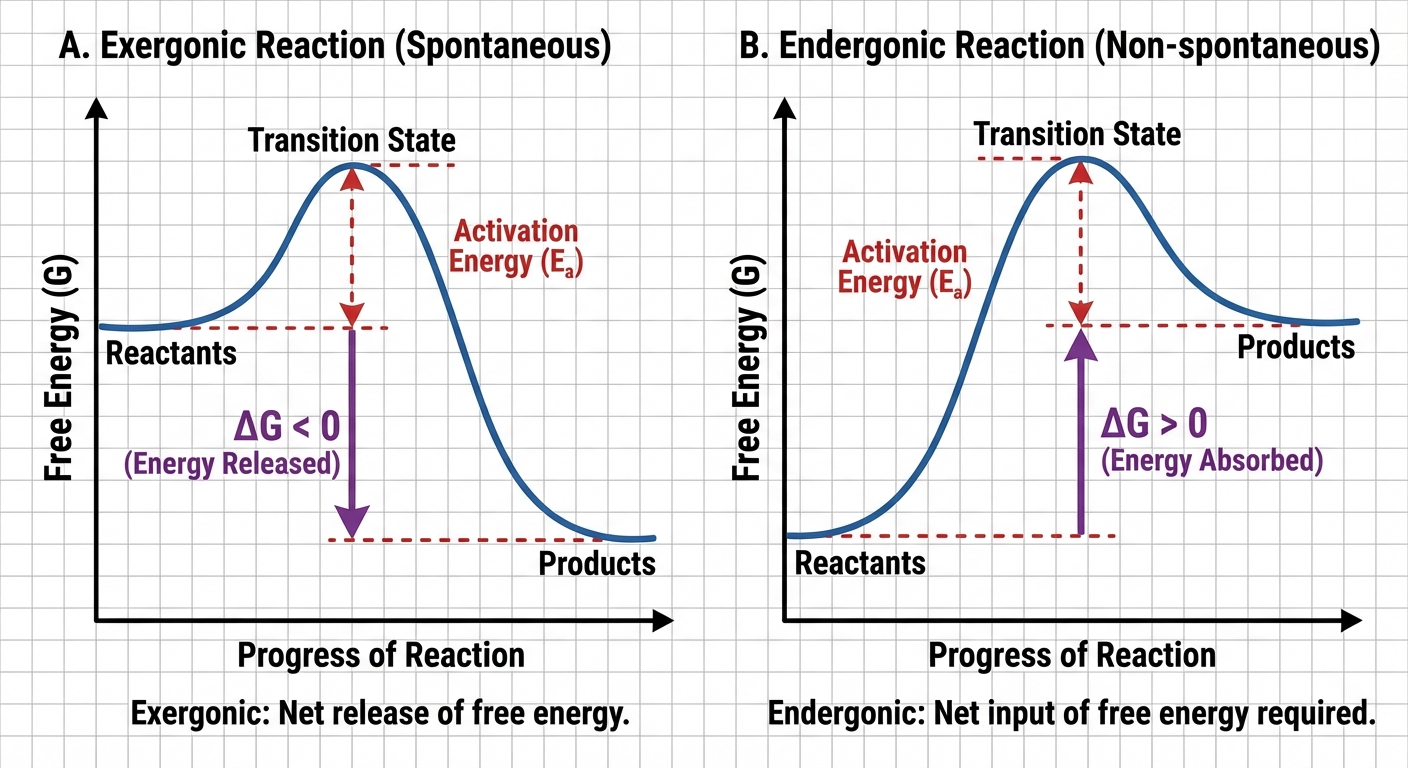
- Exergonic Reaction: A reaction with a net release of free energy ($\Delta G < 0$).
- Anabolic Pathways: Consume energy to build complex molecules from simpler ones.
- Example: Photosynthesis (building sugar from $CO_2$).
- Endergonic Reaction: A reaction that absorbs free energy from its surroundings ($\Delta G > 0$).
Energy Coupling and ATP
Cells manage their energy resources by energy coupling—using an exergonic process to drive an endergonic one. The primary molecule responsible for this is ATP (Adenosine Triphosphate).
- Structure: Composed of the nitrogenous base adenine, a ribose sugar, and a chain of three phosphate groups.
- Hydrolysis: The bonds between the phosphate groups can be broken by hydrolysis. When the terminal phosphate bond is broken, energy is released.
- Phosphorylation: The released inorganic phosphate ($P_i$) is often transferred to another molecule, making that molecule more reactive (less stable).
Introduction to Photosynthesis
Photosynthesis is the process that converts solar energy into chemical energy. It captures light energy and uses it to convert carbon dioxide and water into glucose (or chemical equivalents) and oxygen.
The Chemical Equation
The overall simplified chemical equation is:
Note: While often written as producing glucose, the direct product of the Calvin Cycle is actually a 3-carbon sugar called G3P (Glyceraldehyde 3-phosphate).
Evolution of Photosynthesis
Photosynthesis first evolved in prokaryotic organisms (cyanobacteria). Evidence suggests that prokaryotic photosynthesis was responsible for the oxygenation of the atmosphere (the Great Oxidation Event). Eukaryotic photosynthesis evolved via endosymbiosis—where a eukaryotic host cell engulfed a photosynthetic prokaryote becomes the chloroplast.
Chloroplast Structure
In eukaryotes, photosynthesis occurs in the chloroplast.
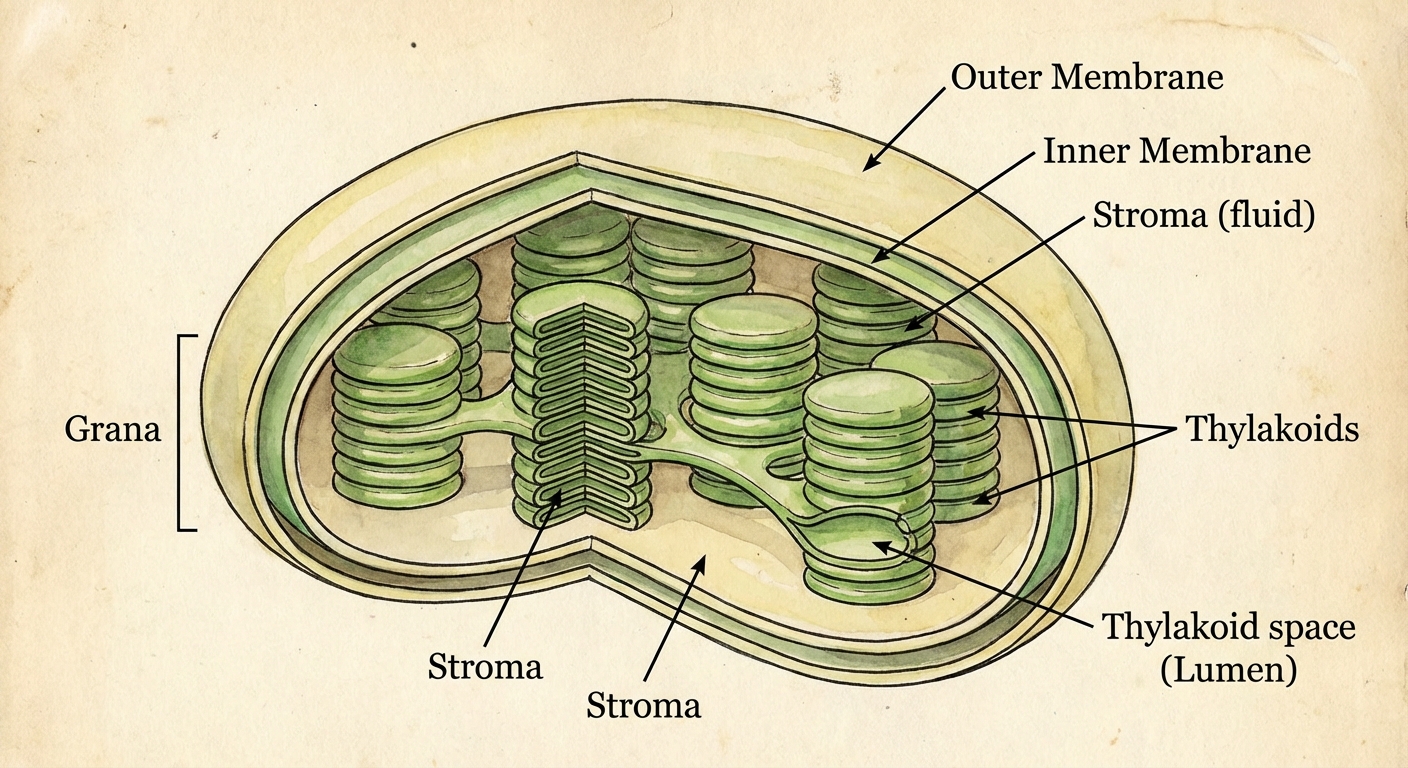
- Thylakoids: Sac-like photosynthetic membranes. The Light-Dependent Reactions occur here (specifically in the membrane and the inner lumen).
- Grana: Stacks of thylakoids.
- Stroma: The dense fluid outside the thylakoids. The Calvin Cycle (Light-Independent Reactions) occurs here.
- Chlorophyll: The green pigment located within the thylakoid membranes that absorbs light energy.
The Light-Dependent Reactions
Goal: Convert solar energy into chemical energy (ATP and NADPH).
Location: Thylakoid Membranes.
Photosystems
Chloroplasts use photosystems, which are reaction-center complexes surrounded by light-harvesting complexes. There are two types, named in order of discovery, not function (hence the numbering can be confusing):
- Photosystem II (PSII): Functions first. Absorbs light best at 680 nm (P680).
- Photosystem I (PSI): Functions second. Absorbs light best at 700 nm (P700).
The Linear Electron Flow (Step-by-Step)
- Photon Absorption (PSII): Light stikes pigments in PSII, exciting electrons to a higher energy state. These electrons are accepted by the primary electron acceptor.
- Photolysis of Water: An enzyme splits water ($H_2O$) into 2 electrons, 2 $H^+$ ions (protons), and 1 oxygen atom.
- The electrons replace those lost by P680.
- The $H^+$ helps generate the gradient.
- The $O$ combines with another $O$ to form $O_2$ (released as a byproduct).
- Electron Transport Chain (ETC): Excited electrons travel from PSII to PSI via an ETC. As electrons fall down the chain, their energy is used to pump protons ($H^+$) from the stroma into the thylakoid space (lumen).
- Photon Absorption (PSI): Light energy re-excites electrons in PSI (P700). These electrons are passed to a second ETC.
- NADPH Production: The enzyme NADP+ reductase transfers electrons to $NADP^+$ to form NADPH (an electron carrier).
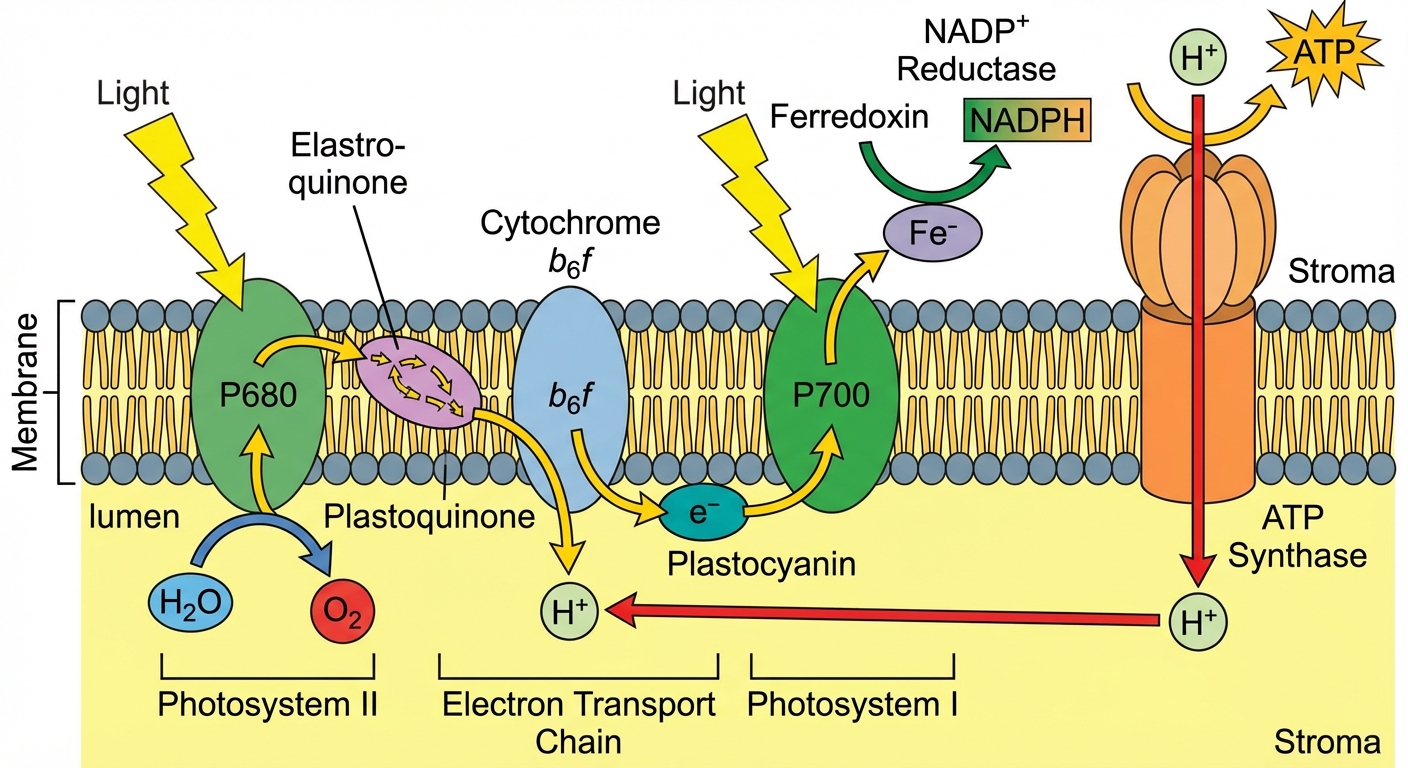
Chemiosmosis and Photophosphorylation
The ETC builds a high concentration of $H^+$ inside the thylakoid lumen. This defines a proton-motive force.
- ATP Synthase: Protons diffuse back out to the stroma through the ATP Synthase channel.
- This flow drives the phosphorylation of ADP to ATP.
- This is called Photophosphorylation.
The Calvin Cycle (Light-Independent Reactions)
Goal: Use ATP and NADPH to reduce $CO_2$ to sugar (G3P).
Location: Stroma.
The cycle is anabolic (builds sugar) and requires 3 turns to synthesize one net molecule of G3P.
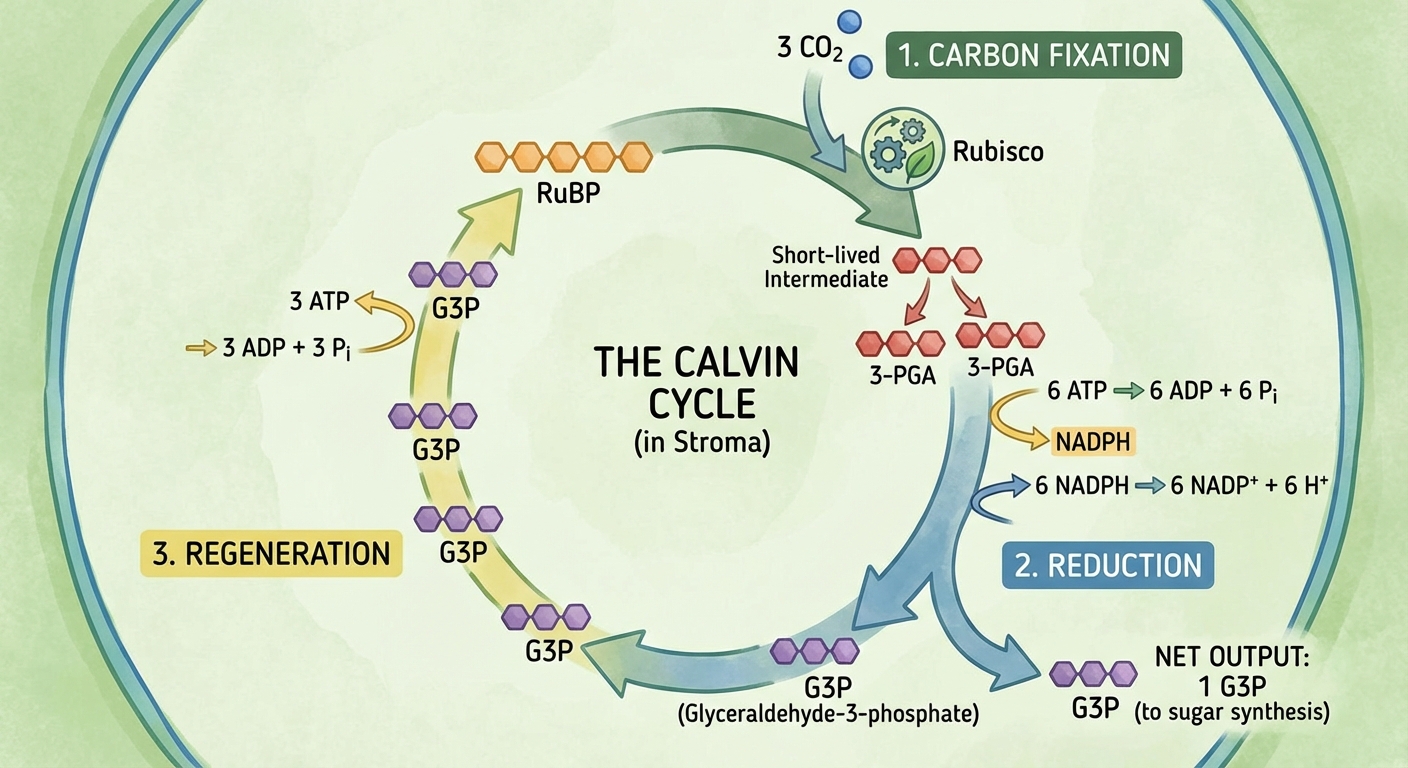
Phase 1: Carbon Fixation
- A $CO_2$ molecule is attached to a 5-carbon sugar named RuBP (ribulose bisphosphate).
- The enzyme that catalyzes this step is Rubisco (the most abundant protein on Earth).
- The resulting 6-carbon intermediate is unstable and splits into two 3-carbon molecules (3-phosphoglycerate).
Phase 2: Reduction
- ATP phosphorylates the 3-carbon intermediate.
- NADPH donates electrons (reduces) the molecule to form G3P.
- For every 3 molecules of $CO_2$ entering, 6 molecules of G3P are formed, but only one exits the cycle to be used by the plant cell. The other 5 must be recycled.
Phase 3: Regeneration of RuBP
- The carbon skeletons of the remaining 5 G3P molecules are rearranged into 3 molecules of RuBP.
- This step requires input of more ATP.
- RuBP is now ready to receive $CO_2$ again.
| Feature | Light-Dependent Reactions | Calvin Cycle |
|---|---|---|
| Location | Thylakoid Membrane | Stroma |
| Inputs | Light, $H_2O$, $NADP^+$, ADP | $CO_2$, ATP, NADPH |
| Outputs | $O_2$, ATP, NADPH | G3P (Sugar), ADP, $NADP^+$ |
Mnemonics & Memory Aids
- OIL RIG: Oxidation Is Loss (of electrons), Reduction Is Gain (of electrons).
- NADPH vs NADH:
- P is for Photosynthesis (NADPH).
- Respiration uses NADH (no P).
- L.I.R. vs L.D.R:
- Light Dependent reactions occur in the Lumen (technically the gradient is built there).
- Calvin Cycle occurs in the Cytoplasm of the chloroplast (the Stroma).
Common Mistakes & Pitfalls
"Plants do photosynthesis, Animals do respiration."
- Correction: Plants do BOTH. They have mitochondria to break down the sugar they make in the chloroplasts to generate ATP for cellular work.
Confusing the Location of the H+ Gradient.
- Correction: In cellular respiration (mitochondria), protons are pumped out of the matrix into the intermembrane space. In photosynthesis (chloroplasts), protons are pumped in to the thylakoid lumen from the stroma.
The "Dark" Reactions.
- Correction: Do not call the Calvin Cycle the "Dark Reactions." It implies they only happen at night. They happen during the day because they require the ATP and NADPH produced by the light reactions.
Rubisco vs RuBP.
- Correction: RuBP is the substrate (the sugar). Rubisco is the enzyme (the protein) that fixes carbon.