AP Environmental Science: Unit 7 - Atmospheric Pollution Guide
Unit 7: Atmospheric Pollution Study Notes
7.1 Introduction to Air Pollution
Air Pollution constitutes the introduction of chemicals, particulate matter, or biological materials that cause harm or discomfort to humans or other living organisms, or damage the natural environment.
Primary vs. Secondary Pollutants
Understanding the source and formation of pollutants is critical for the AP exam.
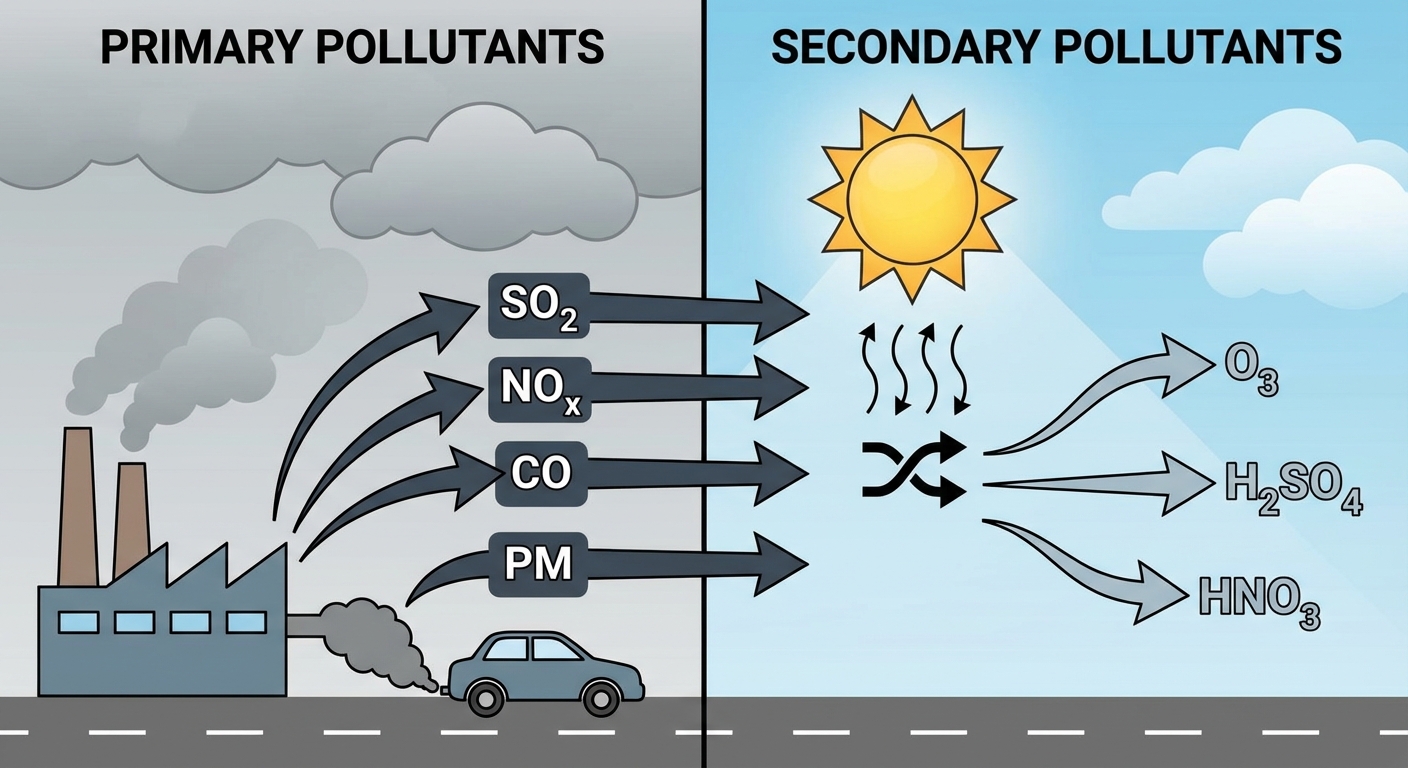
- Primary Pollutants: Emitted directly from a source into the atmosphere in a harmful form.
- Examples: Carbon Monoxide (CO), Carbon Dioxide ($CO2$), Sulfur Dioxide ($SO2$), Nitrogen Oxides ($NO_x$), and suspended particulate matter.
- Sources: Smokestacks, car exhaust pipes, volcanoes.
- Secondary Pollutants: Formed when primary pollutants react with each other or with naturally occurring components (like water vapor or oxygen) in the presence of sunlight.
- Examples: Tropospheric Ozone ($O3$), Sulfuric Acid ($H2SO4$), Nitric Acid ($HNO3$), Peroxyacyl Nitrates (PANs).
The Clean Air Act & Criteria Pollutants
The Clean Air Act (CAA) requires the EPA to set acceptable limits for six "criteria air pollutants" that threaten public health and the environment.
Mnemonic: SPLONC
- Sulfur Dioxide ($SO_2$)
- Particulate Matter (PM)
- Lead (Pb)
- Ozone (Tropospheric $O_3$)
- Nitrogen Dioxide ($NO_2$)
- Carbon Monoxide (CO)
(Note: $CO_2$ is NOT a criteria pollutant under the original CAA, though it is regulated as a greenhouse gas. Do not confuse the two on the exam.)
Categories of Pollution Sources
- Anthropogenic: Human-made sources (burning fossil fuels, agriculture, industrial processes).
- Natural: Non-human sources (volcanic eruptions, forest fires, dust storms, plant terpenes).
- Point Source: A specific, fixed location causing pollution (e.g., a single factory smokestack).
- Non-Point Source: Diffuse sources that are hard to isolate (e.g., runoff from all cars in a city, agricultural pesticides blown by wind).
7.2 Photochemical Smog (Brown Smog)
Photochemical smog is formed when nitrogen oxides ($NO_x$) and volatile organic compounds (VOCs) react with heat and sunlight to produce a variety of pollutants.
The Chemistry of Smog
This reaction is driven by solar energy (UV radiation), meaning smog levels are highest during the summer and at midday.
- Nitrogen Oxides ($NO_x$): Produced by combustion in vehicle engines.
- VOCs (Volatile Organic Compounds): Organic chemicals that evaporate easily (e.g., gasoline fumes, paint solvents, tree sap).
- Process:
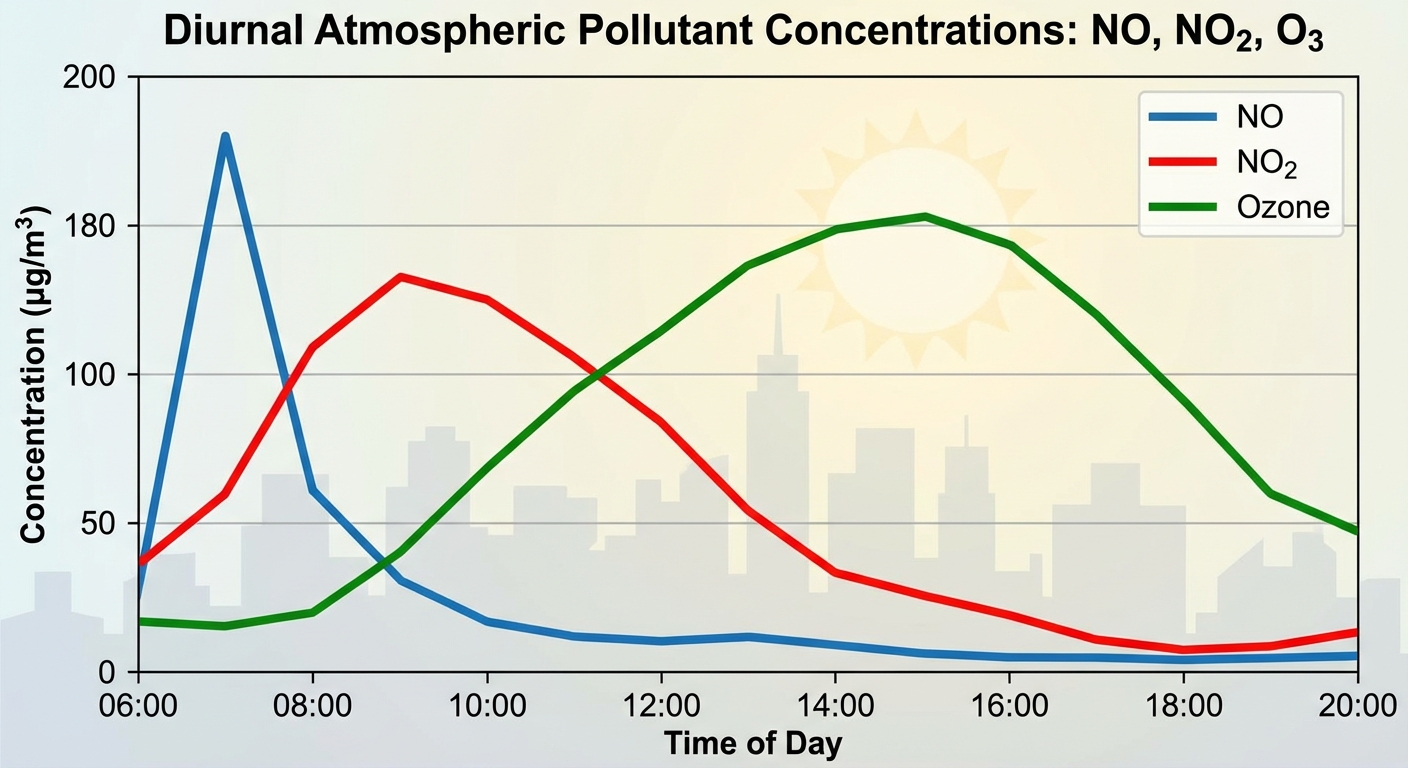
Temporal Formation (Time of Day)
- Morning (6-9 AM): Commuter traffic increases $NO$ and VOC concentrations.
- Mid-morning (9-11 AM): $NO$ reacts with oxygen to form $NO_2$.
- Afternoon (12-4 PM): Intense sunlight breaks down $NO2$ and drives reactions between $O2$ and VOCs, creating peak Ozone ($O_3$) and PANs.
- Evening: As the sun sets, ozone production halts. Ozone may react with remaining $NO$ to degrade, lowering concentrations overnight.
Effects of Photochemical Smog
- Respiratory: Asthma, bronchitis, eye irritation.
- Environmental: Inhibits photosynthesis in plants; damages crops.
- Visibility: Creates a brown haze (due to $NO_2$) blocking sunlight.
7.3 Thermal Inversion
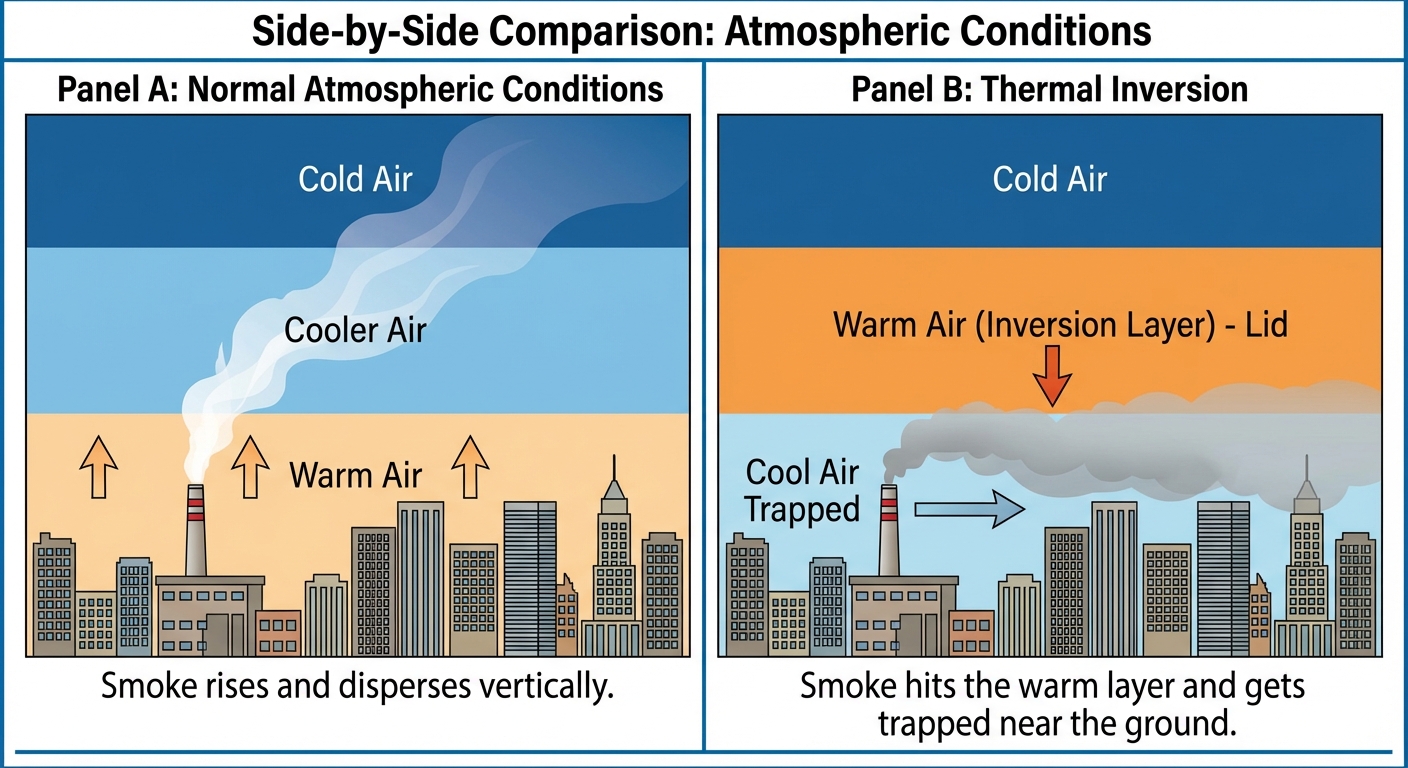
Under normal conditions, the atmosphere cools as altitude increases. This allows warm air (and pollution) to rise and disperse.
The Inversion Mechanism
A Thermal Inversion occurs when a layer of warm air settles over a layer of cooler air near the ground. This "cap" traps pollutants close to the surface.
- Normal Pattern: Warm ground $\rightarrow$ Cool Air $\rightarrow$ Cold Air (Pollution rises and scatters).
- Inversion Pattern: Cool ground $\rightarrow$ Warm Inversion Layer $\rightarrow$ Cold Air (Pollution is trapped).
Common Locations
- Valleys: Cool air sinks into the valley at night, while warm air passes over the mountains, trapping the cool air below.
- Mountain Ranges: Mountains can block air flow, preventing pollutants from dispersing (e.g., Los Angeles, Mexico City).
7.4 Atmospheric CO2 and Particulates
Particulate Matter (PM)
PM refers to solid or liquid particles suspended in the air. It is classified by size:
- PM10 (Dust, smoke, pollen): Can irritate eyes and throat.
- PM2.5 (Combustion particles, organic compounds, metals): More dangerous. Small enough to bypass respiratory filters (nose hairs/cilia), lodge deep in the lungs, and enter the bloodstream.
Sources:
- Natural: Volcanoes, forest fires, dust storms, sea spray.
- Anthropogenic: Fossil fuel combustion (coal/oil), road dust, construction, agriculture.
Carbon Dioxide ($CO_2$)
While not a "toxin" to breath at ambient levels, it is the primary Green House Gas (GHG) responsible for climate change.
- Sources: Respiration (natural), decomposition (natural), volcanic eruptions (natural), and fossil fuel combustion (anthropogenic).
- Trend: Atmospheric levels have risen from ~280 ppm (pre-industrial) to over 400 ppm today.
7.5 Indoor Air Pollutants
Detailed knowledge of specific indoor pollutants is a frequent exam topic. Indoor air pollution can often be worse than outdoor pollution due to poor ventilation.
Developing vs. Developed Countries
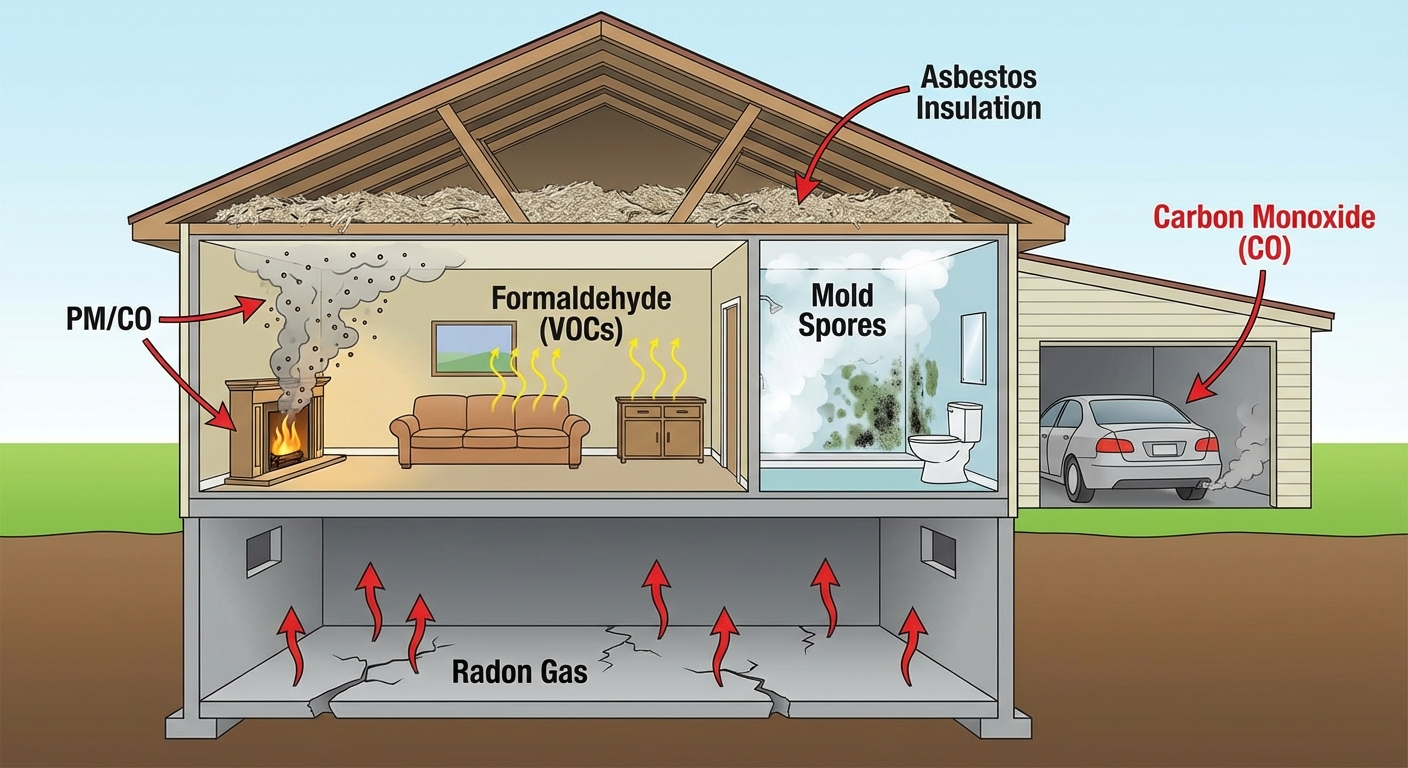
- Developing Nations: Primary indoor pollutants are Carbon Monoxide (CO) and Particulate Matter from burning biomass (wood, dung, coal) indoors for cooking/heating without proper ventilation.
- Developed Nations: Primary issues stem from tightly sealed buildings trapping chemical byproducts (VOCs, Radon).
Key Indoor Pollutants Table
| Pollutant | Source | Health Effect | Remediation |
|---|---|---|---|
| Asbestos | Old insulation, floor tiles (particulate silicate minerals). | Mesothelioma (lung cancer), scarring (Asbestosis). | Encapsulation or professional removal. |
| Carbon Monoxide (CO) | Incomplete combustion (furnaces, gas heaters, wood stoves). | Binds to hemoglobin, blocking oxygen transport; fatal (asphyxiation). | CO detectors, proper maintenance of heaters. |
| Radon-222 | Natural radioactive decay of Uranium in soil/rock. Enters via cracks in foundation. | 2nd leading cause of Lung Cancer (after smoking). | Seal foundation cracks, install ventilation systems. |
| VOCs (Formaldehyde) | Furniture glues, carpets, particleboard, upholstery. | Eye/throat irritation, headaches, carcinogen. | Proper ventilation, air-purifying plants. |
| Lead | Old paint (chipping/dust), old water pipes. | Neurotoxin (brain damage), learning disabilities in children. | Remove lead paint standard, water filtration. |
| Mold | Damp, humid areas (bathrooms, leaks). | Asthma, allergic reactions. | Ventilation, dehumidifiers, fixing leaks. |
7.6 Reduction of Air Pollutants
Methods to reduce air pollution fall into regulatory (laws) and technological solutions.
Technological Solutions
- Vapor Recovery Nozzles: Used at gas stations to capture gasoline fumes (VOCs) that would escape while refueling cars.
- Catalytic Converters: Device in cars that converts harmful pollutants into less harmful ones.
- Wet and Dry Scrubbers: Used in industrial smokestacks (coal plants) to remove Sulfur Dioxide ($SO_2$) and Particulates.
- Mechanism: Uses a fine mists of water and limestone (calcium carbonate) to trap pollutants as sludge.
- Electrostatic Precipitators: Used in coal plants to remove Particulate Matter.
- Mechanism: Uses an electrical charge to attract particles to metal plates, cleaning the "smoke."
Regulatory Solutions
- CAFE Standards (Corporate Average Fuel Economy): Government regulations that mandate higher fuel efficiency for vehicles, reducing fuel consumption and emissions.
- Pollution Credits (Cap and Trade): A market-based approach where companies can buy/sell allowances for emissions (specifically successful with $SO_2$).
7.7 Acid Rain (Acid Deposition)
Acid Deposition refers to rain, snow, fog, or dust with a pH lower than 5.6.
Formation Chemistry
Two primary pollutants drive acid rain:
- Nitrogen Oxides ($NO_x$): From vehicles/coal.
- Sulfur Dioxide ($SO_2$): From coal-burning power plants.
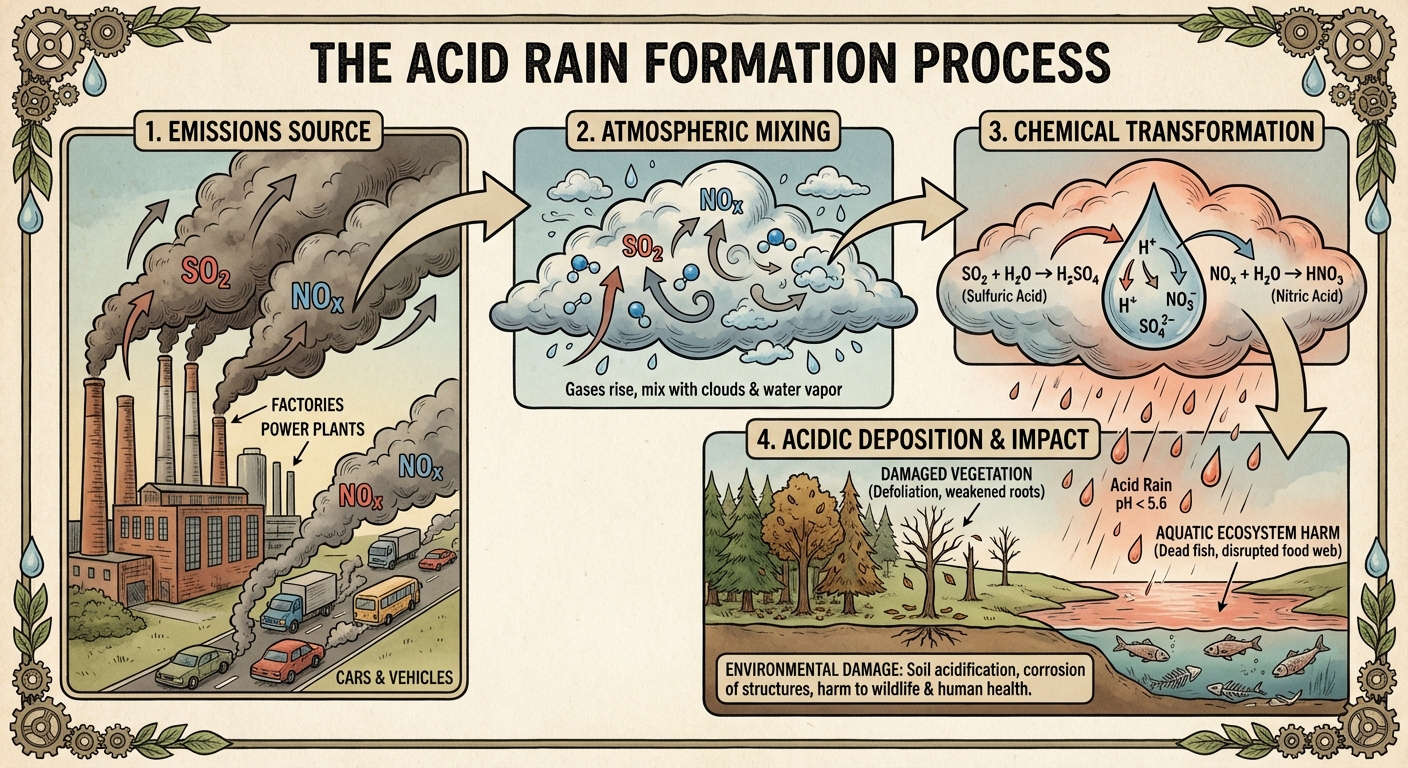
Environmental Impacts
- Soil and Forests: Acid rain leaches essential nutrients (calcium, magnesium) from the soil. It also dissolves Aluminum ions in the soil, which are toxic to plant roots.
- Aquatic Life: Lowers the pH of lakes/rivers beyond the range of tolerance for fish and amphibians. Aluminum runoff suffocates fish by damaging gills.
- Man-made Structures: Corrodes statues and buildings made of limestone or marble (Calcium Carbonate).
Regional Factors
- Limestone Bedrock: Areas with limestone bedrock have a higher buffering capacity. The calcium carbonate neutralizes the acid, minimizing damage.
- Granite Bedrock: Areas with granite (e.g., Northeast US) cannot buffer acid, resulting in severe ecosystem damage.
7.8 Noise Pollution
Noise Pollution is sound at levels high enough to cause physiological stress and hearing loss.
Human Health Effects
- Physiological: Hearing loss, high blood pressure, heart disease.
- Cognitive: Difficulty concentrating, anxiety, sleep disruption.
Ecological Effects
- Masking: Loud noises (ships, traffic, sonar) interfere with animal communication (e.g., whales, birds).
- Example: Whales rely on echolocation to hunt and communicate; sonar can cause beaching or separation from pods.
- Example: Birds may have to alter their singing frequencies or migrate away from urban areas.
- Predator/Prey Confusion: Noise prevents predators from hearing prey or prey from hearing approaching predators.
Common Mistakes & Pitfalls (Unit 7)
- Stratospheric vs. Tropospheric Ozone:
- Mistake: Thinking ozone is always good or always bad.
- Correction: "Good up high, bad nearby." Stratospheric ozone blocks UV radiation (Good). Tropospheric ozone is a lung irritant and smog component (Bad).
- Greenhouse Effect vs. Ozone Depletion:
- Mistake: Thinking CFCs cause global warming or that $CO_2$ causes the ozone hole.
- Correction: These are two separate issues. CFCs destroy the Ozone layer ($O3$). Greenhouse gases ($CO2$, Methane) trap heat.
- Acid Rain vs. Climate Change:
- Mistake: Thinking Acid Rain causes Global Warming.
- Correction: Acid rain is a local/regional issue caused by $SO2$/$NOx$. Climate change is a global issue caused by GHGs.
- CO vs CO2:
- Mistake: Using them interchangeably.
- Correction: CO is a lethal toxin (binds hemoglobin). $CO_2$ is a greenhouse gas (affects climate) and suffocant only at extreme concentrations.