AP Biology Unit 2: Transport Mechanics & Osmoregulation
Principles of Membrane Transport
The survival of a cell depends on its ability to exchange materials with its environment. The plasma membrane allows the cell to maintain internal conditions different from the external environment, a property known as selective permeability.
The Fluid Mosaic Model & Permeability
The structure of the membrane (phospholipid bilayer) determines what can pass through it:
- Small, nonpolar molecules (e.g., $N2$, $O2$, $CO_2$) pass freely across the lipid bilayer.
- Hydrophilic substances (e.g., ions, polar molecules, water) cannot move through the hydrophobic core of the membrane without assistance.
- Transport Proteins allow the passage of hydrophilic substances.
Passive vs. Active Transport: An Overview
There are two main categories of transport based on energy requirements:
- Passive Transport: Movement of molecules from high concentration to low concentration. Does not require metabolic energy (ATP).
- Active Transport: Movement of molecules from low concentration to high concentration (against the gradient). Requires metabolic energy (ATP).
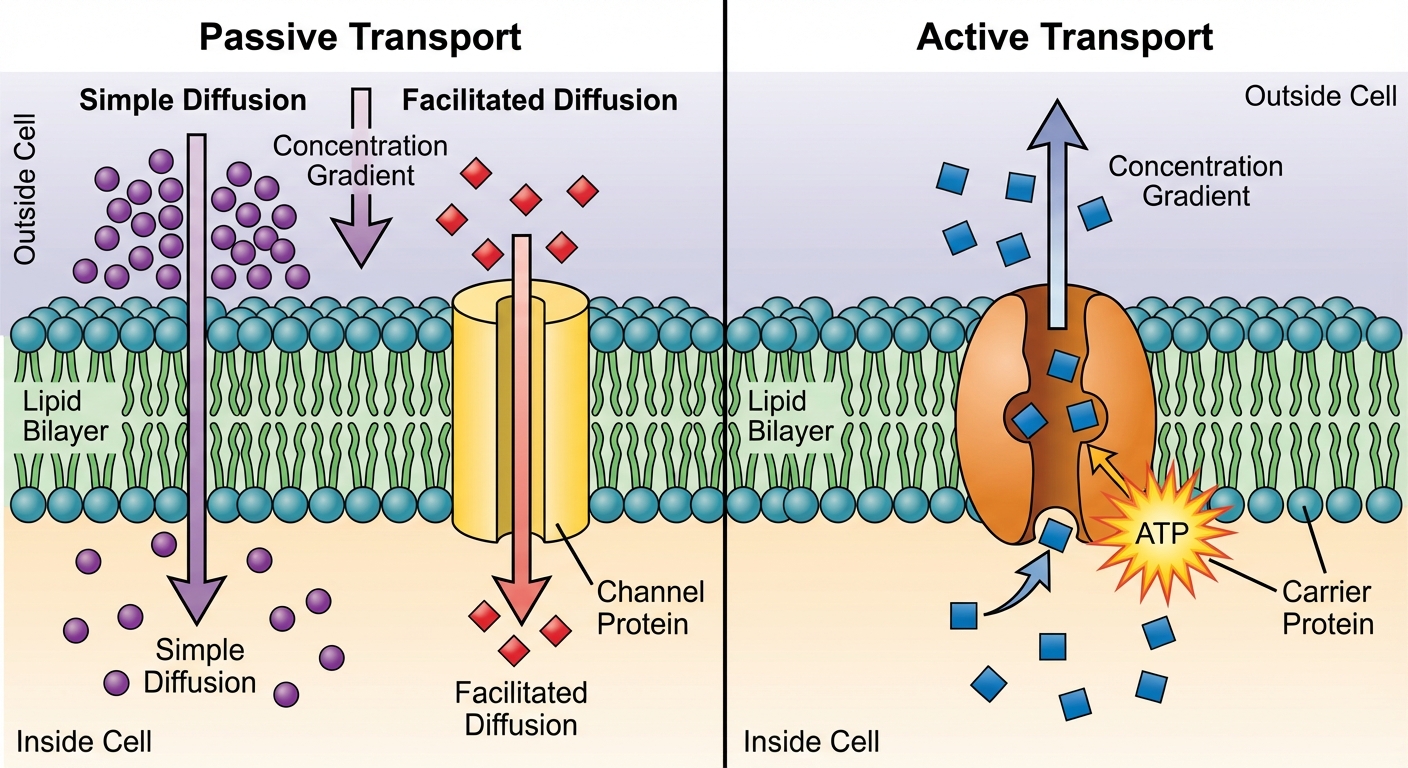
Passive Transport: Diffusion & Facilitated Diffusion
Simple Diffusion
Diffusion is the spontaneous movement of particles of any substance so that they spread out into the available space. A substance diffuses down its concentration gradient.
- Dynamic Equilibrium: Molecules cross the membrane in one direction and the other at roughly equal rates; there is no net change in concentration.
Facilitated Diffusion
For molecules that cannot pass the hydrophobic membrane core, transport proteins speed up the passive movement. There are two types of transport proteins:
- Channel Proteins: Provide a hydrophilic corridor.
- Example: Aquaporins facilitate the massive diffusion of water (osmosis) in kidney and plant cells.
- Example: Ion Channels (gated channels) open or close in response to a stimulus (electrical or chemical).
- Carrier Proteins: Undergo a subtle change in shape that translocates the solute-binding site across the membrane.
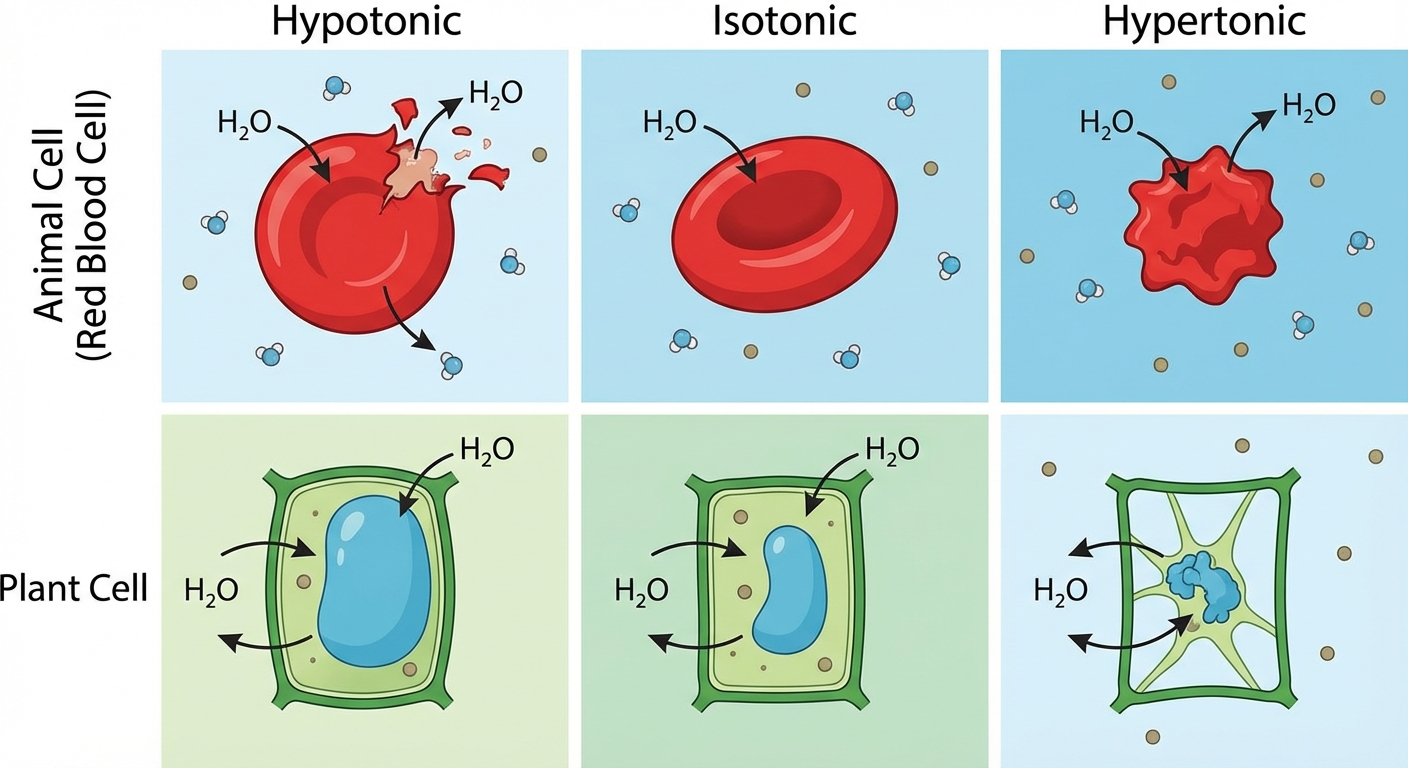
Tonicity and Osmoregulation
Osmosis is the diffusion of free water across a selectively permeable membrane. Water moves from an area of higher free water concentration (lower solute concentration) to an area of lower free water concentration (higher solute concentration).
Understanding Tonicity
Tonicity refers to the ability of a surrounding solution to cause a cell to gain or lose water. These terms are relative comparisons between the environment and the cell.
| Solution Type | Definition | Effect on Animal Cell | Effect on Plant Cell |
|---|---|---|---|
| Hypotonic | Solute concentration is less than inside the cell. | Lysed (Bursts) | Turgid (Normal) |
| Isotonic | Solute concentration is the same as inside the cell. | Normal | Flaccid |
| Hypertonic | Solute concentration is greater than inside the cell. | Shriveled | Plasmolyzed |
Water Potential ($\Psi$)
In AP Biology, you must quantify the movement of water. Water always moves from High Water Potential $\to$ Low Water Potential.
The general formula is:
Where:
- $\Psi$ = Water Potential
- $\Psi_P$ = Pressure Potential (physical pressure on turbidity; usually positive in living plant cells, 0 in open containers)
- $\Psi_S$ = Solute (Osmotic) Potential
Calculating Solute Potential
The formula for solute potential is:
- $i$ = Ionization Constant (number of particles the molecule breaks into. Sucrose = 1, NaCl = 2)
- $C$ = Molar Concentration
- $R$ = Pressure Constant ($0.0831 \text{ liter bars/mole K}$)
- $T$ = Temperature in Kelvin ($^\circ C + 273$)
Key Rule: $\Psi_S$ is always negative (or zero for pure water). Adding solutes lowers water potential.
Mechanisms of Active Transport
Active transport allows cells to maintain concentration gradients that differ from their surroundings (e.g., high $K^+$ and low $Na^+$ inside animal cells).
Primary Active Transport
This directly uses ATP to power the transport.
- The Sodium-Potassium Pump ($Na^+/K^+$ Pump): The major electrogenic pump of animal cells.
- Mechanism: Pumps 3 $Na^+$ out of the cell and 2 $K^+$ into the cell for every 1 ATP molecule consumed.
- Result: Creates a net negative charge inside the cell (membrane potential) and stores energy for other transport work.
Cotransport (Secondary Active Transport)
A transport protein couples the "downhill" diffusion of the solute to the "uphill" transport of a second substance against its own concentration gradient.
- Example: Sucrose-$H^+$ Cotransporter. Plants use an active $H^+$ pump to generate an electrochemical gradient. As $H^+$ diffuses back into the cell (passive), it carries sucrose with it (active) against the sucrose gradient.
Bulk Transport
Large molecules (proteins, polysaccharides) generally cross the membrane in bulk, packaged in vesicles. This requires energy.
Exocytosis
The cell secretes certain biological molecules by the fusion of vesicles with the plasma membrane.
- Example: Pancreas cells secreting insulin.
- Example: Neurons releasing neurotransmitters.
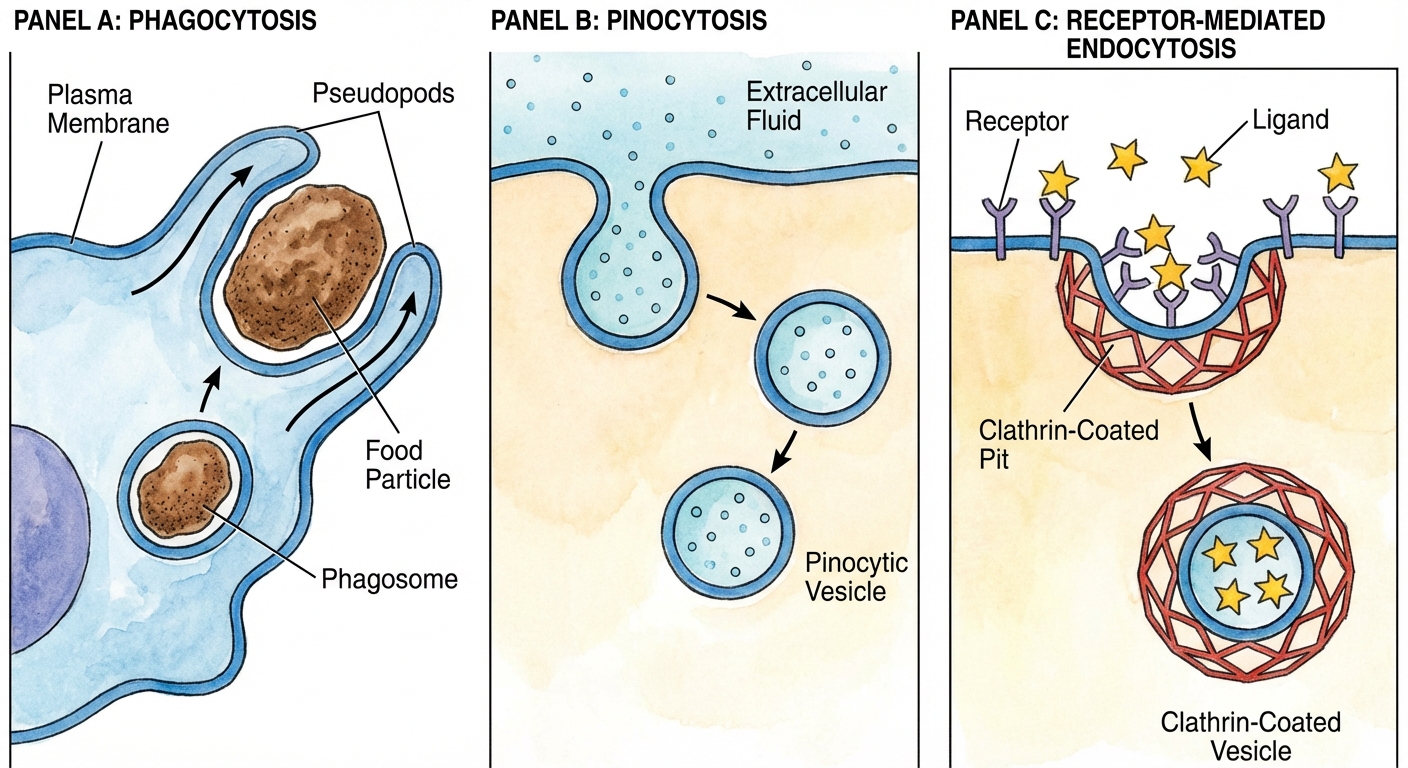
Endocytosis
The cell takes in biological molecules and particulate matter by forming new vesicles from the plasma membrane.
- Phagocytosis ("Cell Eating"): A cell engulfs a particle in a vacuole. The vacuole fuses with a lysosome to digest the particle.
- Pinocytosis ("Cell Drinking"): Molecules are taken up when extracellular fluid is "gulped" into tiny vesicles.
- Receptor-Mediated Endocytosis: Binding of specific solutes to receptors triggers vesicle formation (e.g., cholesterol uptake).
Common Mistakes & Pitfalls
- Water Potential Math: Students often forget the negative sign in the solute potential formula ($-iCRT$). Remember: adding salt makes the number more negative.
- Hypertonic vs. Hypotonic: These are confusing terms. Use the mnemonic: "Hypo acts like a Hippo"—in a hypotonic solution, an animal cell swells up big like a hippo.
- Active Transport Direction: Don't confuse the direction. Active means pushing a boulder up a hill (Low to High). Passive means the boulder rolling down the hill (High to Low).
- $Na^+/K^+$ Pump Ratio: Remember 3-2-1. 3 $Na^+$ out, 2 $K^+$ in, 1 ATP used. A helpful trick: "Salty Banana"—Cells are like salty bananas ($Na^+$ on the outside, $K^+$ on the inside).