AP Chemistry Unit 5: Kinetics — The Mathematics of Moving Molecules
Defining Reaction Rates
Before diving into complex calculations, we must define what we actually mean by "kinetics." While thermodynamics tells us if a reaction will happen (spontaneity), Kinetics tells us how fast it happens and the molecular pathway it takes.
The Definition of Rate
In chemistry, the reaction rate is the change in concentration of a reactant or product per unit of time. Generally, the units are Molarity per second ($M/s$ or $mol/L\cdot s$).
Mathematically, the average rate is expressed as:
There is a crucial convention here: Reaction rates are always reported as positive values.
- Reactants are consumed, so their concentration decreases (negative $\Delta$). We place a negative sign in front of the expression to make the rate positive.
- Products are formed, so their concentration increases (positive $\Delta$).
Stoichiometry and Relative Rates
Not all species in a reaction change at the same speed. Consider the combustion of propane:
For every 1 molecule of propane that burns, 5 molecules of oxygen are consumed. Therefore, oxygen disappears 5 times faster than propane. To equate the rates of the entire reaction, we divide by the stoichiometric coefficient.
For a general reaction $aA + bB \rightarrow cC + dD$:
Example 1: Using Relative Rates
If $O2$ is being consumed at $0.050\; M/s$ in the propane reaction above, what is the rate of $CO2$ production?
- Set up the equality: $-\frac{1}{5}\frac{\Delta[O2]}{\Delta t} = \frac{1}{3}\frac{\Delta[CO2]}{\Delta t}$
- Plug in the rate of $O2$ (remembering consumption is technically negative change, but treating the rate value as scalar magnitude simplifies this if you stick to the equality): $\frac{1}{5}(0.050) = \frac{1}{3}(Rate{CO_2})$
- Solve: $Rate{CO2} = \frac{3}{5}(0.050) = 0.030\; M/s$
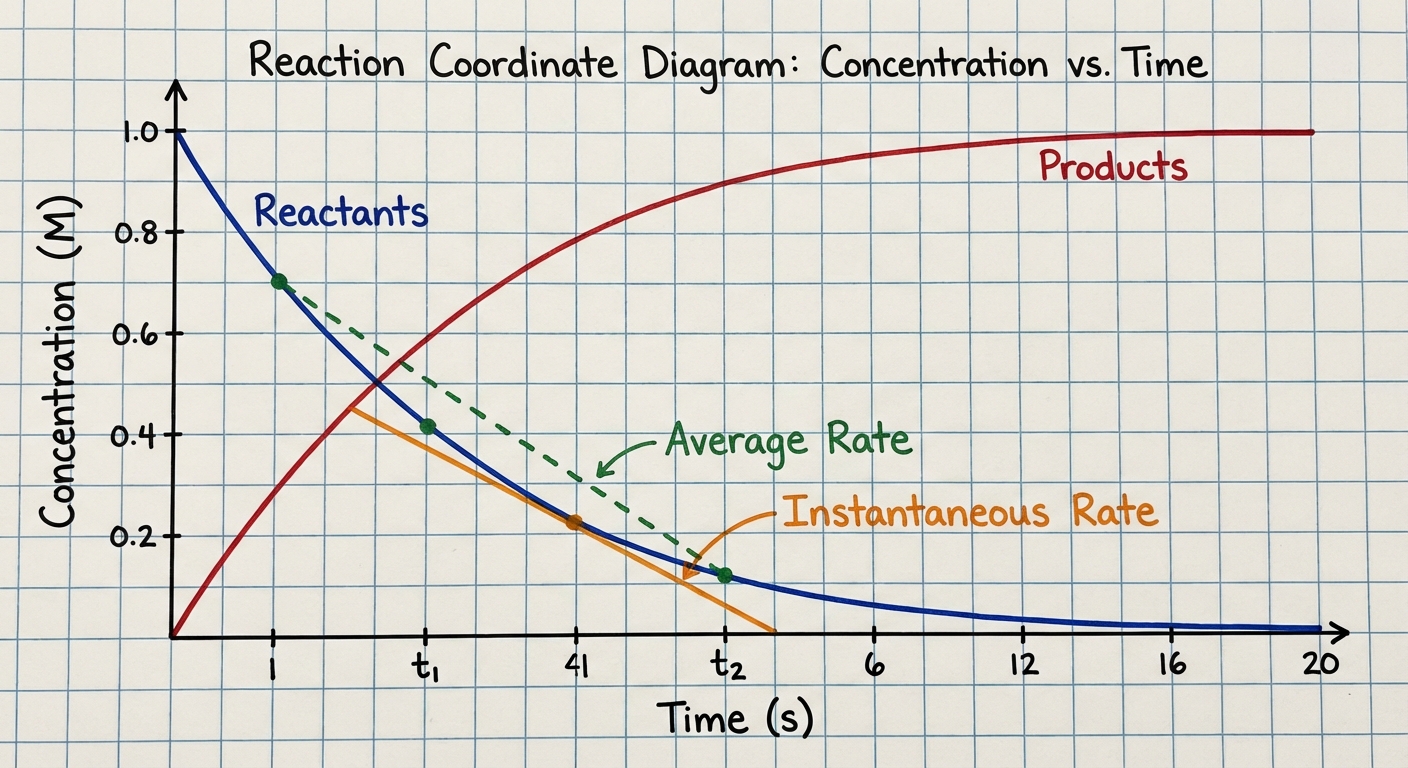
Instantaneous vs. Average Rate
- Average Rate: Calculated over a specific time interval (the slope of the secant line between two points on a graph).
- Instantaneous Rate: The rate at a specific moment in time (the slope of the tangent line to the curve at that point).
The Differential Rate Law
The Differential Rate Law (often just called the "Rate Law") relates the rate of the reaction to the concentration of the reactants. It answers the question: "If I double the concentration of Reactant A, how much faster does the reaction go?"
Structure of the Rate Law:
- $k$: Rate constant (temperature dependent).
- $[A], [B]$: Molar concentrations of reactants.
- $m, n$: Reaction orders (determined experimentally, NOT from coefficients).
Reaction Orders
The "order" describes the mathematical power dependence of the rate on concentration.
| Order | Effect of Doubling Concentration ($[A] \times 2$) | Mathematical relationship |
|---|---|---|
| Zero Order | Rate stays the same ($2^0 = 1$) | Rate $\propto [A]^0$ |
| First Order | Rate doubles ($2^1 = 2$) | Rate $\propto [A]^1$ |
| Second Order | Rate quadruples ($2^2 = 4$) | Rate $\propto [A]^2$ |
The Overall Order of the reaction is the sum of the individual orders: $Order_{overall} = m + n$.
The Method of Initial Rates
This is the standard AP Chemistry FRQ method for determining rate laws. You are given a table of experiments where initial concentrations vary.
Example Strategy:
To find the order with respect to Reactant A, look for two experiments where:
- $[B]$ is held constant.
- $[A]$ changes.
- Compare the ratio of rates to the ratio of concentrations.
The Units of $k$ (The "Rate Constant")
The units of $k$ change depending on the overall order of the reaction. This is a common multiple-choice question.
The Trick: The units of $k$ are always $M^{1-overall} \cdot s^{-1}$.
- Zero Order ($Rate = k$): $M/s$
- First Order ($Rate = k[A]$): $s^{-1}$ or $1/s$
- Second Order ($Rate = k[A]^2$): $M^{-1}s^{-1}$ or $1/(M\cdot s)$
- Third Order: $M^{-2}s^{-1}$
Concentration Changes Over Time (Integrated Rate Laws)
While the Differential Rate Law focuses on Rate vs. Concentration, the Integrated Rate Law focuses on Concentration vs. Time. These equations allow you to calculate how much reactant is left after a certain time period.
Summary of Integrated Rate Laws
The AP Exam requires you to identify the reaction order by looking at which graph creates a straight line.
| Order | Integrated Rate Law | Straight Line Plot ($y=mx+b$) | Linear Slope ($m$) | Half-Life ($t_{1/2}$) |
|---|---|---|---|---|
| Zero | $[A]t - [A]0 = -kt$ | $[A]$ vs. $t$ | $-k$ | $\frac{[A]_0}{2k}$ |
| First | $\ln[A]t - \ln[A]0 = -kt$ | $\ln[A]$ vs. $t$ | $-k$ | $\frac{0.693}{k}$ |
| Second | $\frac{1}{[A]t} - \frac{1}{[A]0} = kt$ | $\frac{1}{[A]}$ vs. $t$ | $+k$ | $\frac{1}{k[A]_0}$ |
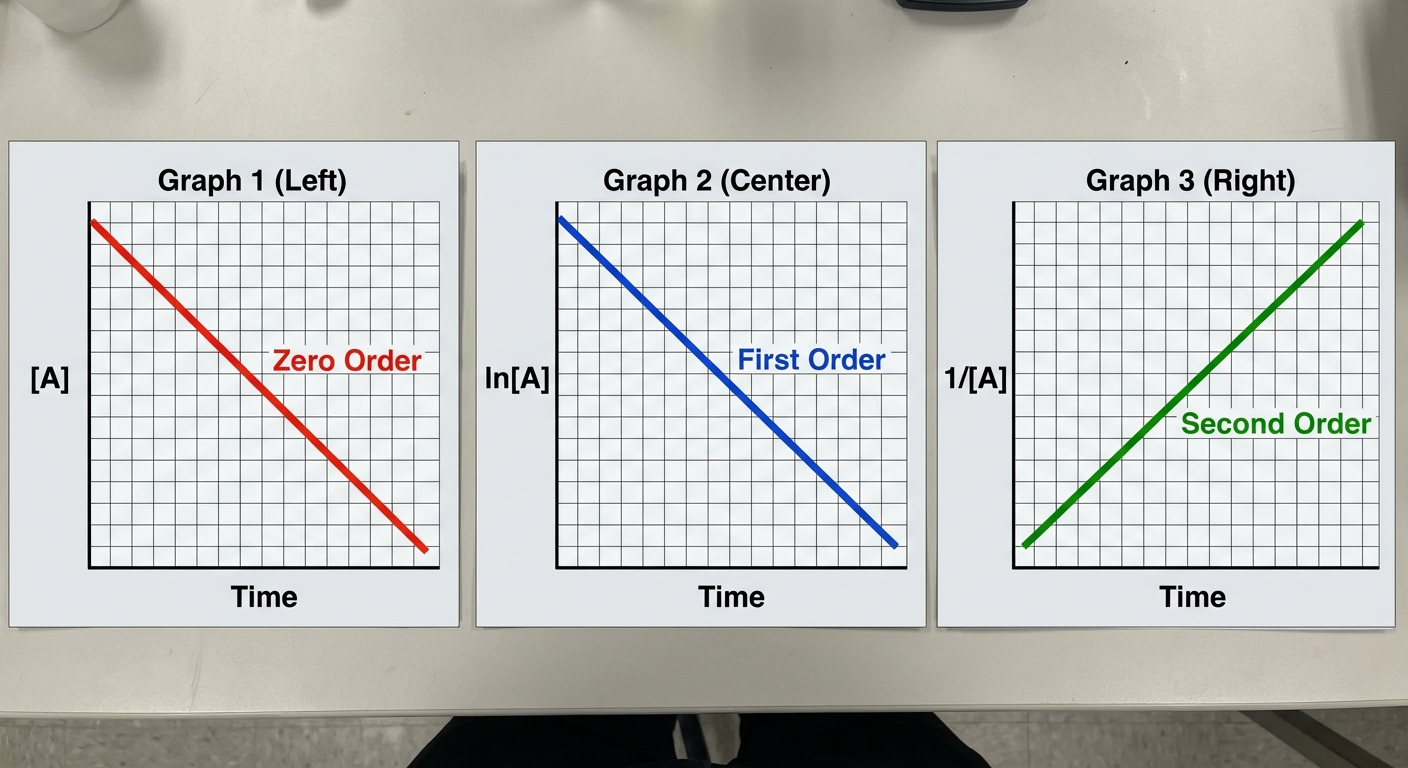
Key Takeaways for Each Order
- Zero Order: The rate is constant. The concentration drops linearly like a straight line down a hill. Common in enzyme-catalyzed reactions where the enzyme is saturated.
- First Order: Radioactive decay is always first order. Notice the half-life equation ($t_{1/2} = 0.693/k$) does not contain concentration. The half-life is constant regardless of how much you start with.
- Second Order: The graph of $1/[A]$ vs. time has a positive slope. This is the only one of the three standard graphs that goes up.
Common Mistakes & Pitfalls
- Confusing Coefficients with Exponents: Never assume the stoichiometric coefficients ($2A + B…$) are the exponents in the rate law ($Rate = k[A]^2[B]^1$). This is only true for elementary steps in a mechanism. For a general reaction, orders must be determined experimentally.
- Units of $k$: Students frequently lose points for incorrect units on $k$. Always check the overall order. If it's overall 2nd order, the units must be $M^{-1}s^{-1}$.
- Intermediate vs. Initial Rate: When using the Method of Initial Rates, ensure you are using values from the very start of the reaction. As the reaction proceeds, the reverse reaction might become significant, complicating the math.
- Graph Identification: Remember the axes!
- Is it $[A]$ vs time? (Straight = Zero Order)
- Is it $\ln[A]$ vs time? (Straight = First Order)
- Is it $1/[A]$ vs time? (Straight = Second Order)