AP Environmental Science: Atmospheric Dynamics & Pollution
Fundamentals of Air Pollution
Air Pollution is defined as the introduction of chemicals, particulate matter, or biological materials that cause harm or discomfort to humans or other living organisms, or damage the natural environment, into the atmosphere.
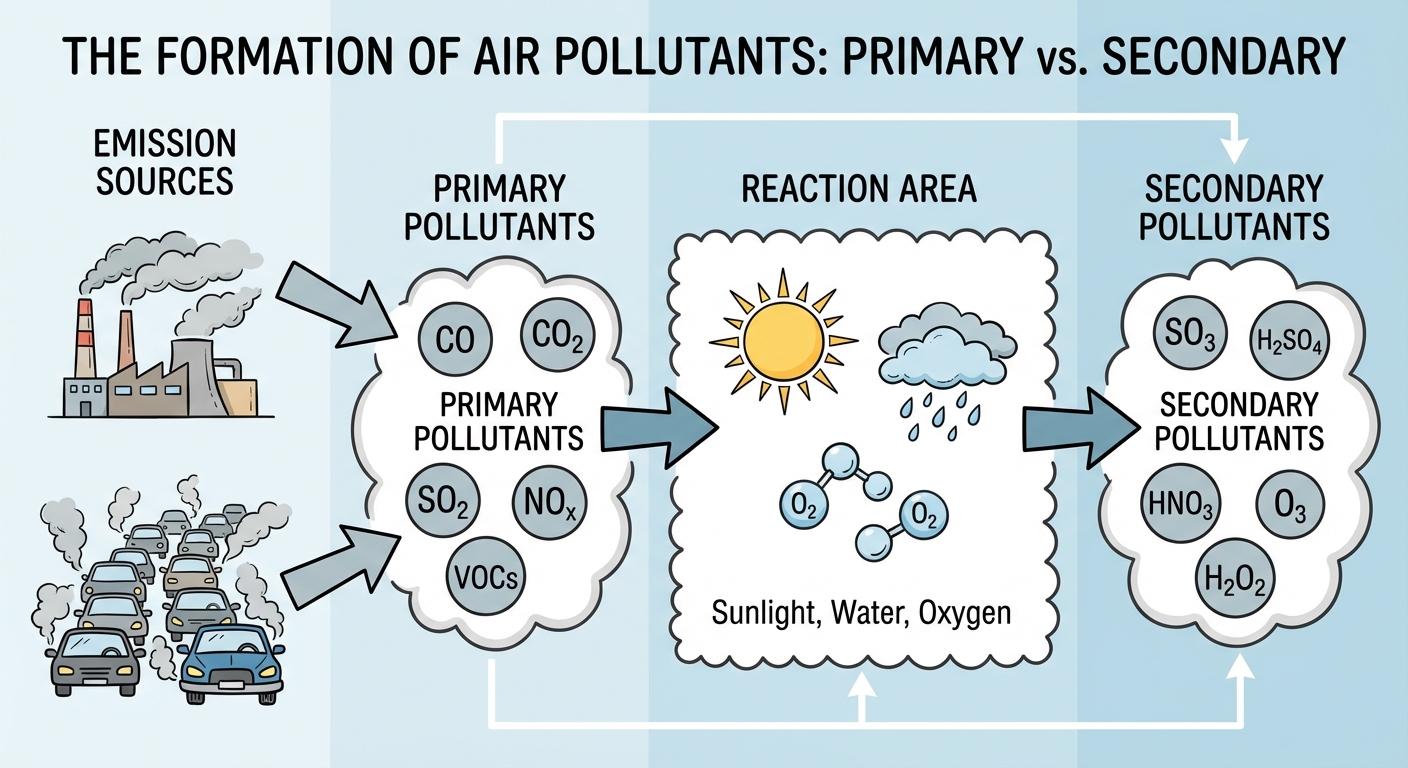
Primary vs. Secondary Pollutants
Understanding the genealogy of a pollutant is critical for the AP exam. Pollutants are classified based on how they originate.
- Primary Pollutants: Compounds coming directly out of a smokestack, exhaust pipe, or natural emission source.
- Examples: Carbon Monoxide ($CO$), Nitrogen Oxides ($NOx$), Sulfur Dioxide ($SO2$), Particulate Matter ($PM$), and Volatile Organic Compounds (VOCs).
- Secondary Pollutants: Primary pollutants that have undergone transformation in the presence of sunlight, water, oxygen, or other compounds.
- Examples: Ozone ($O3$), Sulfuric Acid ($H2SO4$), Nitric Acid ($HNO3$), and Peroxyacyl Nitrates (PANs).
The Clean Air Act and Criteria Pollutants
The Clean Air Act (CAA) requires the EPA to set National Ambient Air Quality Standards (NAAQS) for six criteria pollutants. A helpful mnemonic to remember them is SPLONC:
- Sulfur Dioxide ($SO_2$): From coal combustion; leads to acid rain.
- Particulate Matter ($PM$): Dust, smoke, ash; respiratory irritant.
- Lead ($Pb$): From old gasoline/paint; neurotoxin.
- Ozone ($O_3$): Secondary pollutant; respiratory irritant.
- Nitrogen Dioxide ($NO_2$): From engine combustion; leads to smog and acid rain.
- Carbon Monoxide ($CO$): Incomplete combustion; interferes with oxygen transport in blood.
Note: Carbon Dioxide ($CO_2$) is not one of the original six criteria pollutants under the CAA, although it is now regulated as a greenhouse gas.
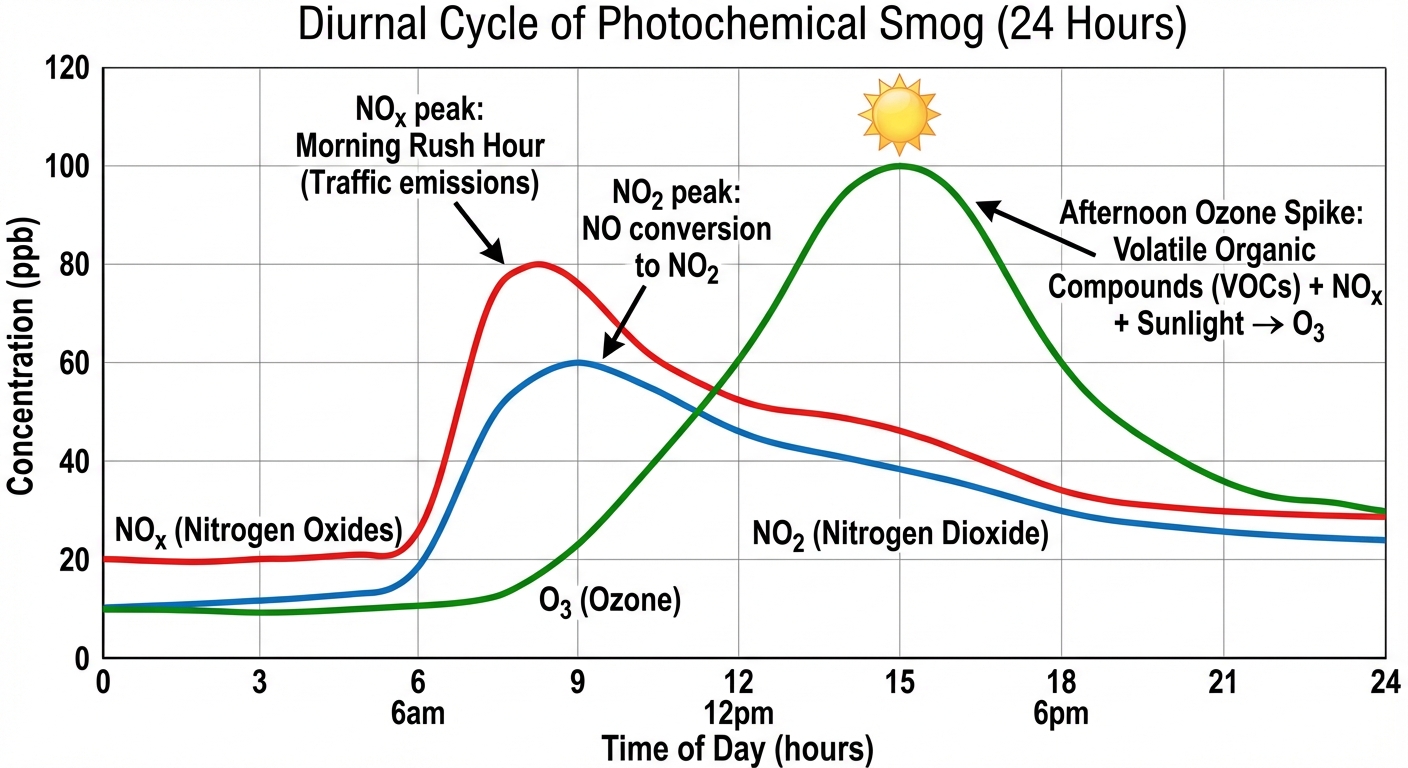
Photochemical Smog
Often called "L.A. Smog" or "Brown Smog," photochemical smog occurs when nitrogen oxides and volatile organic compounds react with heat and sunlight to produce a variety of pollutants.
The Ingredients
- NOx ($NO$ and $NO_2$): Emitted primarily by motor vehicles early in the day.
- VOCs (Volatile Organic Compounds): Organic chemicals that evaporate easily at room temperature. Sources include gasoline, formaldehyde, coniferous trees, and paints.
- Sunlight: The catalyst that drives the reaction.
The Chemical Formation
The formation of smog follows a strict diurnal (daily) cycle.
- Morning: Traffic increases $NO$ and VOC concentrations.
- Mid-day: Sunlight breaks down $NO2$ releasing a free oxygen atom:
This free oxygen atom ($O$) combines with atmospheric oxygen ($O2$) to form Ozone ($O3$): - Afternoon: Normally, ozone reacts with $NO$ to break back down when sunlight fades. However, if VOCs are present, they bind with the $NO$, preventing the ozone breakdown. This leads to a buildup of $O_3$ and the formation of PANs (Peroxyacyl Nitrates).
Environmental & Health Effects
- Respiratory Issues: Asthma, bronchitis, and reduced lung function.
- Plant Damage: Ozone enters stomata, oxidizing plant tissue and inhibiting photosynthesis.
- Economic Loss: Reduced crop yields and visual impairment (haze) reducing tourism.
Thermal Inversion
Under normal calculation conditions, air temperature decreases as altitude increases (the adiabatic lapse rate). Warm air near the surface rises, dispersing pollutants into the upper atmosphere. A Thermal Inversion disrupts this natural convection.
The Mechanism
A thermal inversion occurs when a layer of warm air settles over a layer of cooler air near the ground. Because cool air is denser than warm air, the cool pocket near the surface—containing pollution—cannot rise and mix.
Analogy: Think of a thermal inversion as a "lid" or a "cap" placed on a pot. The pollutants are the steam that cannot escape.
Factors Enhancing Inversion
- Geography: Valleys are most susceptible (e.g., Los Angeles, Mexico City, Santiago) because mountains block horizontal airflow.
- Weather: Clear, calm nights allow the ground to cool rapidly, cooling the air directly above it while the air higher up remains warm.
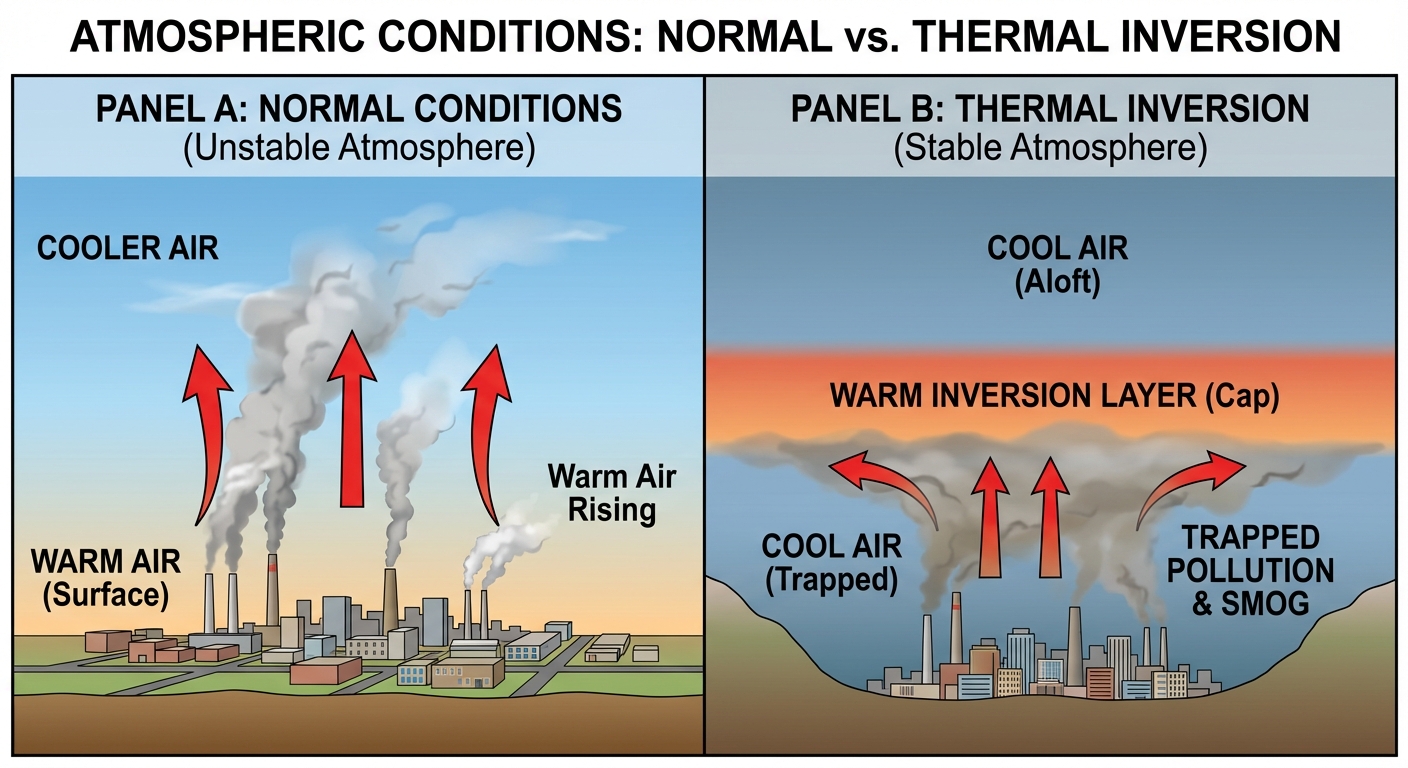
Comparison Table: Normal vs. Inversion
| Condition | Temperature Gradient (Surface $\to$ High) | Dispersal of Pollutants |
|---|---|---|
| Normal | Warm $\to$ Cool | Rapid mixing (Vertical) |
| Inversion | Cool $\to$ Warm $\to$ Cool | Trapped near surface (Stagnant) |
Atmospheric CO2 and Particulates
While chemical reactions create smog, physical accumulation of gas and particles poses distinct risks.
Particulate Matter (PM)
PM refers to solid or liquid particles suspended in the air.
- PM$_{10}$: Particles smaller than 10 micrometers (dust, pollen, mold). These are often trapped by nose hairs and throat mucus but irritate the upper respiratory tract.
- PM$_{2.5}$: Particles smaller than 2.5 micrometers (combustion particles, organic compounds, metals). These are more dangerous as they can penetrate deep into the lung tissue (alveoli) and enter the bloodstream.
Sources:
- Natural: Volcanoes, forest fires, dust storms, sea spray.
- Anthropogenic: Coal combustion, diesel burning, road construction, agriculture.
Atmospheric Carbon Dioxide ($CO_2$)
While often discussed in the context of Climate Change, $CO_2$ is a pollutant in high concentrations due to its toxicity and environmental displacement.
- Natural Sources:
- Respiration: Animals exhaling.
- Decomposition: Breakdown of organic matter.
- Volcanic Eruptions: Release of stored carbon from geosphere.
- Anthropogenic Sources:
- Fossil Fuel Combustion: Coal, oil, natural gas.
- Deforestation: Reduces the biosphere's ability to sequester carbon.
Study Tip: $CO2$ levels fluctuate seasonally. In the Northern Hemisphere spring/summer, photosynthesis increases, pulling $CO2$ down. In fall/winter, leaves decay and photosynthesis drops, raising $CO_2$. This creates the "sawtooth" pattern seen on the Keeling Curve.
Common Mistakes & Pitfalls
Even prepared students often confuse specific atmospheric chemistry concepts. Avoid these errors:
Stratospheric vs. Tropospheric Ozone:
- Mistake: Thinking all ozone is "bad" or created by pollution.
- Correction: Remember the rhyme "Good up high, bad nearby." Stratospheric ozone (natural) blocks UV radiation. Tropospheric ozone (anthropogenic smog) damages lungs.
Ozone Depletion vs. Climate Change:
- Mistake: Suggesting CFCs cause global warming by breaking the ozone layer, which lets in more heat.
- Correction: These are two separate issues. The ozone hole lets in UV radiation (cancer risk), not thermal heat. Greenhouse gases (like CO2 and Methane) trap infrared heat. Do not conflate them.
Photochemical vs. Industrial Smog:
- Mistake: Using the terms interchangeably.
- Correction: Industrial Smog (Grey Smog) is sulfur-based ($SO2$) from coal burning. Photochemical Smog (Brown Smog) is nitrogen-based ($NOx$) from cars and sunlight.