AP Chemistry Unit 3: Physics and Chemistry of Gases
The Ideal Gas Law and Partial Pressures
Fundamental Concepts
The study of gases focuses on the relationships between four macroscopic variables: pressure ($P$), volume ($V$), temperature ($T$), and amount of substance in moles ($n$).
An Ideal Gas is a theoretical gas that fits specific criteria:
- The particles are point masses with negligible volume.
- There are no intermolecular forces (attraction or repulsion) between particles.
- Collisions are completely elastic (no loss of kinetic energy).
While no gas is truly "ideal," most gases behave ideally at low pressures and high temperatures.
The Ideal Gas Equation
The relationships established by Boyle, Charles, and Avogadro are combined into a single equation:
Where:
- $P$ = Pressure (atm, torr, mmHg, or Pa)
- $V$ = Volume (Liters)
- $n$ = Moles of gas
- $T$ = Temperature (Must be in Kelvin)
- $R$ = Ideal Gas Constant
Choosing the Correct R:
The value of $R$ depends on the pressure unit used. You must select the one matching your units from the AP formula sheet.
- If $P$ is in atm: $R = 0.08206 \text{ L atm mol}^{-1} \text{K}^{-1}$
- If $P$ is in torr/mmHg: $R = 62.36 \text{ L torr mol}^{-1} \text{K}^{-1}$
- (Used in energy calculations) $R = 8.314 \text{ J mol}^{-1} \text{K}^{-1}$
Dalton's Law of Partial Pressures
In a mixture of unreactive gases, the total pressure is simply the sum of the individual pressures that each gas would exert if it were alone in the container.
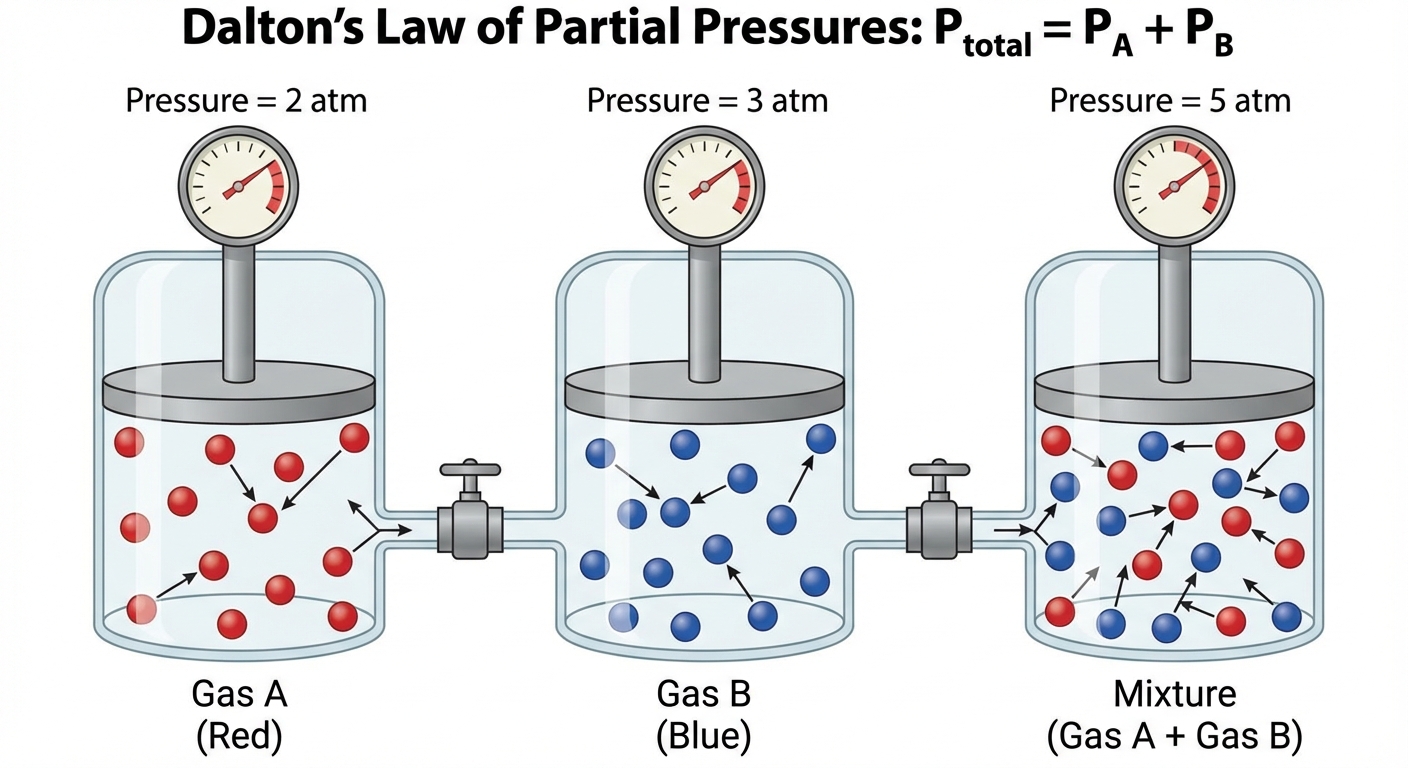
Mole Fraction ($\chi$):
Partial pressure is directly proportional to the number of moles. You can find the partial pressure of a specific gas ($PA$) if you know the total pressure ($P{total}$) and the mole fraction ($X_A$).
Kinetic Molecular Theory (KMT)
Postulates of KMT
The Kinetic Molecular Theory explains why gases follow the Ideal Gas Law based on particle behavior. The key postulates are:
- Random Motion: Gas particles are in constant, random, straight-line motion until they collide with walls or other particles.
- Negligible Volume: The combined volume of the actual gas particles is negligible compared to the total volume of the container (mostly empty space).
- No Forces: There are no attractive or repulsive forces between particles.
- Elastic Collisions: Collisions are perfectly elastic; energy is transferred but standard total kinetic energy is conserved.
- Temperature and KE: The average kinetic energy of the gas particles is directly proportional to the Kelvin temperature.
- Crucial Concept: At a given temperature, all gases in a mixture have the same average Kinetic Energy, regardless of their mass.
Particle Speed and Maxwell-Boltzmann Distributions
While average KE depends only on temperature, the speed of particles depends on both temperature and molar mass.
- $u_{rms}$: Root mean square speed
- $\mathcal{M}$: Molar mass (in kg/mol for this specific energy formula using $R=8.314$)
Interpreting Maxwell-Boltzmann Graphs:
These graphs plot the "Number of Particles" (y-axis) vs. "Particle Speed" (x-axis). 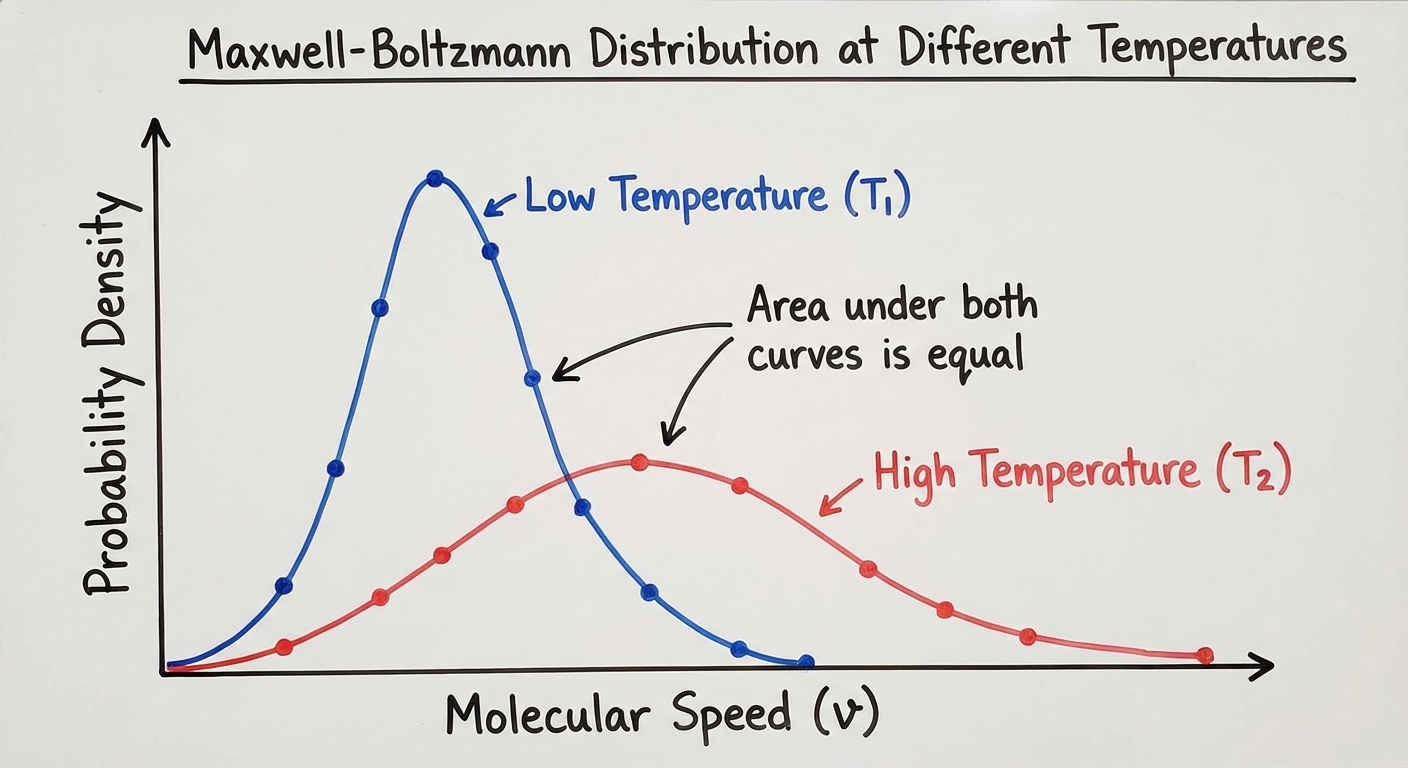
Two Main Trends:
- Effect of Temperature: As Temperature increases, the curve flattens and the peak shifts to the right (higher speed). A wider distribution means a larger range of speeds.
- Effect of Molar Mass: As Molar Mass decreases (lighter gas), the curve behaves like a hot gas—it flattens and shifts right. Lighter particles must move faster to have the same Kinetic Energy as heavy particles.
- Example: At 298K, Helium (light) moves much faster than Xenon (heavy).
Deviations from Ideal Gas Law
Real gases do not always follow $PV=nRT$. Deviations occur when the assumptions of KMT break down.
Conditions for Deviation
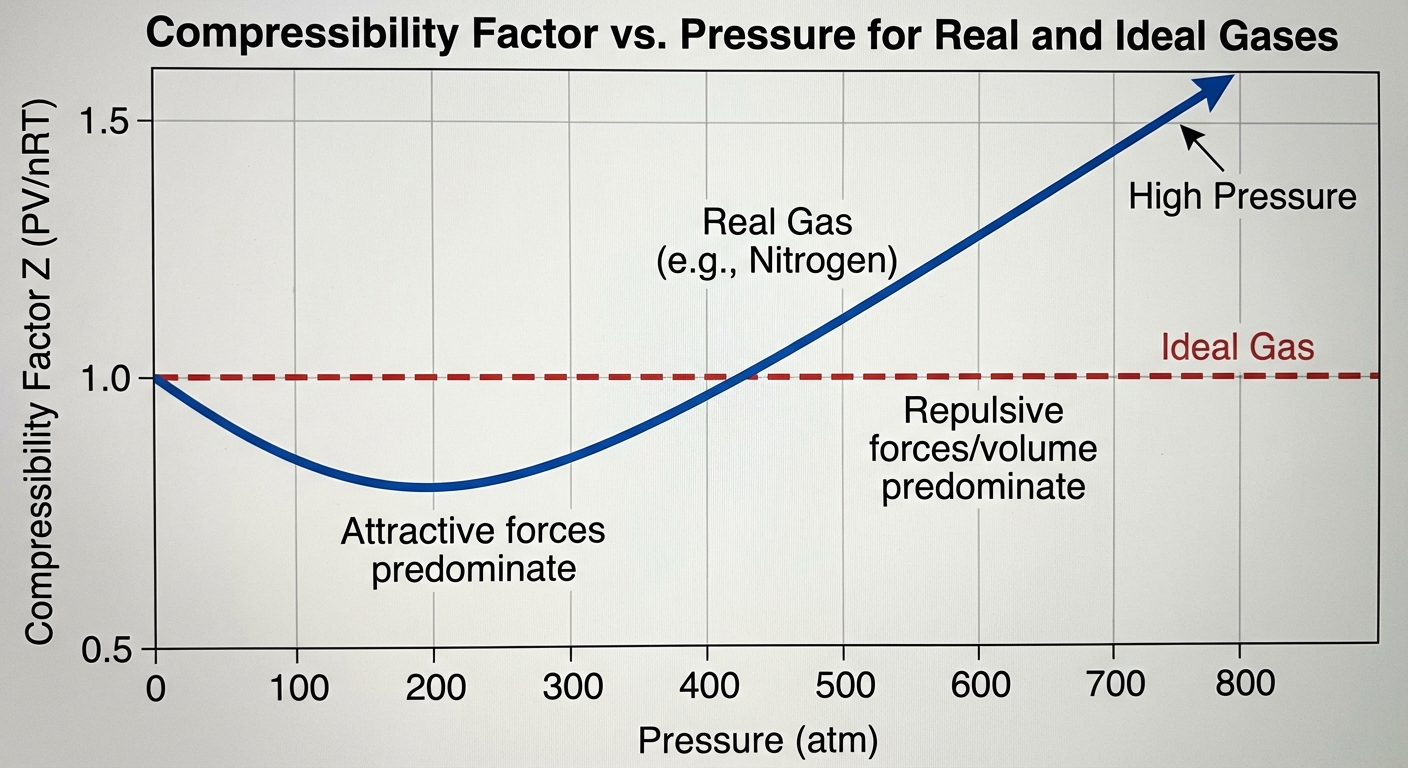
Ideally, we assume particles have no volume and no attraction. This assumption fails under:
- High Pressure: Particles are forced close together. The actual volume of the particles becomes significant compared to the container volume. Predictable result: $V{real} > V{ideal}$.
- Low Temperature: Particles move slowly. Because they have low kinetic energy, they cannot overcome intermolecular forces (IMFs) when they pass near each other. They "stick" slightly, reducing the force of collisions on the walls. Predictable result: $P{real} < P{ideal}$.
The Van der Waals Equation (Conceptual)
You rarely calculate with this on the AP exam, but you must understand the corrections:
| Correction Term | Purpose | Relationship |
|---|---|---|
| $+ \frac{n^2a}{V^2}$ | Corrects Pressure for IMFs | The constant $a$ relates to the stickiness/strength of IMFs. A gas with strong Hydrogen Bonding has a large $a$. |
| $- nb$ | Corrects Volume for particle size | The constant $b$ relates to molecular size. Large molecules have a large $b$. |
Deviation Plot
Consider a graph of $Z = \frac{PV}{nRT}$ vs Pressure.
- For an ideal gas, $Z = 1$ always.
- For real gases, calculating $Z$ usually results in a value $
Common Mistakes & Pitfalls
Celsius vs. Kelvin:
- Mistake: Plugging Celsius directly into $PV=nRT$.
- Correction: ALWAYS add 273 to Celsius. $0^\circ C$ is not zero energy; $0K$ is.
Confusing KE and Speed:
- Mistake: Thinking heavier gases have lower Kinetic Energy than lighter gases at the same temperature.
- Correction: At the same $T$, average KE is the same. Mass only affects speed (Heavy = Slow; Light = Fast).
Maxwell-Boltzmann Peak Height:
- Mistake: Drawing a high-temperature curve with a higher peak than a low-temperature curve.
- Correction: The area under the curve (total number of particles) must remain constant. If the curve stretches right (higher speed), it MUST get shorter (flatten).
Pressure Units for R:
- Mistake: Using $R = 8.314$ for a has pressure calculation in standard atmospheres.
- Correction: Check your units. $8.314$ involves Joules ($kg\cdot m^2/s^2$) and is usually for calculating speed ($u_{rms}$) or energy, not basic $PV=nRT$ unless utilizing Pascals/m³.