AP Chemistry Unit 3: Mechanics of Mixtures and Solubility
Understanding Solutions and Mixtures
To master Unit 3 in AP Chemistry, you must understand how matter interacts not just as pure substances, but as mixtures. At the highest level, a mixture describes a physical combination of two or more substances where the identity of each substance remains unchanged.
Mixtures fall into two primary categories:
- Heterogeneous Mixtures: Components are not distinct and not uniformly distributed (e.g., salad dressing, concrete).
- Homogeneous Mixtures (Solutions): Components are uniformly distributed at the microscopic level. In AP Chemistry, we focus heavily on these.
The Components of a Solution
A solution is composed of two distinct parts:
- Solvent: The substance present in the greatest abundance (usually water in aqueous solutions).
- Solute: The substance being dissolved.
Molarity (M)
The standard unit of concentration in chemistry is Molarity. It quantifies the number of solute particles in a specific volume of solution.
When calculating Molarity:
- [ ] Brackets (e.g., $[Na^+]$) denote molar concentration.
- The volume in the denominator is the total volume of the solution, not just the volume of the solvent added.
Representations of Solutions
The College Board frequently asks students to draw or analyze particulate diagrams (particle-level drawings). You must be able to visualize what happens when a solute dissolves in a solvent.
Liquid Solutions and Dissolution
When an ionic compound (a salt) dissolves in water, it undergoes dissociation. The crystal lattice breaks apart, and the individual ions are stabilized by water molecules. This stabilization is determined by Coulombic interactions between the charged ions and the polar water molecules.
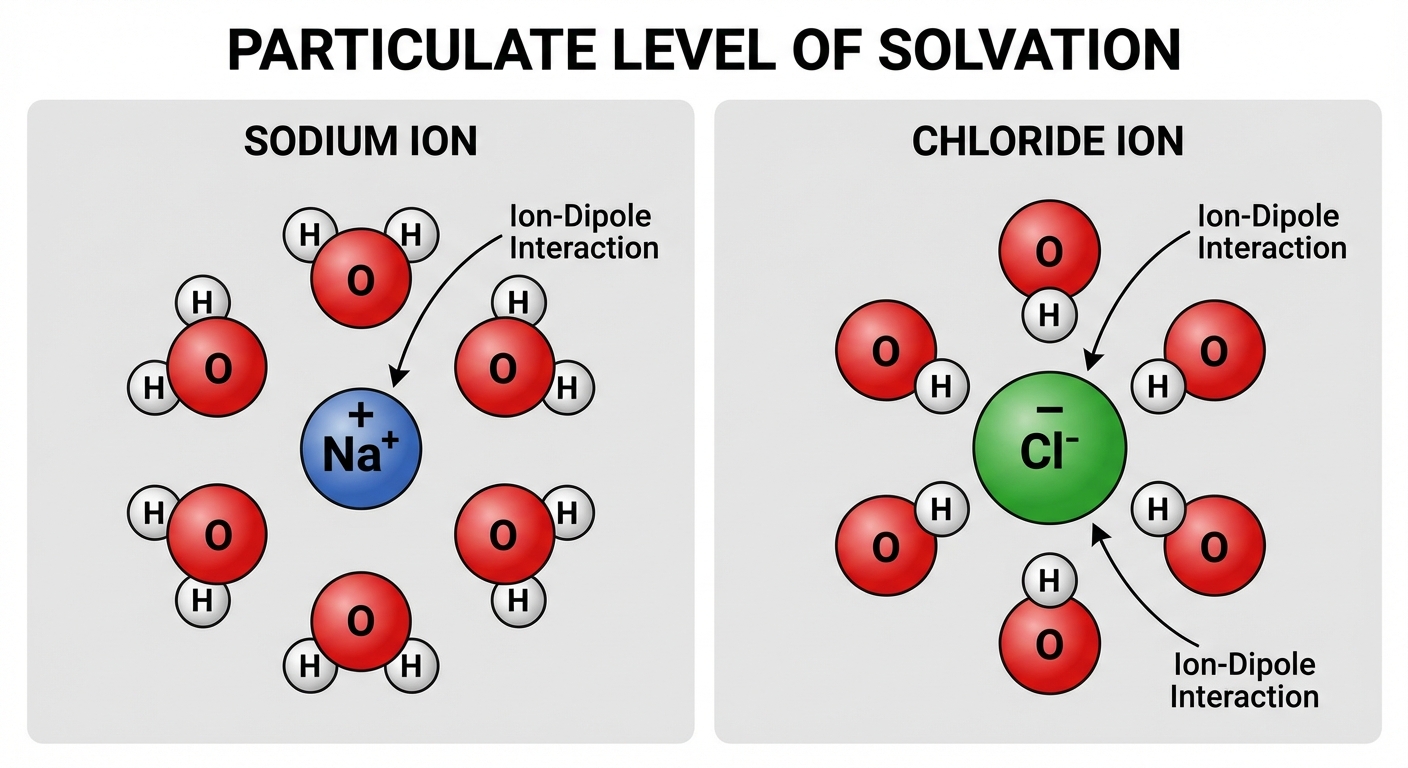
Key Rules for Drawing Hydration Shells
When drawing the solvatation (or hydration) of ions, orientation is critical:
- Cations (Positive Ions): The partially negative oxygen atom ($\delta-$) of the water molecule must face the cation.
- Anions (Negative Ions): The partially positive hydrogen atoms ($\delta+$) of the water molecule must face the anion.
- Scale: If the anion is significantly larger than the cation (e.g., $I^-$ vs $Li^+$), your drawing should reflect that size difference.
Note on Molecular Solutes:
Covalent compounds like sucrose (sugar) or ethanol generally do not dissociate into ions (unless they are acids). They remain as intact molecules surrounded by water.
Solubility and Intermolecular Forces
Why do some things dissolve while others settle at the bottom? The answer lies in the strength of Intermolecular Forces (IMFs).
The Golden Rule: "Like Dissolves Like"
This phrase is a simplification of energetic stability. Substances with similar intermolecular forces tend to be soluble in one another.
| Solute Type | Solvent Type | Result | Interaction Dominance |
|---|---|---|---|
| Polar | Polar | Soluble | Dipole-Dipole or H-bonding |
| Nonpolar | Nonpolar | Soluble | London Dispersion Forces |
| Polar | Nonpolar | Insoluble | The polar molecules prefer to stick to each other rather than mix with nonpolar molecules. |
| Ionic | Polar | Soluble | Ion-Dipole |
Miscibility
- Miscible: Two liquids that mix in all proportions (e.g., Ethanol and Water—both can Hydrogen bond).
- Immiscible: Two liquids that form distinct layers (e.g., Oil and Water—Oil is nonpolar, Water is polar).
Energetics of Solubility
For a solution to form, the energy released by the new solute-solvent interactions must be comparable to or greater than the energy required to break the original solute-solute and solvent-solvent interactions.
Separation of Solutions and Mixtures
Chemists separate mixtures by exploiting differences in the physical properties of their components.
Distillation
Distillation separates components based on differences in boiling points and vapor pressure.
- Process: The mixture is heated. The substance with the weaker IMFs (higher vapor pressure, lower boiling point) vaporizes first.
- Application: Separating alcohol from water, or crude oil refinement.
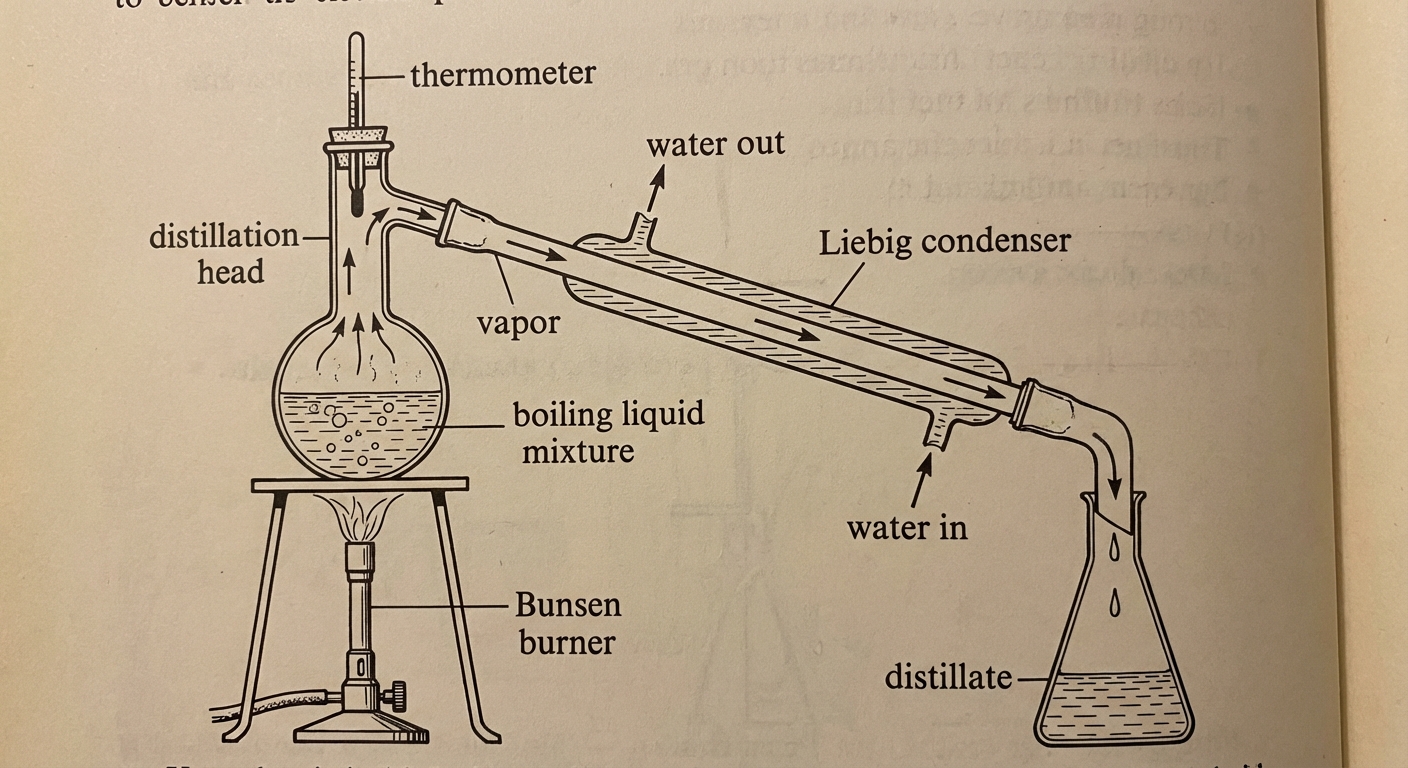
Chromatography
Chromatography separates components based on differences in particle polarity and their affinity for the stationary phase versus the mobile phase.
There are two main phases in Paper/Thin-Layer Chromatography (TLC):
- Mobile Phase: The solvent moving up the paper (the eluent).
- Stationary Phase: The paper or solid strip itself.
The Concept:
- If the component has strong IMFs with the mobile phase (solvent), it travels further up the paper.
- If the component has strong IMFs with the stationary phase, it stays lower on the paper and moves slower.
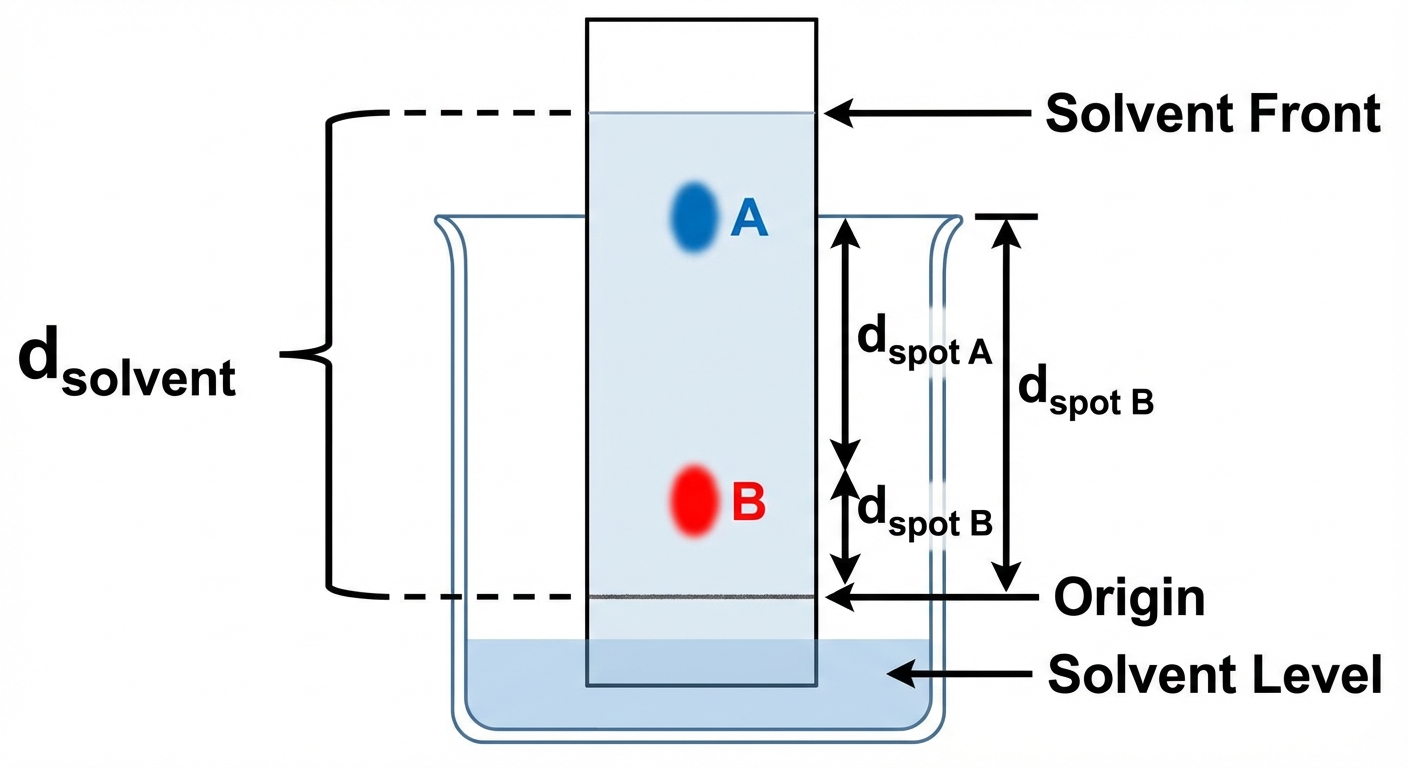
Retention Factor ($R_f$)
The distance traveled is quantified by the $R_f$ value:
- $R_f$ is always less than 1.
- A high $R_f$ indicates high attraction to the mobile phase.
Example Scenario
Imagine a nonpolar solvent is used on polar paper.
- Component A travels very high ($R_f = 0.9$). It prefers the solvent, so it must be nonpolar.
- Component B stays low ($R_f = 0.2$). It prefers the paper, so it must be polar.
Common Mistakes & Pitfalls
Molarity Volume Error:
- Mistake: Using the volume of the solvent in the denominator for Molarity.
- Correction: Always use the total volume of the solution. If you add water to salt, the volume changes slightly.
Water Orientation:
- Mistake: Drawing water molecules randomly around an ion.
- Correction: Check your dipoles! Oxygen faces cations; Hydrogen faces anions.
Dissociation vs. Decomposition:
- Mistake: Thinking polyatomic ions break apart when dissolving (e.g., thinking $NO_3^-$ becomes $N$ and $O$ atoms).
- Correction: Polyatomic ions stay intact. $NaNO3$ becomes $Na^+$ and $NO3^-$.
"Like Dissolves Like" as an Explanation:
- Mistake: Writing "It dissolves because like dissolves like" on an FRQ.
- Correction: This is a rule of thumb, not an explanation. You must reference the specific Intermolecular Forces involved (e.g., "Ethanol is soluble in water because both can form Hydrogen bonds, which are energetically favorable").