Strategies for Air Quality and Environmental Safety
Reducing Air Pollutants
Air pollution reduction relies on a combination of regulatory frameworks (like the Clean Air Act) and specific technologies designed to filter, neutralize, or capture pollutants before they enter the atmosphere. In AP Environmental Science, you must distinguish between regulatory "command-and-control" approaches and physical "end-of-pipe" technologies.
Technological Interventions
Technology is often applied at the source of emission, such as coal-burning power plants or automobile tailpipes.
1. Vapor Recovery Nozzles
This technology is primarily found at gasoline pumps. When you pump gas, volatile organic compounds (VOCs)—specifically benzene and other hydrocarbons—can escape into the atmosphere as fumes.
- Mechanism: A specialized nozzle captures these vapors and returns them to the underground storage tank.
- Benefit: Reduces the formation of photochemical smog (since VOCs are a precursor).
2. Catalytic Converters
Mandated on all automobiles in the US since 1975, catalytic converters utilize rare earth metals (Platinum, Palladium, Rhodium) to facilitate chemical reactions.
The Reaction Process:
It converts three harmful pollutants into less harmful compounds:
- Nitrogen Oxides ($NOx$) $\rightarrow$ Nitrogen Gas ($N2$) + Oxygen ($O_2$)
- Carbon Monoxide ($CO$) $\rightarrow$ Carbon Dioxide ($CO_2$)
- Hydrocarbons (VOCs) $\rightarrow$ Water ($H2O$) + Carbon Dioxide ($CO2$)
Note: While catalytic converters successfully reduce toxic emissions, they do produce $CO_2$, which is a greenhouse gas.
3. Scrubbers (Wet and Dry)
Scrubbers are large air pollution control devices used in industrial smokestacks (coal power plants) to remove particulates and/or gases.
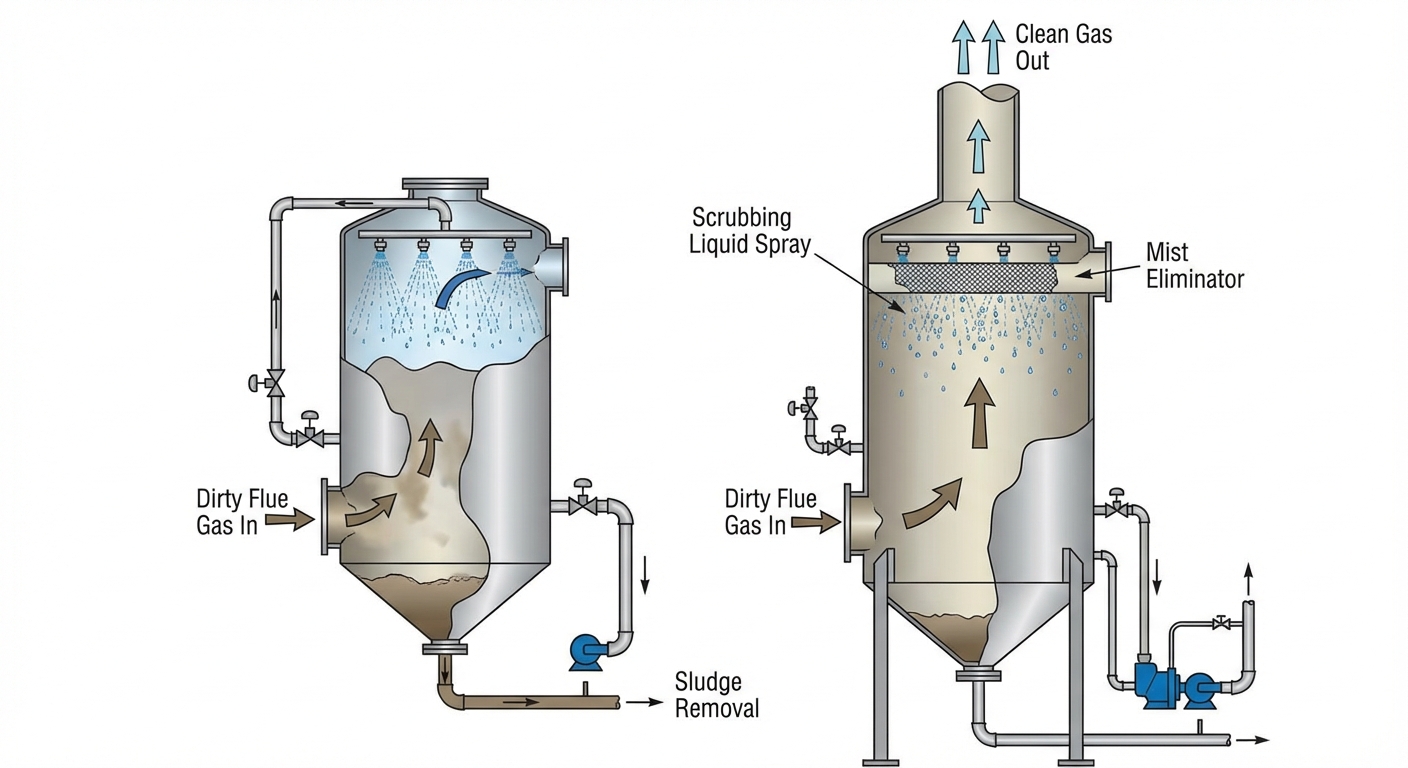
| Feature | Wet Scrubber | Dry Scrubber |
|---|---|---|
| Target Pollutants | Sulfur Oxides ($SO_x$), PM (Particulate Matter) | Primarily Acid Gases ($SO_x$, HCl) |
| Mechanism | Uses a liquid spray (mist) to wash pollutants out of the gas stream. The liquid often contains chemical neutralizing agents (like lime). | Uses dry chemical reagents (pulverized limestone) injected into dirty exhaust gases to neutralize them. |
| Byproduct | Wet toxic sludge (must be disposed of carefully). | Dry powder/waste. |
4. Electrostatic Precipitators
Used in coal-burning power plants to remove Particulate Matter (PM).
- Mechanism: Dirty gas enters a chamber containing negatively charged wires. The particulates pick up a negative charge and are attracted to positively charged collection plates.
- Efficiency: Very high (up to 99%) for removing fine dust and fly ash.
Legislation: The Clean Air Act (CAA)
The Clean Air Act (1970, 1990) is the primary U.S. federal law regulating air emissions. It established the National Ambient Air Quality Standards (NAAQS) for six "criteria pollutants."
Mnemonic: NOSCLP
To remember the six criteria pollutants regulated by the CAA, use the detailed acronym NOSCLP:
- N: Nitrogen Oxides ($NO_x$)
- O: Ozone ($O_3$) — specifically tropospheric (ground-level) ozone
- S: Sulfur Dioxide ($SO_2$)
- C: Carbon Monoxide ($CO$)
- L: Lead ($Pb$)
- P: Particulate Matter (PM)
Acid Deposition (Acid Rain)
Acid deposition refers to any form of precipitation (rain, snow, fog, hail) with acidic components, such as sulfuric or nitric acid. While normal rain is slightly acidic ($\text{pH} \approx 5.6$) due to atmospheric $CO_2$, acid rain typically has a pH below 5.6, often ranging from 4.0 to 4.5.
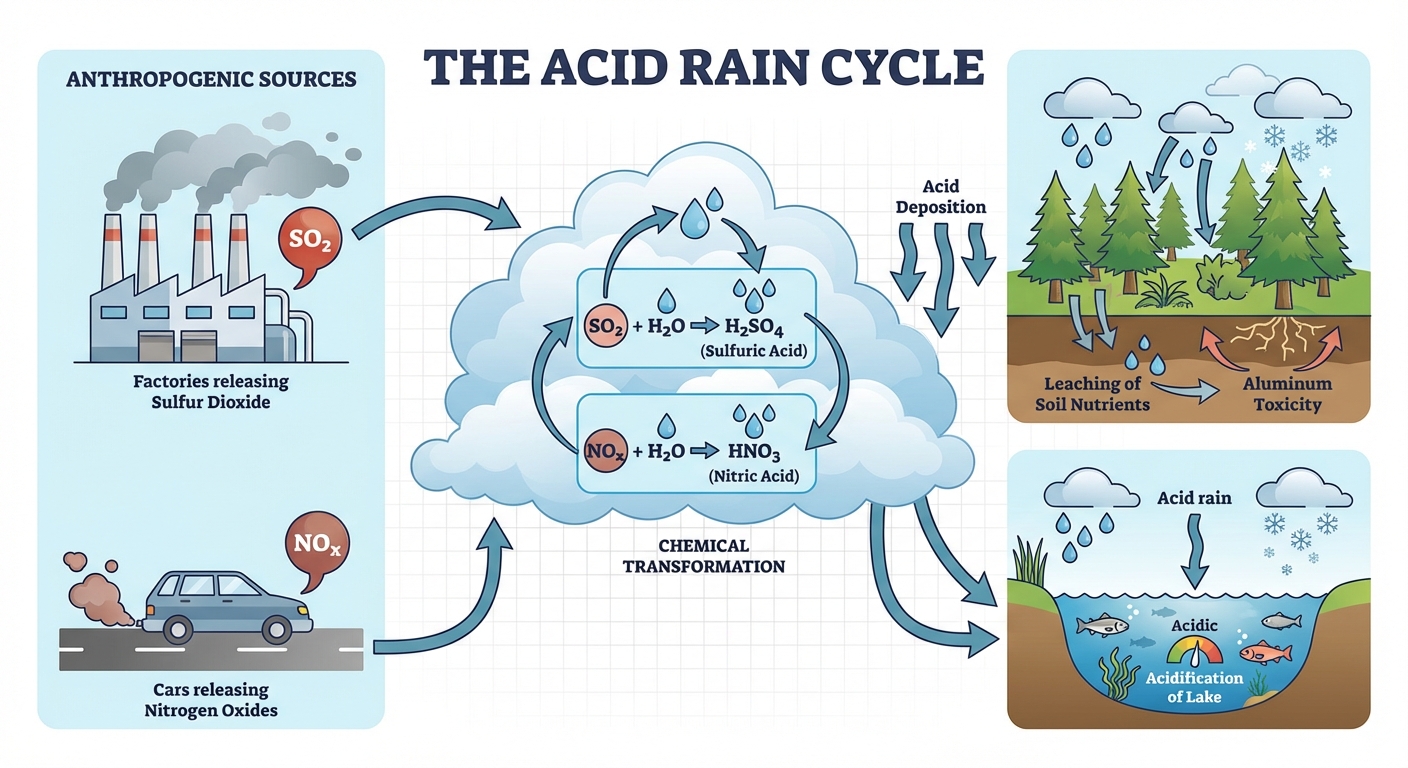
Formation Chemistry
Acid rain is a secondary pollutant formed from primary pollutants released by combustion.
Nitrogen Oxides: Released from vehicle exhaust and coal burning.
(Reacts with water vapor to form Nitric Acid)Sulfur Oxides: Released primarily from coal-burning power plants.
(Reacts with water/oxygen to form Sulfuric Acid)
Environmental Impacts
1. Soil and Geology
- Leaching of Nutrients: Acidic water dissolves positively charged soil nutrients like Calcium ($Ca^{2+}$) and Magnesium ($Mg^{2+}$), washing them out of the root zone.
- Aluminum Toxicity: Acid rain dissolves aluminum ions ($Al^{3+}$) typically bound to soil particles. Free aluminum is toxic to plant roots and prevents nutrient uptake.
2. Aquatic Ecosystems
- pH Tolerance: Most aquatic species have a specific pH range. If pH drops below 5.0, many fish species cannot reproduce.
- Aluminum Effect: Leached aluminum runs off into lakes and streams, accumulating in fish gills and causing suffocation by stimulating excess mucus production.
3. Anthropogenic Structures
- Acid rain degrades limestone and marble structures (calcium carbonate) through chemical weathering, damaging statues, bridges, and buildings.
Mitigation
- Liming: Adding crushed limestone (calcium carbonate) to acidified lakes or soils to neutralize the acid and raise the pH.
Noise Pollution
Noise pollution is sound at levels high enough to cause physiological stress and hearing loss. It works on a logarithmic scale (decibels, dB), meaning an increase of 10 dB represents a ten-fold increase in sound intensity.
Sources
- Transportation: Cars, trucks, trains, airplanes.
- Construction: Jackhammers, machinery.
- Industrial: Manufacturing plants.
- Domestic: Lawn mowers, loud music.
Effects on Humans
- Physiological: Permanent hearing loss, high blood pressure (hypertension), cardiovascular disease.
- Psychological: Stress, anxiety, insomnia, and difficulty communicating.
Effects on Wildlife
Animals rely heavily on sound for survival, making them highly vulnerable to noise pollution.
- Masking Communication: Loud ambient noise interferes with vocalizations used for mating calls or territorial warnings. (Example: Songbirds in cities sing at higher frequencies or louder volumes to be heard over traffic).
- Marine Disruption: Sonar (from military or mapping vessels) and shipping noise interfere with the echolocation of whales and dolphins. This can cause:
- Communication blocking.
- Disorientation leading to beaching.
- Physical damage to hearing organs.
- Predator/Prey Relationships: Noise can prevent predators from hearing prey movement, or prevent prey from hearing approaching predators.
Common Mistakes & Pitfalls
- Confusing Acid Rain with Ozone Depletion: These are two distinct issues. Acid rain is caused by $SOx$ and $NOx$ lowering the pH of precipitation. Ozone depletion is caused by CFCs breaking down the stratospheric ozone layer. They are not the same mechanism.
- Specifics of Catalytic Converters: Students often forget that catalytic converters regulate NOx and CO, not Carbon Dioxide ($CO2$). In fact, they create $CO2$ as a byproduct of combustion cleaning.
- Clean Water vs. Acid Water: A common misconception is that dirty water looks murky. Ecosystems impacted by acid rain often appear crystal clear because the acidity has killed the phytoplankton and algae that usually make water turbid. Clear water does not always mean healthy water.
- Logarithmic Scale of dB: Remember that a sound of 70 dB is 10 times more intense than 60 dB, not just "10 more" units.