AP Biology Unit 1: The Molecular Basis of Life
The Molecular Basis of Life
1. Structure of Water and Hydrogen Bonding
Life on Earth is dependent on the unique properties of water, which arise directly from its molecular structure.
Polarity and Hydrogen Bonds
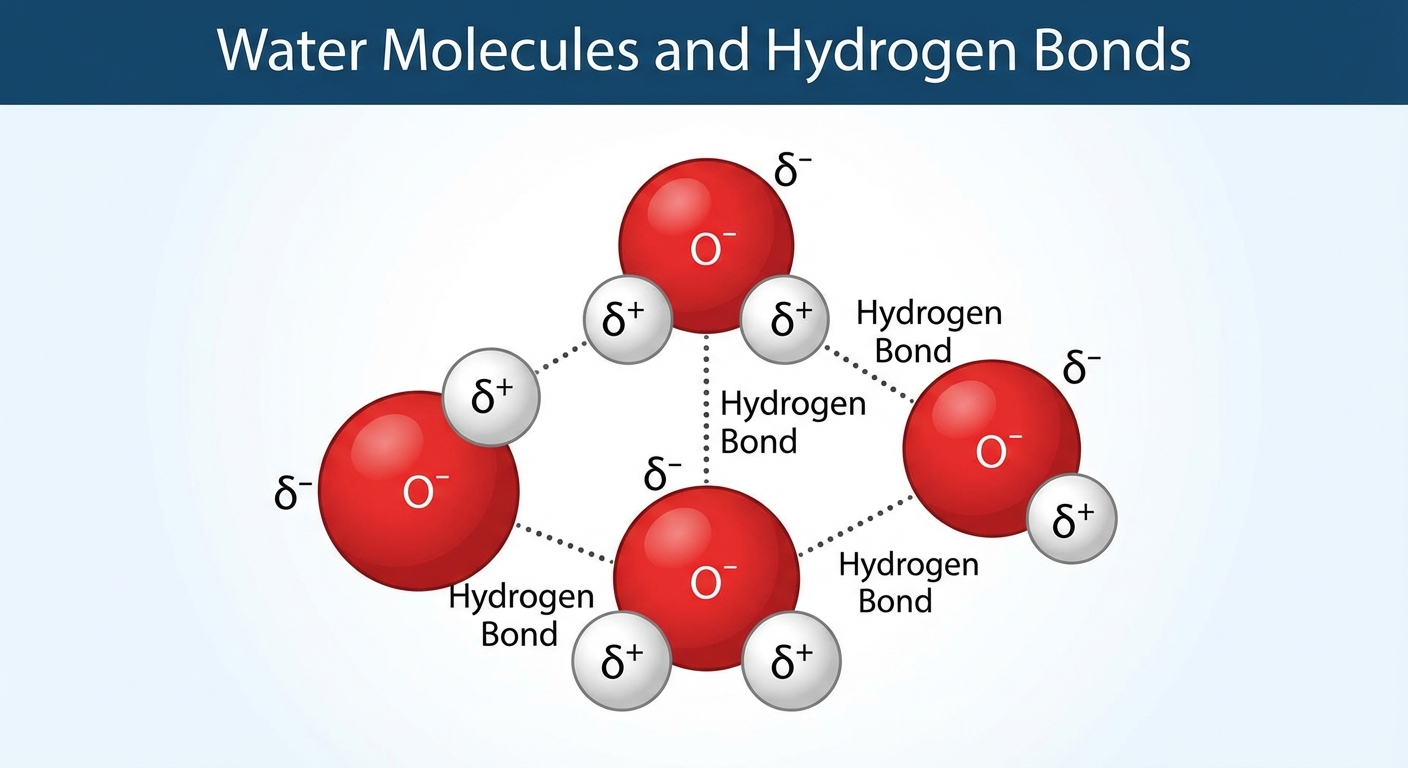
Water ($H_2O$) is a polar molecule. Oxygen consists of 8 protons and is highly electronegative (electron-greedy), while hydrogen has only 1 proton. This causes the shared electrons in the covalent bonds to spend more time near the oxygen atom.
- Polarity: The oxygen region has a partial negative charge ($\delta^-$), and the hydrogen regions have a partial positive charge ($\delta^+$).
- Hydrogen Bonds: The $\delta^+$ hydrogen of one water molecule is attracted to the $\delta^-$ oxygen of a neighboring molecule. These weak intermolecular forces are called hydrogen bonds.
Emergent Properties of Water
The hydrogen bonds between water molecules result in four life-supporting properties:
Cohesion and Adhesion
- Cohesion: Water molecules stick to each other due to hydrogen bonding. This creates surface tension (allows water striders to walk on water).
- Adhesion: Water molecules stick to other polar surfaces (like cell walls).
- Capillary Action: The combined force of cohesion and adhesion allows water to move up plant roots and stems against gravity.
High Specific Heat
- Water resists changes in temperature because heat energy must first be used to break hydrogen bonds before the molecules can move faster.
- Biological Significance: this stabilizes ocean temperatures and helps organisms maintain homeostasis (stable body temperature).
Expansion Upon Freezing
- Unlike most substances, water is less dense as a solid than as a liquid. At $0^\circ C$, water molecules form a crystalline lattice where hydrogen bonds keep molecules further apart.
- Biological Significance: Ice floats, insulating the liquid water below and allowing aquatic life to survive winter.
Versatile Solvent
- Water is the "solvent of life" because its polarity allows it to dissolve ionic compounds (salts) and polar molecules (sugars).
- Hydrophilic: "Water-loving" substances (polar/ionic).
- Hydrophobic: "Water-fearing" substances (non-polar, like oils) that do not dissolve in water.
2. The Elements of Life
While there are over 90 naturally occurring elements, biology is primarily built from a small subset.
CHONPS Mnemonic
Memorize the six major elements that make up 99% of living matter:
- Carbon
- Hydrogen
- Oxygen
- Nitrogen
- Phosphorus
- Sulfur
Biological Roles of Elements
| Element | Found in Major Macromolecules |
|---|---|
| Carbon | All organic molecules (Backbone of life) |
| Nitrogen | Proteins and Nucleic Acids |
| Phosphorus | Nucleic Acids and Phospholipids |
| Sulfur | Proteins (specifically Cysteine/Methionine) |
Note: Trace elements (like Iron in hemoglobin or Magnesium in chlorophyll) are required in minute quantities but are essential for function.
pH: Acids and Bases
Biochemical reactions are sensitive to pH (the concentration of $H^+$ ions).
- Acid: Increases $[H^+]$ concentration ($pH < 7$).
- Base: Reduces $[H^+]$ concentration ($pH > 7$).
- Buffers: Substances that minimize changes in pH by accepting $H^+$ when it is in excess and donating $H^+$ when it is depleted.
- Example: Carbonic acid-bicarbonate buffer system in human blood.
3. Introduction to Biological Macromolecules
Carbon: The Backbone
Carbon is tetravalent, meaning it can form 4 covalent bonds. This allows for complex, diverse, and large molecular structures (chains, rings, branching).
Synthesis and Breakdown
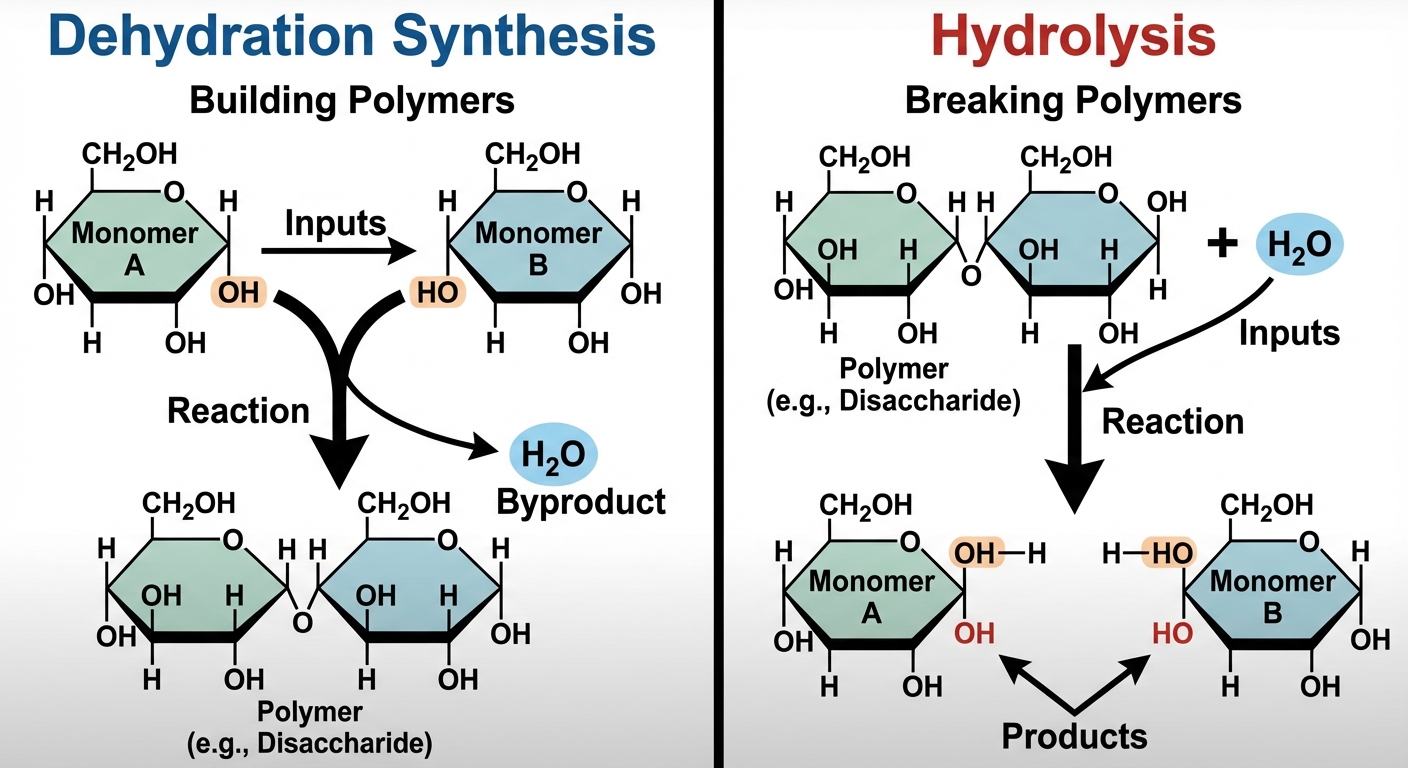
Macromolecules (except lipids) are polymers built from repeating units called monomers.
Dehydration Synthesis (Condensation Reaction):
- Joins two monomers together.
- Removes a water molecule ($H$ from one monomer, $OH$ from the other).
- Creates a covalent bond.
- Example: Glucose + Fructose $\rightarrow$ Sucrose + $H_2O$.
Hydrolysis:
- Breaks polymers apart.
- Adds a water molecule to break the bond.
- Example: Digestion of starch into glucose.
4. Carbohydrates
Function: Fuel (energy) and building material.
Elements: C, H, O (Ratio roughly 1:2:1, e.g., $C6H{12}O_6$).
Structure Hierarchy
- Monosaccharides (Monomers): Simple sugars like Glucose, Fructose, Galactose.
- Disaccharides: Two monomers joined by a Glycosidic Linkage (e.g., Sucrose, Maltose, Lactose).
- Polysaccharides (Polymers): Long chains of sugars.
Storage vs. Structure
The structure of the glucose monomer ($\alpha$ vs $\beta$ isomers) determines the function of the polymer.
| Type | Name | Found In | Function | Linkage Structure |
|---|---|---|---|---|
| Storage | Starch | Plants | Energy Storage | $\alpha$-glucose (helical/branched) |
| Storage | Glycogen | Animals (Liver/Muscle) | Energy Storage | Highly branched |
| Structural | Cellulose | Plant Cell Walls | Support | $\beta$-glucose (straight, rigid) |
| Structural | Chitin | Fungi/Insects | Exoskeletons | Glucose + Nitrogen group |
5. Lipids
Function: Energy storage, insulation, cell membranes, hormones.
Elements: C, H, O (and P in phospholipids).
Key Trait: Hydrophobic (Non-polar). They are not true polymers.
Types of Lipids
Triglycerides (Fats)
- Composed of 1 Glycerol + 3 Fatty Acids joined by Ester Linkages.
- Saturated Fats: All single bonds between carbons. Solid at room temperature (e.g., butter). "Saturated with hydrogen."
- Unsaturated Fats: Contains at least one double bond ($C=C$), causing a "kink" in the chain. Liquid at room temperature (e.g., plant oils).
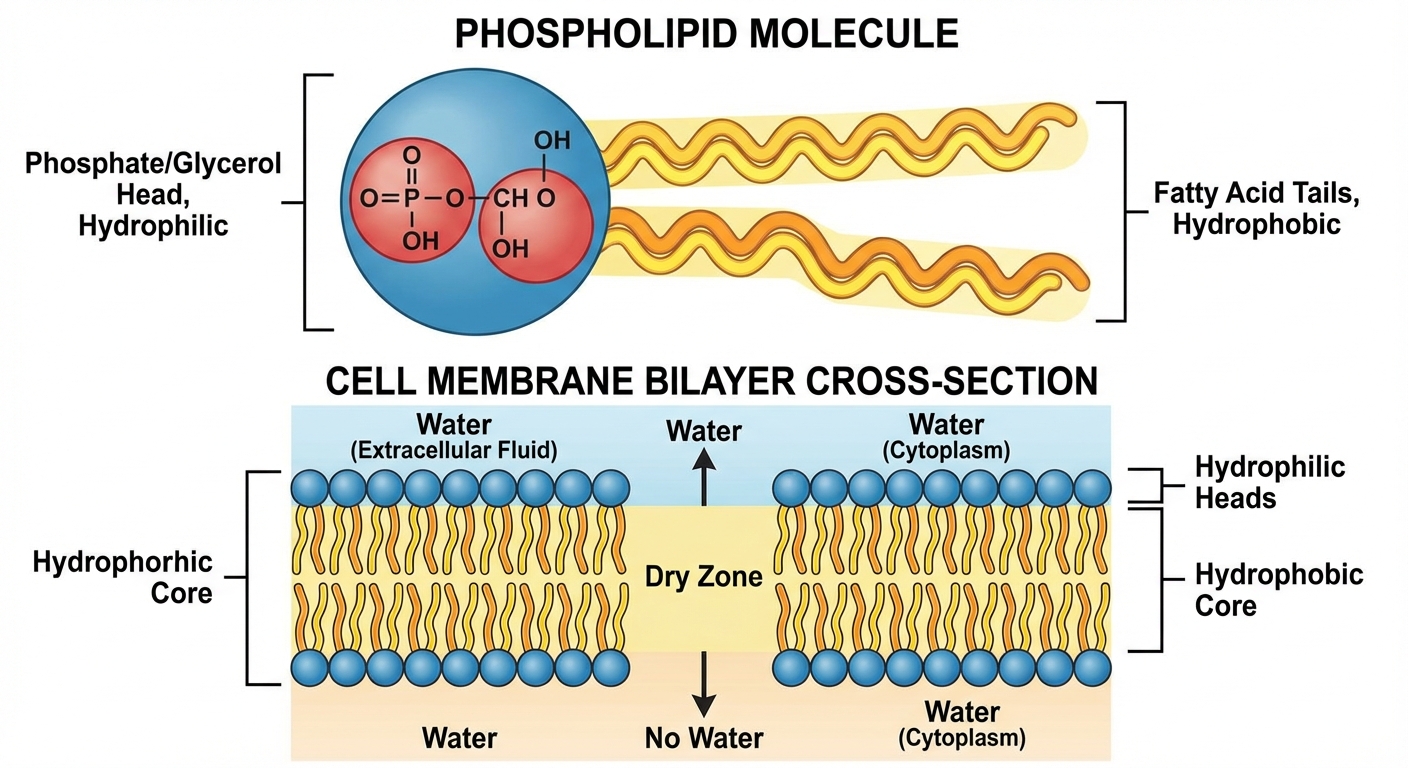
Phospholipids
- Structure: 1 Glycerol + 2 Fatty Acids + 1 Phosphate Group.
- Amphipathic: Has both a hydrophilic region (head) and hydrophobic region (tail).
- Forms the Phospholipid Bilayer of cell membranes.
Steroids
- Structure: Carbon skeleton with four fused rings.
- Example: Cholesterol (stabilizes membranes) and sex hormones (estrogen/testosterone).
6. Proteins
Function: Enzymes, defense (antibodies), storage, transport, hormones, receptors, movement, structure.
Elements: C, H, O, N, S.
Monomer: Amino Acid.
Amino Acid Structure
Every amino acid has a central carbon bonded to:
- An Amino group ($-NH_2$)
- A Carboxyl group ($-COOH$)
- A Hydrogen atom
- An R-Group (Side chain)
The R-Group: Contains the specific chemical properties (hydrophobic, hydrophilic, or ionic). It determines how the protein folds.
Polypeptide Formation
- Amino acids link via Peptide Bonds formed by dehydration synthesis.
- Directionality: Chains have an N-terminus (amino end) and a C-terminus (carboxyl end). New amino acids are always added to the C-terminus.
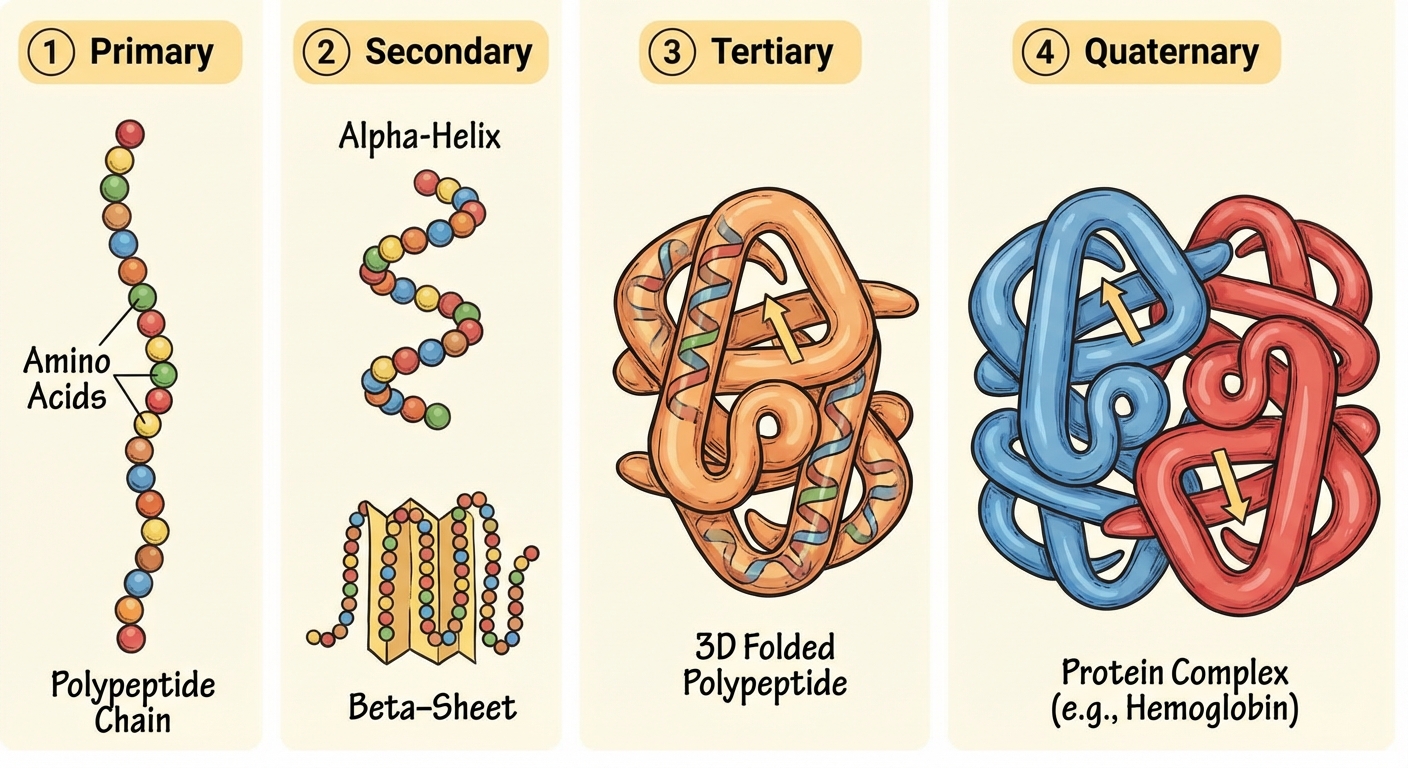
Levels of Protein Structure
Protein shape determines function. A change in shape is denaturation.
- Primary Structure: The unique linear sequence of amino acids (determined by DNA). Held by peptide bonds.
- Secondary Structure: Coils and folds in the polypeptide chain due to Hydrogen Bonding between the backbone components (not R-groups). Forms $\alpha$-helices and $\beta$-pleated sheets.
- Tertiary Structure: 3D shape formed by interactions between R-groups.
- Interactions: Hydrophobic interactions, Van der Waals forces, Hydrogen bonds, Ionic bonds, and Disulfide bridges (covalent bonds between Sulfur atoms in Cysteine).
- Quaternary Structure: Association of two or more polypeptide chains (e.g., Hemoglobin).
7. Nucleic Acids
Function: Store, transmit, and express hereditary information.
Elements: C, H, O, N, P.
Monomer: Nucleotide.
Nucleotide Structure
- Pentose Sugar (5-carbon).
- Phosphate Group (Negative charge).
- Nitrogenous Base.
DNA vs. RNA
| Feature | DNA (Deoxyribonucleic Acid) | RNA (Ribonucleic Acid) |
|---|---|---|
| Sugar | Deoxyribose | Ribose |
| Strands | Double-stranded (Double Helix) | Single-stranded |
| Bases | A, G, C, Thymine (T) | A, G, C, Uracil (U) |
| Directionality | Antiparallel ($5' \to 3'$ and $3' \to 5'$) | $5' \to 3'$ |
Directionality
- 5' End: Has a free Phosphate group.
- 3' End: Has a free Hydroxyl ($-OH$) group.
- DNA and RNA are synthesized in the $5' \to 3'$ direction.
Base Pairing Rules (Chargaff's Rules)
- Purines (A, G) pair with Pyrimidines (C, T, U).
- Context: Purines have 2 rings; Pyrimidines have 1 ring.
- A pairs with T (2 Hydrogen Bonds).
- G pairs with C (3 Hydrogen Bonds—stronger connection).
Common Mistakes & Pitfalls
- Bond Confusion: Students often confuse peptide bonds (covalent, between amino acids) with hydrogen bonds (weak, between secondary structure elements).
- Hydrolysis vs. Dehydration: Remember, if you are building muscle (protein), you are engaging in synthesis, which releases water (you sweat?). If you are digesting food, you add water (hydrolysis) to break bonds.
- Saturated vs. Unsaturated: Saturated means the molecule is "full" of hydrogens (straight chain). Unsaturated has a double bond which removes hydrogens (kinked chain).
- Nucleic Acid Directionality: Never forget that DNA strands are antiparallel. If one strand runs $5' \to 3'$, the complementary strand runs $3' \to 5'$. Exam questions often trick you by flipping the strand direction.
- Identifying Macromolecules:
- Uses $P$? It’s likely a Nucleic Acid or Phospholipid.
- Uses $S$? It’s a Protein.
- Ratio 1:2:1 ($C:H:O$)? It’s a Carbohydrate.