Comprehensive Guide to Kinetics: Energy, Collisions, and Catalysts
The Collision Model
For a chemical reaction to occur, it isn't enough for molecules simply to exist in the same container. They must interact physically. The Collision Model provides the foundational theory for understanding reaction rates based on particle behavior using Kinetic Molecular Theory (KMT).
The Three Requirements for a Successful Reaction
According to the collision model, three specific conditions must be met for a reaction to occur:
- Collision: The reactant particles must physically collide.
- Proper Orientation (Steric Factor): The particles must collide in a specific geometric alignment that exposes the reactive sites (atoms/bonds) to one another. If they hit "backwards," they simply bounce off.
- Sufficient Energy: The collision must possess enough kinetic energy to break existing chemical bonds. This energy threshold is called the Activation Energy ($E_a$).
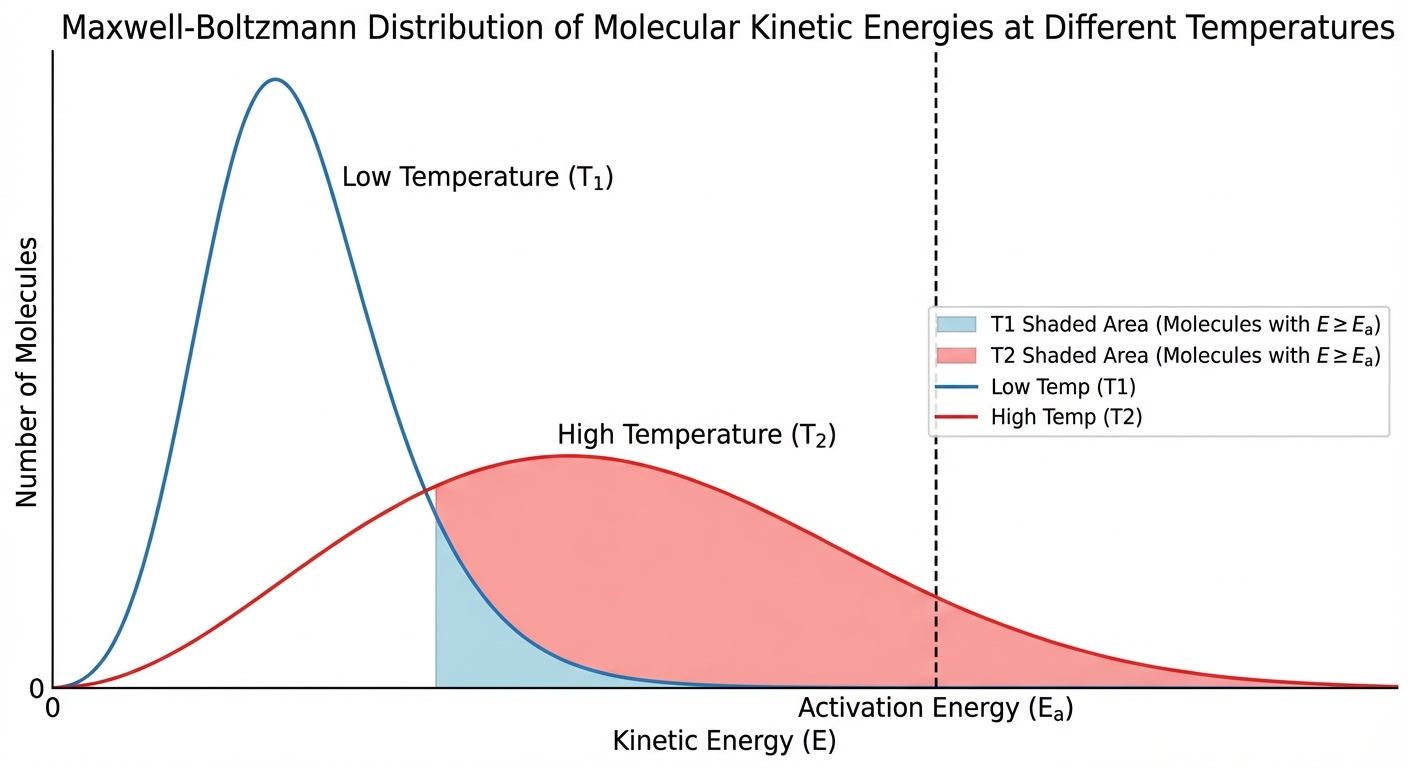
Maxwell-Boltzmann Distributions & Temperature
The relationship between collision energy and reaction rate is best visualized using a Maxwell-Boltzmann distribution graph.
- Temperature's Role: Increasing temperature does not change the activation energy ($E_a$). Instead, it shifts the curve of particle kinetic energies to the right and flattens it.
- The Result: A greater fraction of molecules now possess kinetic energy $\ge E_a$. This leads to a higher frequency of effective collisions and a faster reaction rate.
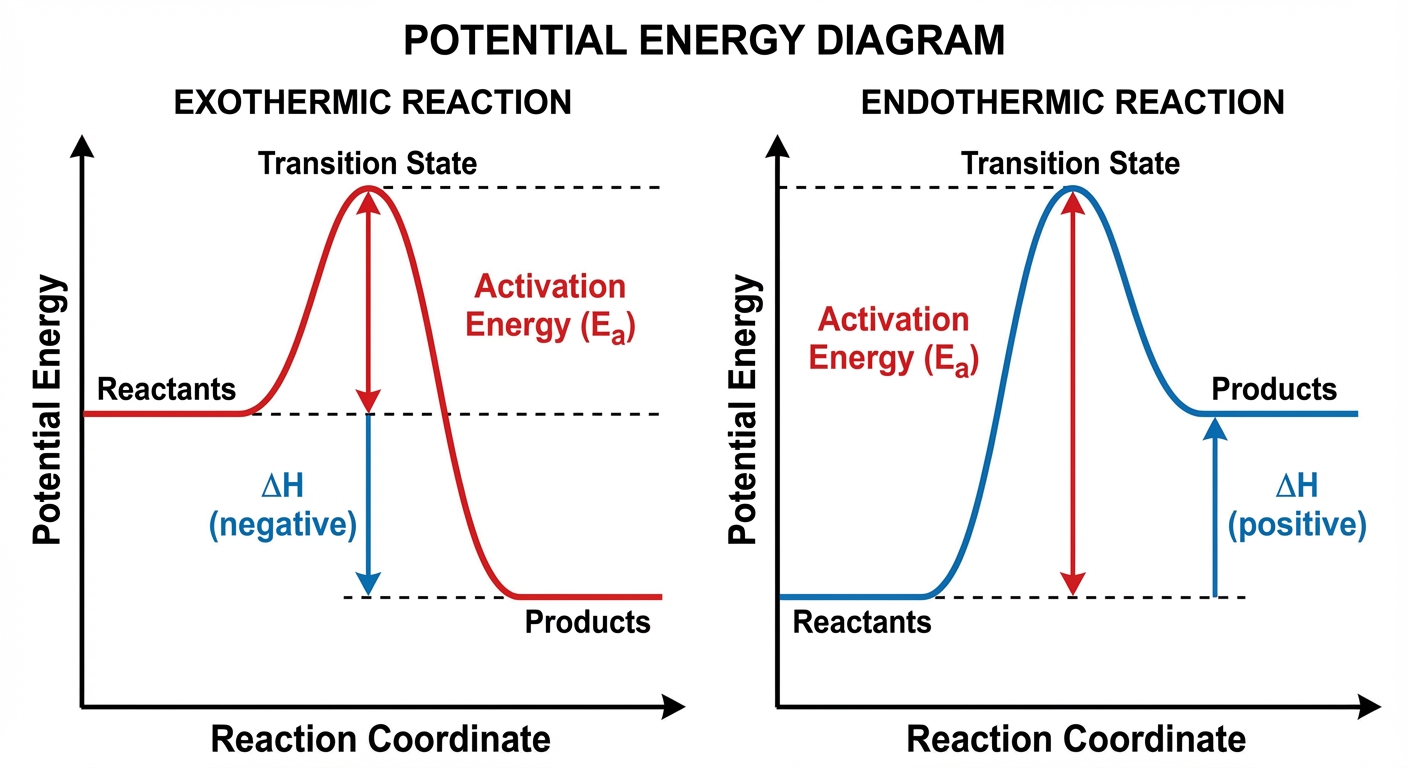
Reaction Energy Profiles
A Reaction Energy Profile (or potential energy diagram) maps the changing potential energy of a system as it moves from reactants to products. It is the "topographical map" of a chemical reaction.
Key Components of the Profile
- Reactants & Products: The flat plateaus at the start and end of the curve.
- Activation Energy ($E_a$): The energy difference between the Reactants and the peak of the curve.
- This is the energy "hump" or barrier the system must overcome.
- Transition State (Activated Complex): The absolute peak of the curve. This is a high-energy, unstable arrangement of atoms where bonds are in the process of breaking and forming. It cannot be isolated.
- Enthalpy Change ($\Delta H$): The energy difference between Products and Reactants.
Exothermic vs. Endothermic Profiles
- Exothermic Reaction: The products are lower in energy than the reactants ($\Delta H < 0$). Energy is released.
- Endothermic Reaction: The products are higher in energy than the reactants ($\Delta H > 0$). Energy is absorbed.
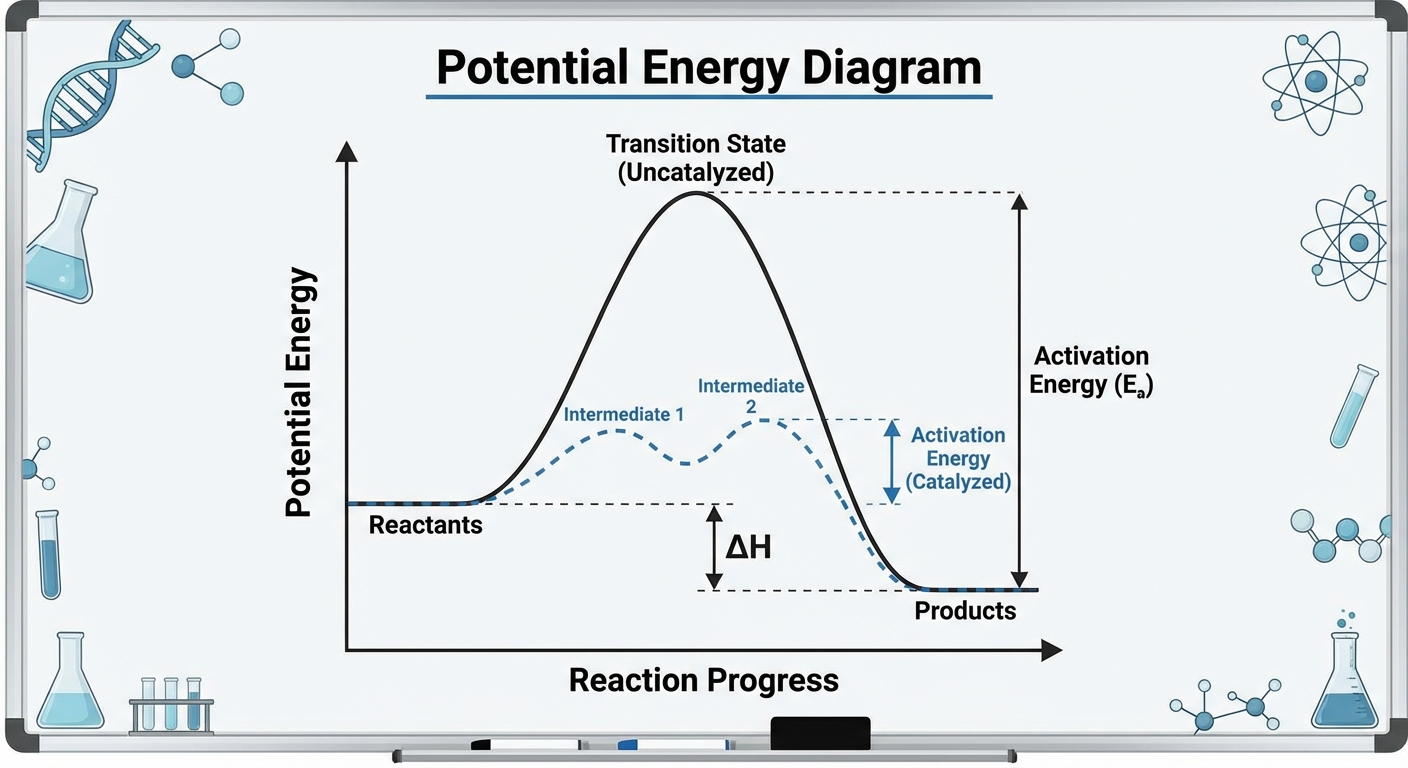
Multistep Reaction Energy Profiles
Most complex reactions do not occur in a single collision event; they happen in a series of steps called a reaction mechanism. The energy profile for these reactions looks like a roller coaster with multiple hills.
Interpreting the Hills and Valleys
- Number of Hills: Each hump represents an elementary step in the mechanism. If there are two hills, it is a two-step mechanism.
- Valleys (Intermediates): The local minimum (valley) between two hills represents a Reaction Intermediate. Unlike the transition state, an intermediate is a distinct chemical species that is produced in one step and consumed in a subsequent step. It has a slight duration of stability.
- Rate-Determining Step (RDS): The reaction can only go as fast as its slowest step. On an energy profile, the step with the highest activation energy barrier (the tallest hill relative to the valley preceding it) is the Rate-Determining Step.
Memory Aid: Think of a convoy of trucks driving over a mountain range. The entire convoy is slowed down by the steepest, most difficult mountain pass. That pass is the Rate-Determining Step.
Identifying Species in Mechanisms
In a written mechanism (equations) versus the graph:
- Intermediate: Appears in the products of an early step and reactants of a later step. (Cancels out when summing steps).
- Catalyst: Appears in the reactants of an early step and products of a later step. (Is regenerated).
Catalysis
A Catalyst is a substance that increases the rate of a chemical reaction without being consumed in the overall process.
How Catalysts Work
Catalysts generally provide an alternative reaction pathway (mechanism) that has a lower activation energy ($E_a$) than the uncatalyzed pathway.
By lowering the threshold ($E_a$), a larger fraction of molecules at the same temperature will have sufficient energy to collide effectively. See the Arrhenius relationship:
If $E_a$ decreases, the exponent becomes less negative, and the rate constant $k$ increases exponentially.
Types of Catalysis
- Acid-Base Catalysis: Reactants gain or lose a proton to become more reactive.
- Surface Catalysis (Heterogeneous): A reactant creates a bond with the surface of a solid (adsorption), which weakens internal bonds within the reactant or orients the reactant favorably (improves steric factor).
- Enzyme Catalysis: Biological molecules bind substrates in an "active site," stabilizing the transition state and dramatically lowering $E_a$.
What a Catalyst Does NOT Do
It is vital to distinguish between kinetics (rate) and thermodynamics (equilibrium).
| Property | Effect of Catalyst |
|---|---|
| Activation Energy ($E_a$) | Decreases (Lowers the barrier) |
| Mechanism | Changes (Provides alternate path) |
| Reaction Rate | Increases (Both forward and reverse) |
| Enthalpy ($\Delta H$) | No Change (Start and end points are the same) |
| Equilibruim Constant ($K$) | No Change (Ratio of products/reactants at equilibrium is constant) |
| Gibbs Free Energy ($\Delta G$) | No Change |
Common Mistakes & Pitfalls
Confusing Temperature and Activation Energy:
- Mistake: "Increasing temperature lowers the activation energy."
- Correction: Temperature NEVER changes the value of $Ea$. Temperature gives particles more kinetic energy so they can overcome the fixed $Ea$ barrier. Only a catalyst changes $E_a$.
Catalysts and Yield:
- Mistake: "Adding a catalyst increases the amount of product produced."
- Correction: Catalysts get you to equilibrium faster, but they do not change the position of equilibrium. You get the same yield, just sooner.
Identifying Intermediates vs. Transition States:
- Mistake: Confusing the top of the hill with the valley.
- Correction: Transition States are at the peaks (maxima); Intermediates are in the valleys (minima). You can isolate an intermediate; you cannot isolate a transition state.
Orientation Neglect:
- Mistake: Assuming collision rate equals reaction rate.
- Correction: Don't forget the steric factor. Millions of collisions happen for every successful reaction because molecules must hit with the correct geometric alignment.