Comprehensive Guide to AP Environmental Science: Biogeochemical Cycles
Introduction to Biogeochemical Cycles
Life on Earth depends on the recycling of essential elements. Unlike energy, which flows through an ecosystem in one direction (Sun $\rightarrow$ Producers $\rightarrow$ Consumers $\rightarrow$ Heat), matter is recycled. A Biogeochemical Cycle refers to the movement of matter within and between ecosystems involving biological, geological, and chemical processes.
To master this section of AP Environmental Science (APES), you must understand the Law of Conservation of Matter: matter creates nor destroys itself; it only changes forms. In these cycles, we track specific elements as they move between sources (reservoirs that release more material than they accept) and sinks (reservoirs that accept more material than they release).
Key Concepts
- Reservoir (Pool): A place where a nutrient sits for a period of time (e.g., the atmosphere, the deep ocean).
- Flux: The rate of movement of nutrients between reservoirs.
- Residence Time: The average time an element stays in a particular reservoir.
The Carbon Cycle
Carbon is the fundamental building block of life, essential for forming carbohydrates, lipids, proteins, and nucleic acids. The carbon cycle is unique because it has both a "fast" biological track and a "slow" geological track.
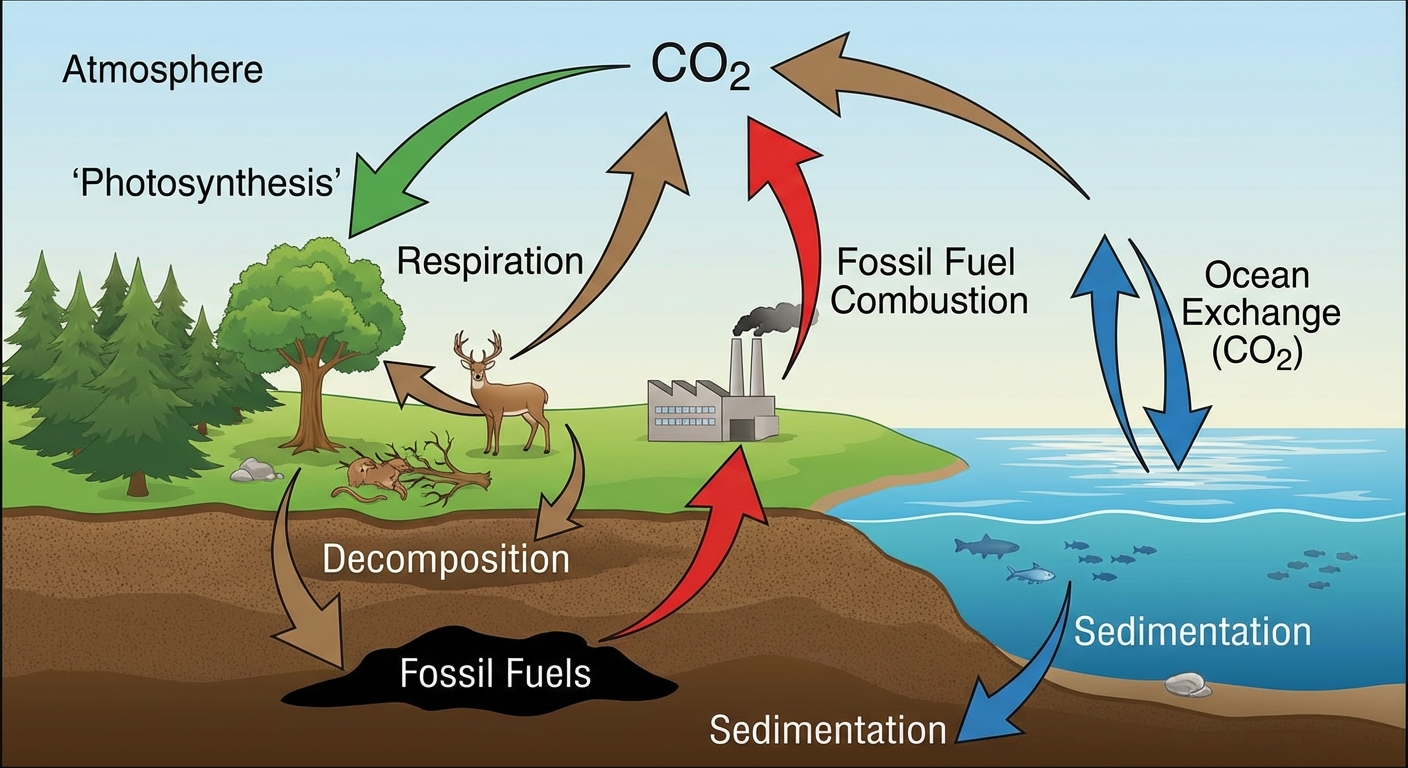
Major Reservoirs
- Sedimentary Rock (Limestone): The largest reservoir of carbon. Carbon is trapped here for millions of years.
- Oceans: The second-largest reservoir. Contains dissolved $CO_2$ and carbonate ions.
- Fossil Fuels: Coal, oil, and natural gas formed from ancient biomass.
- Biosphere: Living plants and animals.
- Atmosphere: Primarily as carbon dioxide ($CO2$) and methane ($CH4$). While accurate measurements vary, the atmospheric concentration is rapidly increasing due to human activity.
Key Processes
1. Photosynthesis & Respiration (Fast Cycle)
This is the primary exchange between the biosphere and the atmosphere.
- Photosynthesis: Producers (plants, algae) take in atmospheric $CO2$ and convert it into glucose.
- Cellular Respiration: Organisms (including plants) break down glucose for energy, releasing $CO2$ back into the atmosphere.
2. Sedimentation & Burial (Slow Cycle)
- Dissolved $CO2$ combines with calcium in the ocean to form calcium carbonate ($CaCO3$).
- Marine organisms use this for shells/skeletons. When they die, they sink and form limestone sediments over millions of years.
- Organic matter buried without oxygen can transform into Fossil Fuels.
3. Air-Sea Gas Exchange
- There is a constant abiotic flux of $CO2$ dissolving into the ocean and diffusing back out. As atmospheric $CO2$ rises, the ocean absorbs more, leading to Ocean Acidification.
4. Human Impacts
- Combustion of Fossil Fuels: digging up "old" carbon (sinks) and burning it releases it rapidly into the atmosphere ($CO_2$), contributing to global climate change.
- Deforestation: Removes carbon, cutting down trees that act as carbon sinks.
The Nitrogen Cycle
Nitrogen is a critical limiting factor for plant growth and is required to build amino acids (proteins) and nucleic acids (DNA/RNA). The Nitrogen Cycle is often the most difficult for students because it relies heavily on specific bacteria for chemical transformations.
Crucial Fact: The atmosphere is 78% Nitrogen gas ($N_2$), but this form is biologically unavailable to most plants and animals.
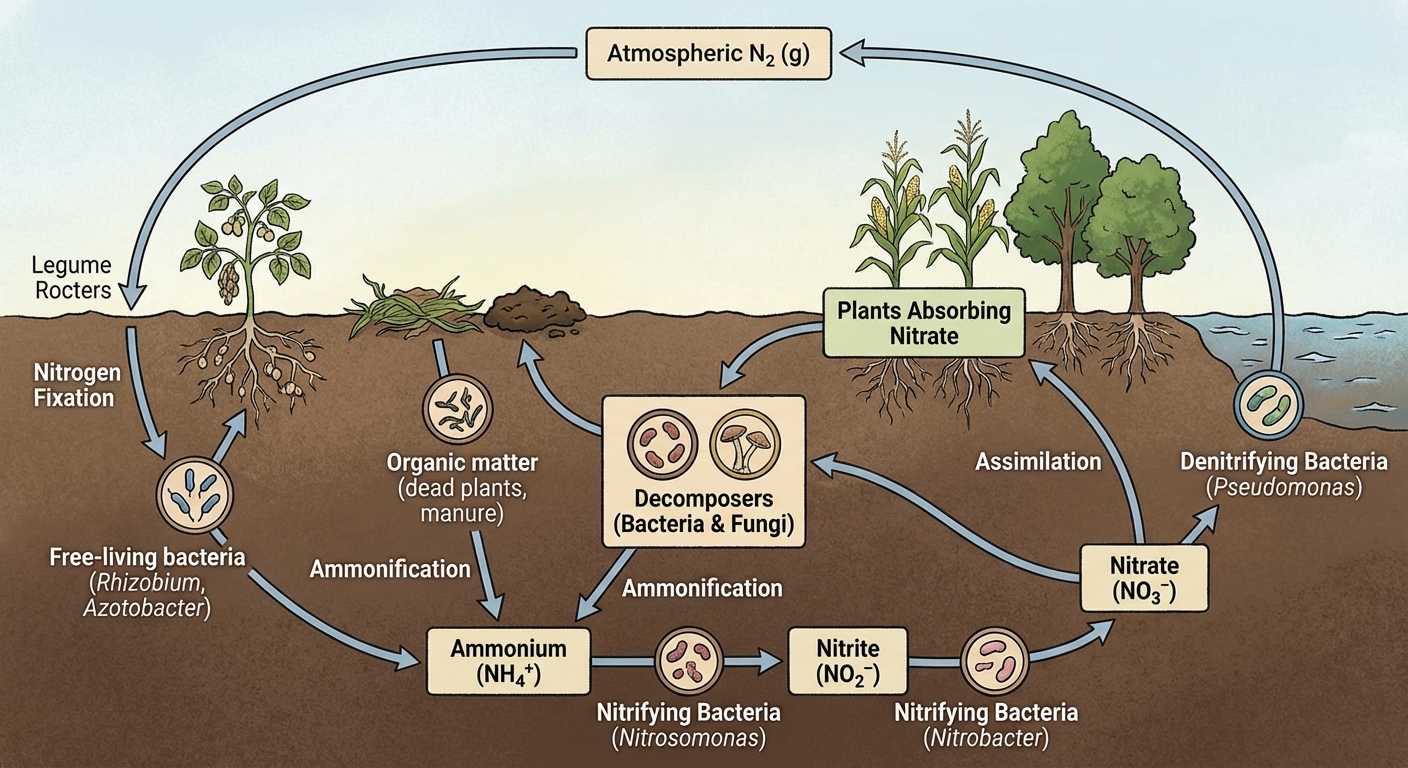
The Major Reservoir
- Atmosphere: The largest reservoir ($N_2$ gas).
The "FixNAAD" Mnemonic
Use this acronym to remember the steps and the corresponding products:
| Step Name | Process | Product / Result |
|---|---|---|
| Fixation | $N_2$ converted to usable forms | Ammonia ($NH3$) / Ammonium ($NH4^+$) |
| Nitrification | Soil bacteria convert ammonium | Nitrites ($NO2^-$) $\rightarrow$ Nitrates ($NO3^-$) |
| Assimilation | Plants take up Nitrate via roots | Organic N (Proteins/DNA) |
| Ammonification | Decomposers break down dead matter | Ammonium ($NH_4^+$) returned to soil |
| Denitrification | Bacteria convert Nitrate back to gas | Nitrogen Gas ($N_2$) |
Detailed Process Explanations
Nitrogen Fixation:
- Biotic: Specialized bacteria (e.g., Rhizobium) living in root nodules of legumes (peas, beans) convert $N2$ into ammonia ($NH3$).
- Abiotic: Lightning strikes or industrial processes (fertilizer production) convert $N_2$ into nitrates.
Nitrification:
- Ammonium is converted by soil bacteria first into Nitrite ($NO2^-$) and then into Nitrate ($NO3^-$). Nitrate is the preferred form of nitrogen for plants.
Assimilation:
- Plants absorb Nitrate through root hairs and incorporate it into their tissues. Animals assimilate nitrogen by eating plants.
Ammonification (Mineralization):
- Fungal and bacterial decomposers break down organic nitrogen found in dead bodies and waste products, converting it back into inorganic ammonium ($NH_4^+$).
Denitrification:
- In oxygen-poor (anaerobic) soils, such as waterlogged wetlands, bacteria convert Nitrate back into nitrous oxide ($N2O$) and eventually Nitrogen gas ($N2$), returning it to the atmosphere.
Human Impacts
- Excess Fertilizer: Using synthetic fertilizers adds huge amounts of Nitrates to soil. Runoff causes eutrophication (algal blooms) in water bodies.
- Combustion: Burning fossil fuels releases Nitrogen Oxides ($NO_x$), a precursor to photochemical smog and acid rain.
The Phosphorus Cycle
Phosphorus is essential for ATP (energy), DNA, and cell membranes (phospholipids). The Phosphorus cycle is distinct for being the slowest cycle and having no atmospheric phase.
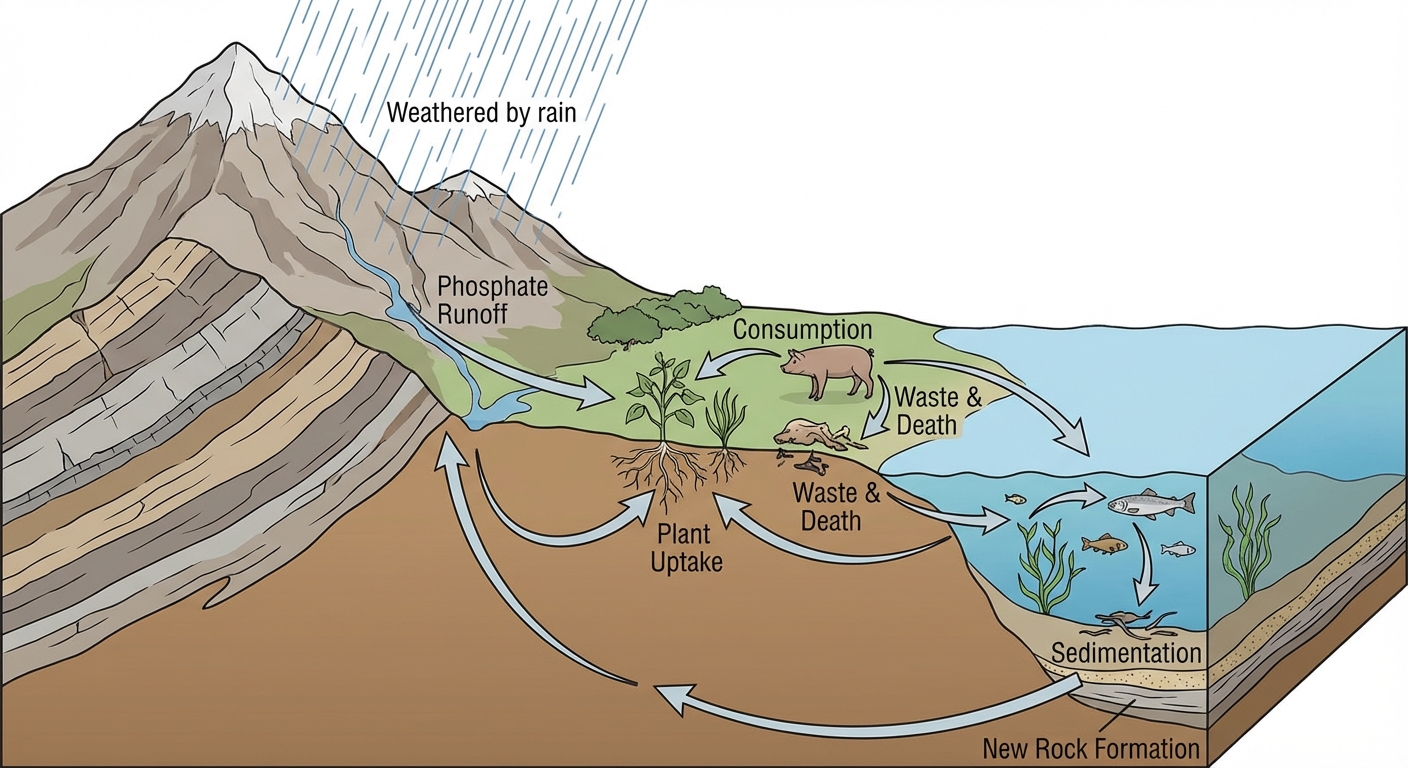
Major Reservoirs
- Lithosphere (Rocks/Sediment): The primary reservoir. Phosphorus is found in phosphate rocks.
Key Processes
Weathering & Erosion:
- Rain and weathering break down phosphate rocks, releasing phosphate ions ($PO_4^{3-}$) into the soil and water.
Assimilation:
- Plants absorb phosphate via roots from the soil. Animals obtain it by eating plants.
Excretion & Decomposition:
- Phosphorus returns to the soil through animal waste (guano is a rich source) and the decomposition of dead organic matter.
Sedimentation & Uplift:
- Phosphates in water can settle to the bottom, forming new sedimentary rocks. Geological forces eventually lift these rocks to the surface, but this takes millions of years.
Limiting Factor
Because phosphorus does not exist as a gas and weathers slowly, it is often the limiting factor for plant growth in many ecosystems. This is why it is a key ingredient in fertilizers.
Human Impacts
- Fertilizers & Detergents: Synthetic fertilizers and phosphate-heavy detergents in runoff lead to rapid eutrophication in aquatic systems (hypoxic "dead zones").
The Hydrologic (Water) Cycle
Water moves biological elements through the ecosystem. The cycle is powered by the Sun.
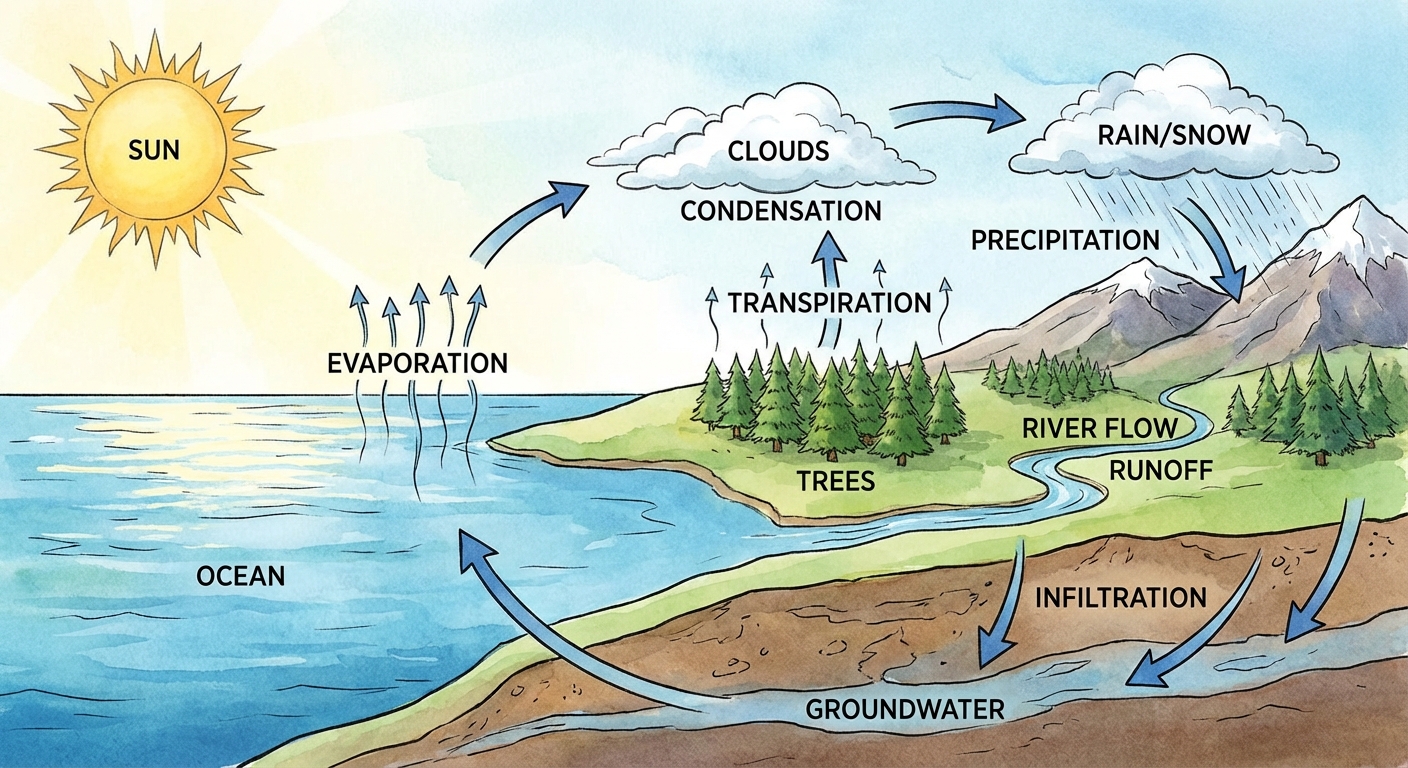
Key Processes
- Evaporation: Solar energy heats water in oceans/lakes, turning it into vapor.
- Transpiration: The process by which plants release water vapor into the air through stomata in their leaves. Combined with evaporation, this is often called Evapotranspiration.
- Condensation: Water vapor cools and forms clouds.
- Precipitation: Rain, snow, or hail brings water back to Earth.
- Runoff vs. Infiltration:
- Runoff: Water moves across the land surface into streams/rivers.
- Infiltration (Percolation): Water soaks into the ground, recharging aquifers (groundwater reservoirs).
Human Impacts
- Impervious Surfaces: Concrete and asphalt prevent infiltration, increasing runoff and flash flooding.
- Deforestation: Reduces transpiration, altering local rainfall patterns.
- Groundwater Depletion: Pumping water from aquifers faster than nature refills them (e.g., Ogallala Aquifer).
Summary Comparison Table
| Cycle | Major Reservoir (Sink) | Atmospheric Component? | Key Process/Driving Force | Human Impact |
|---|---|---|---|---|
| Carbon | Sedimentary Rock | Yes ($CO2, CH4$) | Photosynthesis/Resp. | Burning Fossil Fuels |
| Nitrogen | Atmosphere ($N_2$) | Yes ($N_2$) | Bacterial Action | Fertilizer, Smog |
| Phosphorus | Rocks | NO | Weathering/Erosion | Fertilizer / Soap |
| Water | Ocean | Yes (Vapor) | Sun (Heat) | Paving / Irrigation |
Common Mistakes & Pitfalls
Confusing the Atmosphere:
- Mistake: Thinking Phosphorus cycles through the air.
- Correction: Phosphorus has no gas phase; it never enters the atmosphere. This makes it a very slow cycle.
Nitrogen Fixation vs. Nitrification:
- Mistake: Switching the definitions.
- Correction: Fixation is turning gas ($N2$) into a solid ($NH3$). Nitrification is turning that solid into a plant-food version ($NO_3^-$).
Plants and Nitrogen Gas:
- Mistake: Believing plants "breathe in" Nitrogen like they do $CO_2$.
- Correction: Plants cannot use atmospheric $N_2$. They rely entirely on bacteria in the soil to process it first.
Source vs. Sink Logic:
- Mistake: Assuming fossil fuels are a source of carbon naturally.
- Correction: Fossil fuels are naturally a sink (holding carbon). When humans burn them, we artificially turn them into a massive source.
Eutrophication Agents:
- Mistake: Thinking Carbon causes eutrophication.
- Correction: Eutrophication (algae blooms) is primarily caused by excess Nitrogen and Phosphorus.