AP Chemistry Unit 7: Mechanics of Equilibrium and Calculations
The Magnitude of the Equilibrium Constant
The equilibrium constant ($K$) is a numerical value that describes the ratio of product concentrations to reactant concentrations when a chemical system has reached equilibrium. The magnitude of this number is crucial because it indicates the extent of the reaction.
The general expression for the reaction $aA + bB \rightleftharpoons cC + dD$ is:
K_{eq} = \frac{[C]^c [D]^d}{[A]^a [B]^b}
Interpreting the Value of K
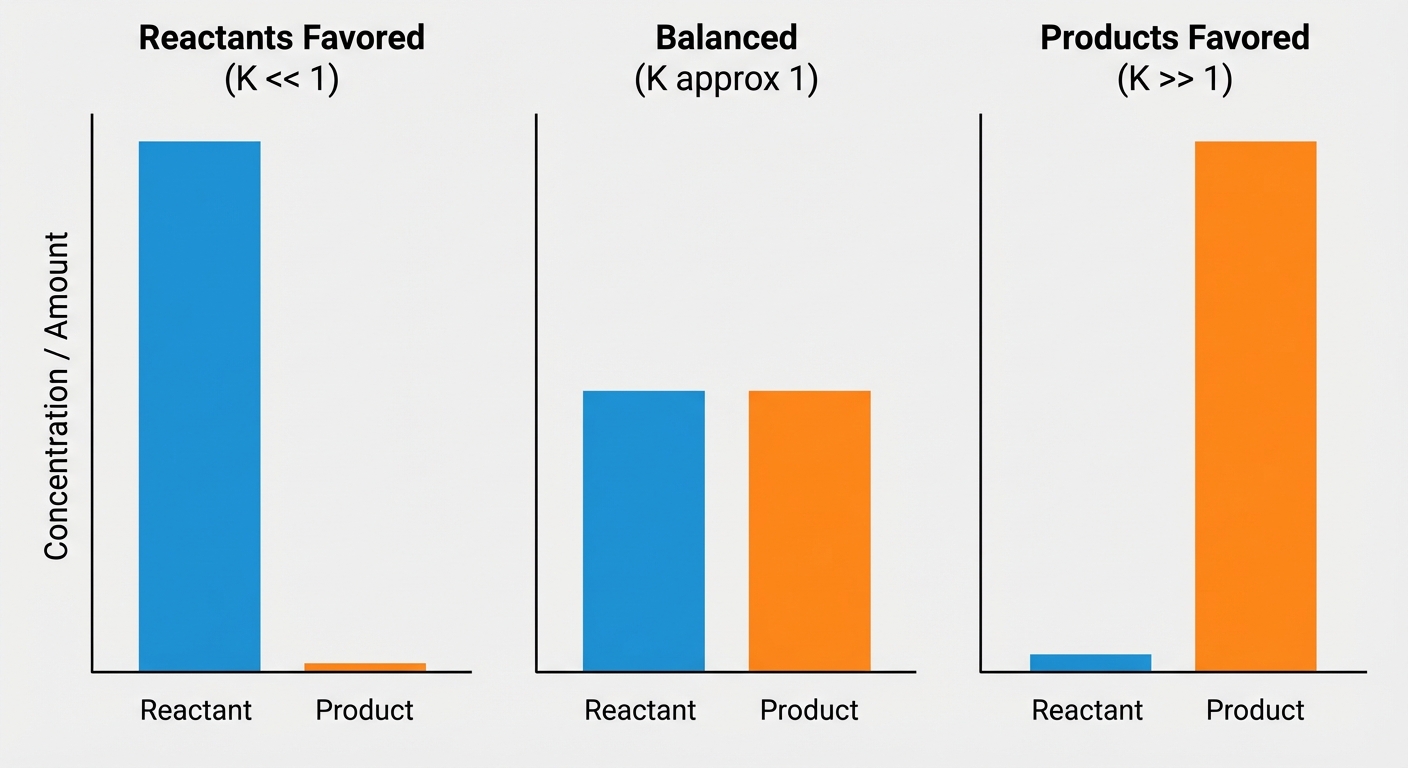
Depending on the size of $K$, we can predict which species (reactants or products) dominate the mixture at equilibrium.
$K \gg 1$ (Generally $K > 1000$): Products Favored
- The numerator (products) is much larger than the denominator (reactants).
- The reaction goes virtually to completion.
- At equilibrium, the system consists mostly of products.
$K \ll 1$ (Generally $K < 0.001$): Reactants Favored
- The denominator (reactants) is much larger than the numerator (products).
- The reaction barely proceeds forward.
- At equilibrium, the system consists mostly of reactants.
$K \approx 1$: Comparable Amounts
- Neither reactants nor products strongly dominate.
- Significant amounts of both reactants and products are present at equilibrium.
$Kc$ vs. $Kp$
While $K_c$ uses molar concentrations (M), reactions involving gases often use partial pressures (atm). The relationship is derived from the Ideal Gas Law:
Kp = Kc(RT)^{\Delta n}
Where:
- $R$ is the ideal gas constant ($0.08206 \text{ L·atm/(mol·K)}$)
- $T$ is temperature in Kelvin
- $\Delta n$ = (moles of gaseous product) $-$ (moles of gaseous reactant)
Properties and Manipulation of the Equilibrium Constant
Sometimes, the reaction you are studying is a variation of a known reaction. The value of $K$ changes in predictable ways when the stoichiometric equation is manipulated.
1. Reversing the Reaction
If you reverse the equation, the products become reactants and vice versa. Mathematically, the expression is inverted.
- Reaction: $A \rightleftharpoons B$ has constant $K_1$
- Reversed: $B \rightleftharpoons A$ has constant $K_{new}$
- Rule:
K{new} = \frac{1}{K1}
2. Multiplying Coefficients
If you multiply the stoichiometric coefficients by a factor $n$, the new equilibrium constant is the original constant raised to the power of $n$.
- Reaction: $nA \rightleftharpoons nB$
- Rule:
K{new} = (K1)^n
3. Adding Reactions (Hess's Law for K)
If a reaction is the sum of two or more elementary steps, the overall equilibrium constant is the product (multiplication) of the constants for the individual steps.
- Step 1: $A \rightleftharpoons B$ ($K_1$)
- Step 2: $B \rightleftharpoons C$ ($K_2$)
- Overall: $A \rightleftharpoons C$
- Rule:
K{total} = K1 \times K_2
Example:
Given: $2A \rightleftharpoons B$ ($K=4$). What is $K$ for $1/2 B \rightleftharpoons A$?
- Reverse the reaction: $B \rightleftharpoons 2A \Rightarrow K' = 1/4 = 0.25$
- Multiply coefficients by $1/2$: $1/2 B \rightleftharpoons A \Rightarrow (0.25)^{1/2} = \sqrt{0.25} = 0.5$
Calculating Equilibrium Concentrations
To solve for equilibrium concentrations given initial conditions, we use the R.I.C.E. table method. This helps organize the stoichiometry and changes in mass.
The Reaction Quotient ($Q$)
Before calculating, calculate $Q$ to determine which direction the reaction will shift to reach equilibrium. $Q$ is calculated exactly like $K$, but using current/initial concentrations rather than equilibrium ones.
- If $Q < K$: Ratio is too small. Shift Right (towards products).
- If $Q > K$: Ratio is too large. Shift Left (towards reactants).
- If $Q = K$: The system is at equilibrium.
The R.I.C.E. Table
- Reaction: The balanced chemical equation.
- Initial: Starting concentrations (M) or pressures (atm).
- Change: The change required to reach equilibrium, represented by $x$. Use stoichiometry coefficients here.
- Equilibrium: The algebraic sum of Initial and Change ($I + C$).
Worked Example
Consider the reaction $2NO2(g) \rightleftharpoons N2O4(g)$ with $Kc = 8.0$. If you start with $0.10 \text{ M } NO_2$ and no product, what are the equilibrium concentrations?
Step 1: Set up the Table
| $2NO_2$ | $\rightleftharpoons$ | $N2O4$ | |
|---|---|---|---|
| I | $0.10$ | $0$ | |
| C | $-2x$ | $+x$ | |
| E | $0.10 - 2x$ | $x$ |
Note: Reactants decrease ($-$), Products increase ($+$). The coefficient 2 on $NO_2$ means calculation is $-2x$.
Step 2: Substitute into Equilibrium Expression
8.0 = \frac{[N2O4]}{[NO_2]^2} = \frac{x}{(0.10 - 2x)^2}
Step 3: Solve for x
Take the square root of both sides (since the denominator is a perfect square and the numerator coefficient is 1 relative to K if simplified, but here we can just do algebra):
\sqrt{8.0} = \frac{\sqrt{x}}{0.10 - 2x} \quad \text{(Wait, this is messy without a perfect square on the left.)}
Correction strategy: Since this isn't a perfect square, you would normally expand to a quadratic equation: $4x^2 - 1.4x + 0.08 = 0$ (approx). On the AP exam, you often get perfect squares OR you use the "Small x Optimization."
The "5% Rule" (Small x Approximation)
If $K$ is very small ($10^{-4}$ or smaller), we assume $x$ is negligible compared to the initial concentration.
Example: $(0.10 - 2x) \approx 0.10$. This simplifies math significantly. Always verify that $x$ is indeed less than 5% of the initial value.
Representations of Equilibrium
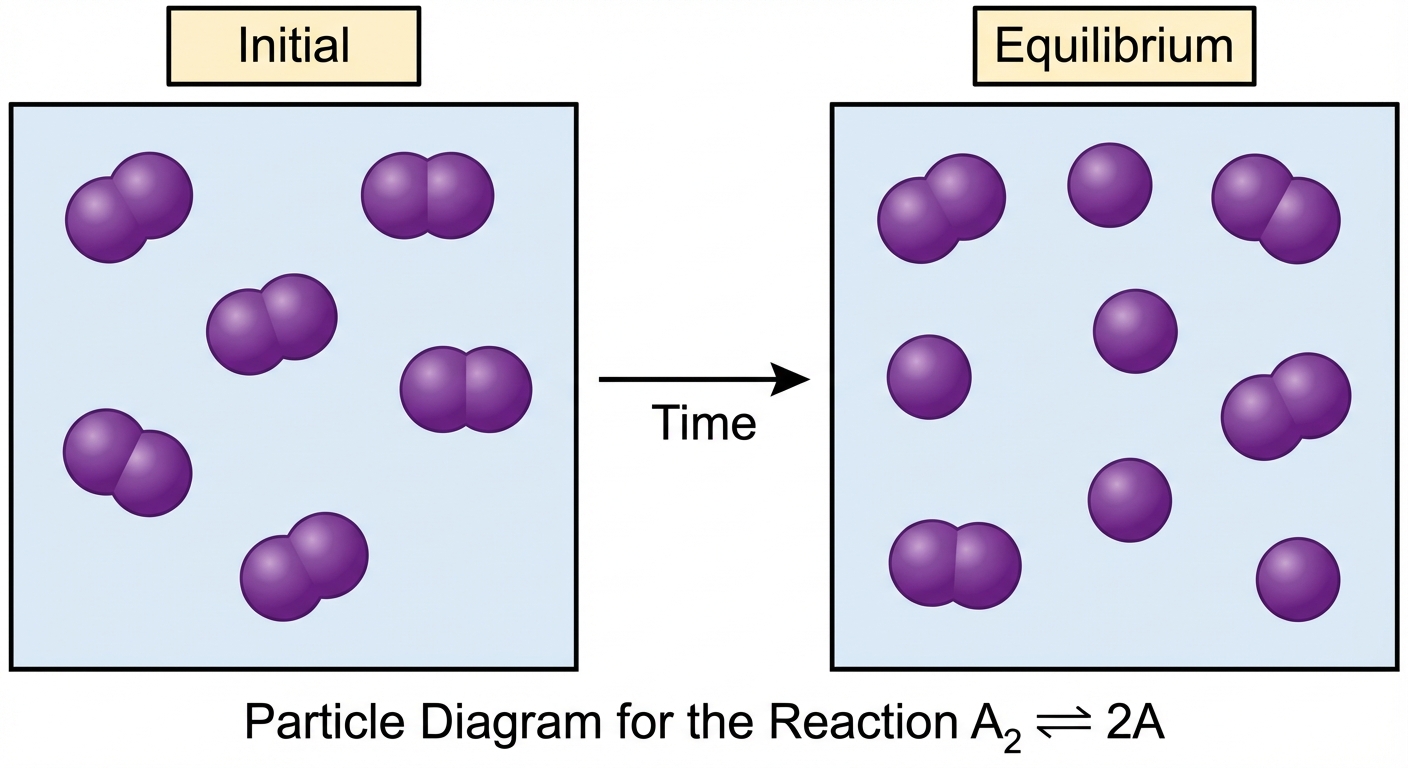
On the AP exam, you must translate between mathematical calculations and particulate models (diagrams of atoms/molecules).
Particle Diagrams
A particle diagram represents a snapshot of the reaction mixture.
- Counting: The number of particles drawn represents relative moles (or concentration).
- Ratios: If $K=1$, you should see roughly balanced amounts of reactant and product particles.
- Stoichiometry: If the reaction is $A \rightarrow 2B$, every time one particle of A disappears, two particles of B must appear.
Common Mistakes & Pitfalls
- Solids and Liquids: Never include pure solids ($s$) or liquids ($l$) in the $K$ or $Q$ expression. Their concentrations are constant. Only include gas ($g$) and aqueous ($aq$) species.
- Confusing Initial vs. Equilibrium: Students often plug initial concentrations into the $K$ expression. Only Equilibrium row values go into $K$. Initial values go into $Q$.
- Forgetting Coefficients in Powers: In the expression $K = \frac{[B]^2}{[A]}$, students often forget to square the concentration of B.
- Rate vs. Equilibrium: A large $K$ means the reaction goes far to the right; it does not mean the reaction is fast. Rate is determined by activation energy ($E_a$), not $K$.
- Unit Mismatch: Do not mix Molarity and partial pressure in the same calculation without converting via $PV=nRT$.