AP Environmental Science: Stratospheric Ozone Dynamics
Stratospheric Ozone Protection
The "Ozone Layer" Basics
Stratospheric Ozone ($O_3$) differs fundamentally from the ground-level ozone studied in air pollution units. While tropospheric ozone is a respiratory irritant and pollutant (smog), stratospheric ozone is a critical shield for life on Earth. A helpful mnemonic to remember this distinction is:
"Good up high, bad nearby."
This layer, located approximately 16–30 km above Earth's surface, acts as a planetary sunscreen. It absorbs the majority of the sun's harmful ultraviolet (UV) radiation before it reaches the surface.
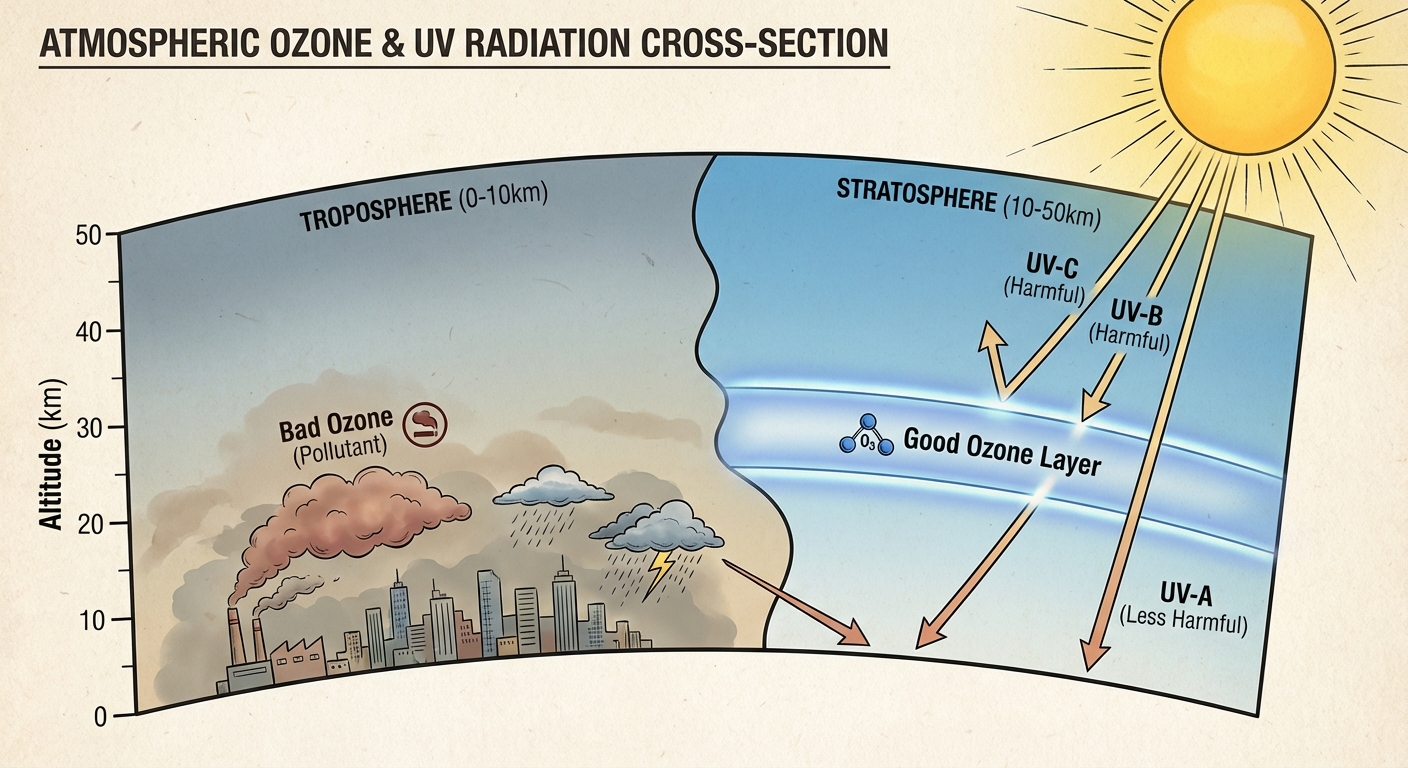
Ultraviolet Radiation and Filtration
Not all solar radiation is the same. The ozone layer filters specific wavelengths:
- UV-A: The longest wavelength. Not absorbed by ozone. Reaches Earth; causes skin aging.
- UV-B: Medium wavelength. Strongly absorbed by ozone. Causes sunburn and skin cancer.
- UV-C: The shortest, most dangerous wavelength. Entirely absorbed by stratospheric ozone and oxygen.
Natural Formation and Breakdown
Ozone is continuously formed and broken down in a natural, balanced cycle driven by sunlight.
Formation: High-energy UV-C strikes an oxygen molecule ($O2$), splitting it.
Then, free oxygen atoms collide with $O2$ to form ozone:Breakdown:UV radiation strikes an ozone molecule, splitting it back into $O2$ and a free oxygen atom, absorbing the UV energy in the process.
Ozone Depletion
Anthropogenic Causes: The Role of CFCs
In the mid-20th century, humans introduced Chlorofluorocarbons (CFCs). These chemicals were used as coolants (refrigerators/ACs), propellants (aerosol sprays), and blowing agents (styrofoam/insulation) because they were non-flammable and chemically stable in the troposphere.
However, because they are so stable, they do not break down rain or dissolve in water. They drift upward into the stratosphere. Once there, high-energy UV radiation breaks them apart, releasing Chlorine ($Cl$).
The Chemical Mechanism of Depletion
The destruction of ozone by chlorine is a catalytic process. This means a single chlorine atom can destroy up to 100,000 ozone molecules before being removed from the stratosphere.
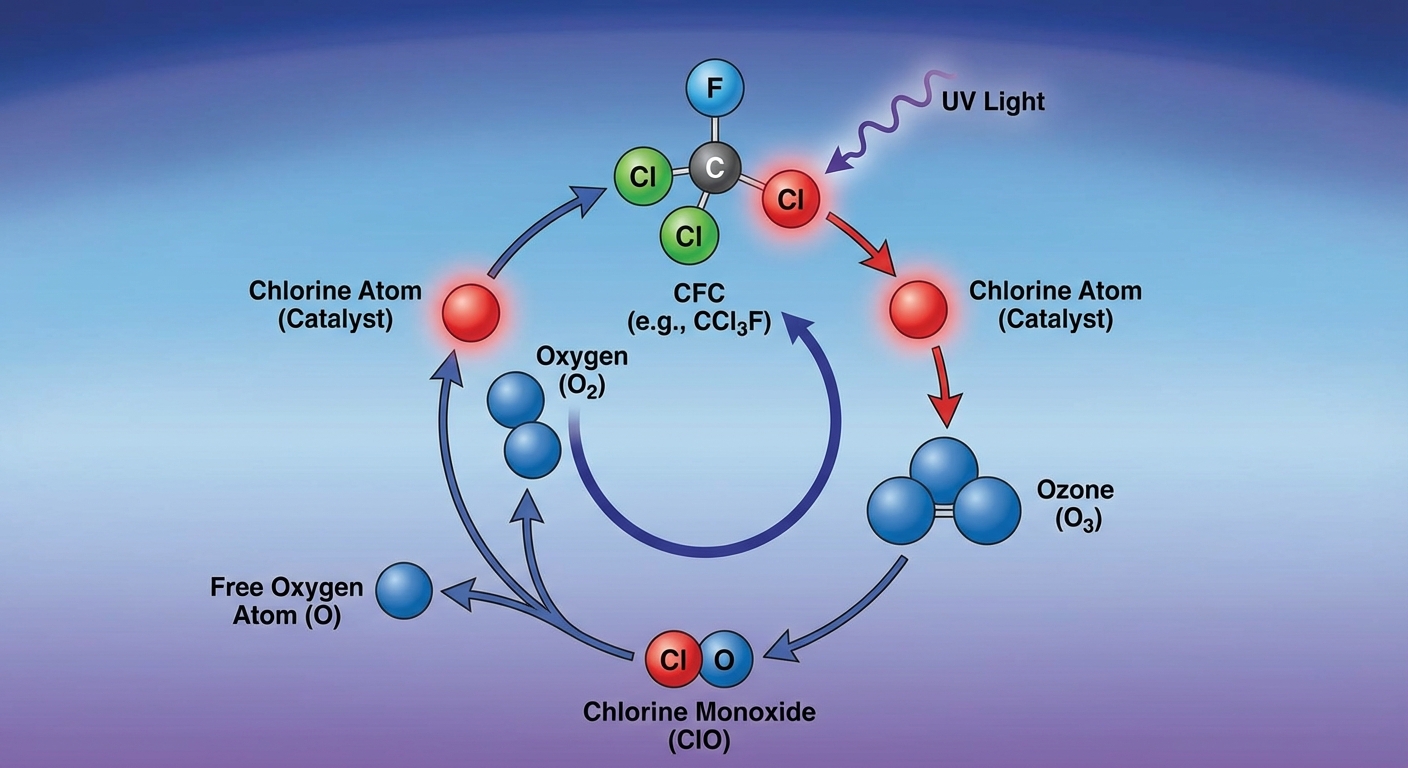
Step 1: The Attack
UV radiation breaks a chlorine atom free from a CFC molecule. The chlorine atom attacks an ozone molecule, pulling an oxygen atom away to form Chlorine Monoxide ($ClO$) and leaving normal oxygen ($O2$).
Step 2: The Reset
A free oxygen atom exerts a pull on the oxygen in the Chlorine Monoxide. The bond breaks, forming $O2$ and releasing the Chlorine atom back into the atmosphere to find another ozone molecule to destroy.
Because the Chlorine is regenerated at the end, it acts as a catalyst. It is not consumed in the reaction.
The Antarctic Ozone "Hole"
Depletion is most severe over Antarctica during its spring (September–October). This occurs due to:
- Polar Stratospheric Clouds (PSCs): Ice clouds that form only in extreme cold.
- Surface Chemistry: These clouds provide a surface for chlorine nitrates to convert into reactive chlorine gas ($Cl_2$) during the dark winter.
- Spring Thaw: When the sun rises in spring, UV radiation splits the accumulated $Cl_2$, unleashing a massive pulse of ozone-destroying chlorine atoms.
Environmental and Health Effects
As the ozone layer thins, more UV-B radiation hits the surface:
- Human Health: Increased rates of skin cancer (melanoma), cataracts (clouding of the eye lens), and suppression of the immune system.
- Marine Ecosystems: Disruption of phytoplankton photosynthesis. Since phytoplankton are the base of the marine food web, this affects the entire ocean ecosystem.
- Terrestrial Plants: Damage to plant DNA, reduced crop yields, and inhibition of photosynthesis.
Reducing Ozone Depletion
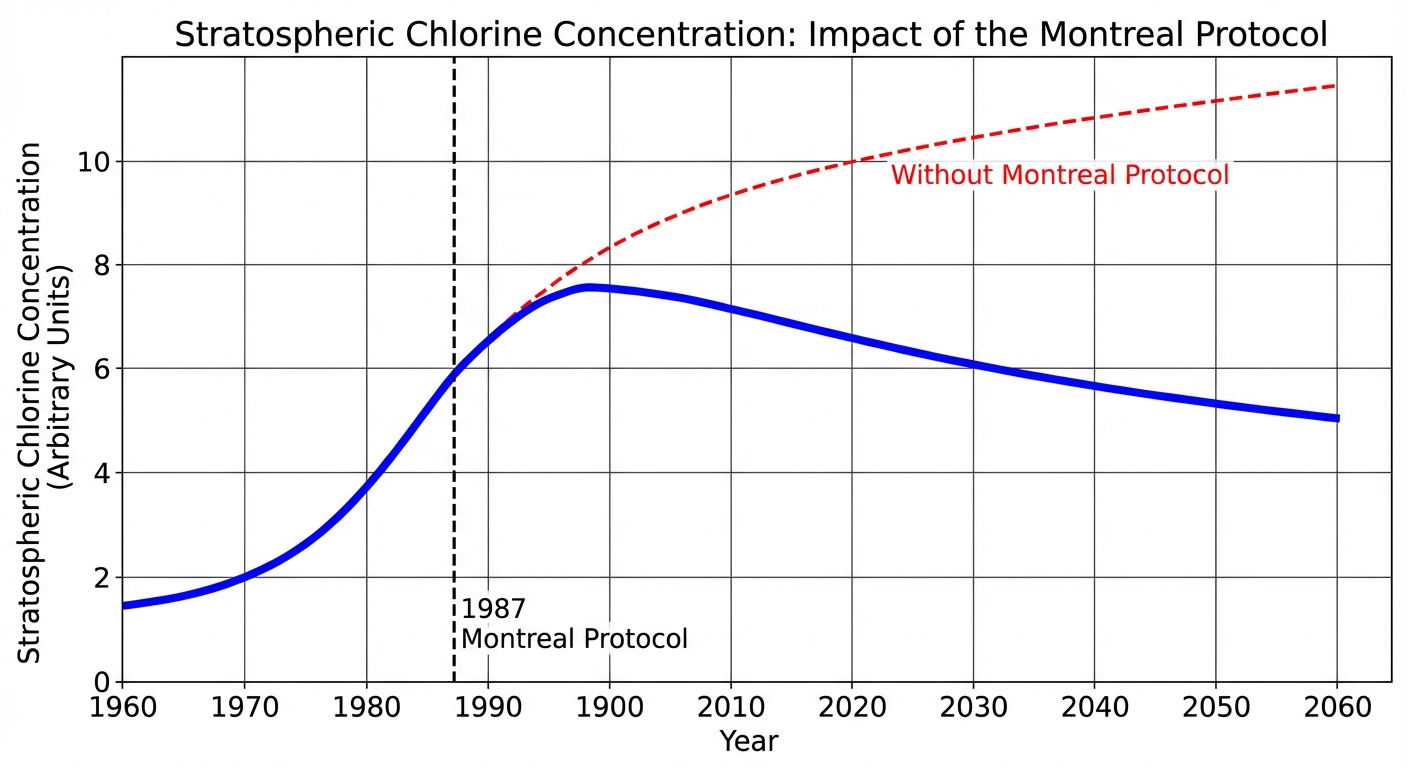
The Montreal Protocol (1987)
The Montreal Protocol is widely considered the most successful global environmental treaty in history.
- Goal: To phase out the production and consumption of Ozone Depleting Substances (ODS), specifically CFCs and Halons.
- Result: Global reduction of CFC emissions by over 98%. The ozone layer is currently healing and is projected to recover to 1980 levels by the middle of the 21st century.
Replacements: HCFCs and HFCs
Industries transitioned away from CFCs to alternatives, but these solutions have their own trade-offs.
HCFCs (Hydrochlorofluorocarbons): A temporary replacement. They still contain chlorine but break down faster in the troposphere. They deplete ozone, but much less than CFCs. They act as a transition chemical and are also being phased out.
HFCs (Hydrofluorocarbons): The long-term replacement (found in modern air conditioners).
- Pro: They contain no chlorine, so they do not deplete the ozone layer.
- Con: They are extremely potent Greenhouse Gases (heat-trapping potential thousands of times higher than $CO_2$).
The Kigali Amendment
Recognizing the climate threat of HFCs, the Kigali Amendment (2016) to the Montreal Protocol aims to phase down HFCs in favor of climate-friendly alternatives (like hydrofluoroolefins or natural refrigerants).
Common Mistakes and Pitfalls
- MISTAKE: Confusing the Ozone Hole with Global Warming.
- Correction: These are separate issues. The ozone hole creates more UV radiation (cancer risk), not more heat. While CFCs are greenhouse gases, the main mechanism of global warming is the greenhouse effect trapping infrared radiation, while ozone depletion involves UV radiation breaking chemical bonds.
- MISTAKE: Thinking Ozone Depletion causes the Greenhouse Effect.
- Correction: Ozone depletion technically cools the stratosphere slightly. Increased UV striking earth contributes a negligible amount of heat energy compared to the greenhouse effect.
- MISTAKE: Assuming HFCs are perfect solutions.
- Correction: HFCs saved the ozone layer but are contributing largely to climate change. This is a classic example of an environmental trade-off.
- MISTAKE: Thinking Ozone Depletion is greatest at the Equator.
- Correction: Depletion is greatest at the Poles (especially Antarctica) due to the unique weather conditions (Polar Stratospheric Clouds) needed to concentrate the reactive chlorine.