Unit 8: Study Guide for Acid-Base Fundamentals
The Nature of Acids and Bases
Acid-base chemistry is central to understanding chemical reactivity in aqueous solutions. In AP Chemistry, we move beyond simple definitions to understand how and why protons move between molecules.
Definitions and Models
There are two primary models used to define acids and bases in this unit. While the Arrhenius definition is historically significant, the Brønsted-Lowry model is the standard for AP Chemistry equilibrium problems.
Arrhenius Definition
- Acid: A substance that increases the concentration of hydrogen ions ($H^+$) in aqueous solution.
- Base: A substance that increases the concentration of hydroxide ions ($OH^-$) in aqueous solution.
- Limitation: Only applies to aqueous solutions and fails to explain bases like ammonia ($NH_3$) that lack an $OH$ group.
Brønsted-Lowry Definition
- Acid: A proton ($H^+$) donor.
- Base: A proton ($H^+$) acceptor.
- Key Concept: Acid-base reactions are essentially proton transfer reactions.
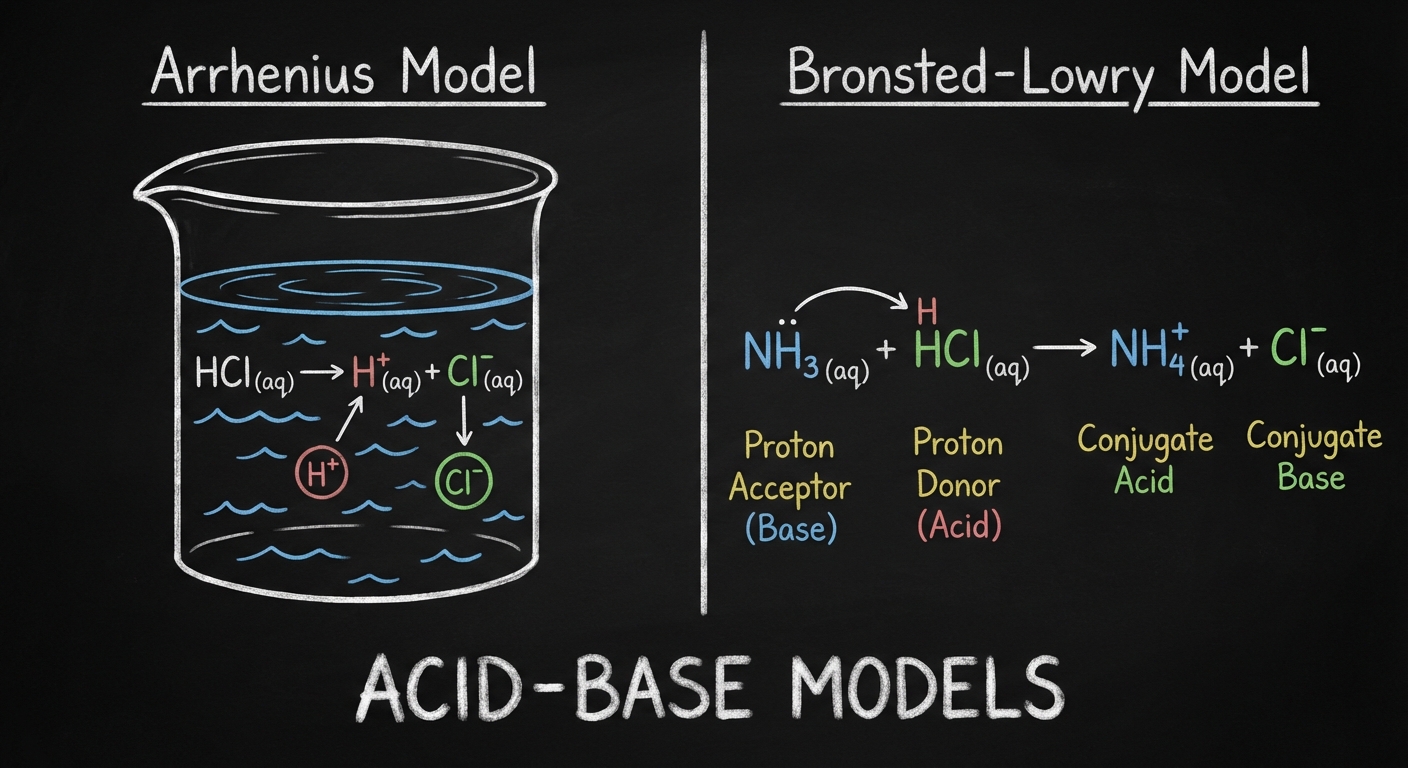
Conjugate Acid-Base Pairs
In a Brønsted-Lowry reaction, an acid turns into a base (after losing a proton), and a base turns into an acid (after gaining a proton). These are called conjugate pairs.
General Equation:
- $HA$ is the acid; $A^-$ is its conjugate base.
- $B$ is the base; $HB^+$ is its conjugate acid.
Conjugate Strength Rule: The stronger the acid, the weaker its conjugate base. Conversely, a strong base has a negligible conjugate acid.
Amphoterism (Amphiprotic Species)
Some substances can act as either an acid or a base depending on what they react with. The most common example is water.
- Acting as a base: $HCl + H2O \rightarrow Cl^- + H3O^+$
- Acting as an acid: $NH3 + H2O \rightleftharpoons NH_4^+ + OH^-$
pH, pOH, and the Autoionization of Water
The Ion-Product Constant ($K_w$)
Even pure water conducts electricity slightly because it acts as both an acid and a base with itself. This is called autoionization.
The equilibrium constant for this process is $Kw$. At $25^\circ$C:
Important Note on Temperature: Because autoionization is endothermic, $K_w$ increases as temperature increases. Therefore, at $50^\circ$C, the pH of neutral water is less than 7 (though it is still neutral because $[H^+] = [OH^-]$).
The pH Scale and Calculations
The pH scale is logarithmic, meaning a change of 1 pH unit represents a 10-fold change in acidity.
Key Formulas:
| Calculation | Formula |
|---|---|
| pH | $pH = -\log[H^+]$ |
| pOH | $pOH = -\log[OH^-]$ |
| Relationship | $pH + pOH = 14.00$ (at $25^\circ$C) |
| Finding Conc. | $[H^+] = 10^{-pH}$ |
Strong Acids and Bases
Strong electrolytes dissociate completely ($100\%$) in water. This makes calculation simple because the concentration of the ion is derived directly from the concentration of the molecule.
The Strong Acids (Memorize these 6-7):
- $HCl$ (Hydrochloric)
- $HBr$ (Hydrobromic)
- $HI$ (Hydroiodic)
- $HNO_3$ (Nitric)
- $H2SO4$ (Sulfuric - only the first proton is strong)
- $HClO_4$ (Perchloric)
The Strong Bases:
Group 1 Hydroxides (e.g., $NaOH$, $KOH$) and heavy Group 2 Hydroxides (e.g., $Ba(OH)2$, $Sr(OH)2$).
Worked Example:
Calculate the pH of $0.020 M$ $Ba(OH)_2$.
- Stoichiometry: $Ba(OH)_2 \rightarrow Ba^{2+} + 2OH^-$
- $[OH^-] = 2 \times 0.020\,M = 0.040\,M$
- $pOH = -\log(0.040) = 1.40$
- $pH = 14.00 - 1.40 = 12.60$
Weak Acid and Base Equilibria
Most acids and bases are weak, meaning they only partially dissociate. We must use equilibrium constants to define their strength.
Dissociation Constants ($Ka$ and $Kb$)
For a weak acid $HA$:
For a weak base $B$:
Concept Check: A larger $K_a$ value implies the equilibrium lies further to the right, meaning the acid is stronger (more dissociated).
Relationship Between $Ka$ and $Kb$
For any conjugate acid-base pair:
This mathematical relationship proves that as acid strength increases ($Ka \uparrow$), the conjugate base strength decreases ($Kb \downarrow$).
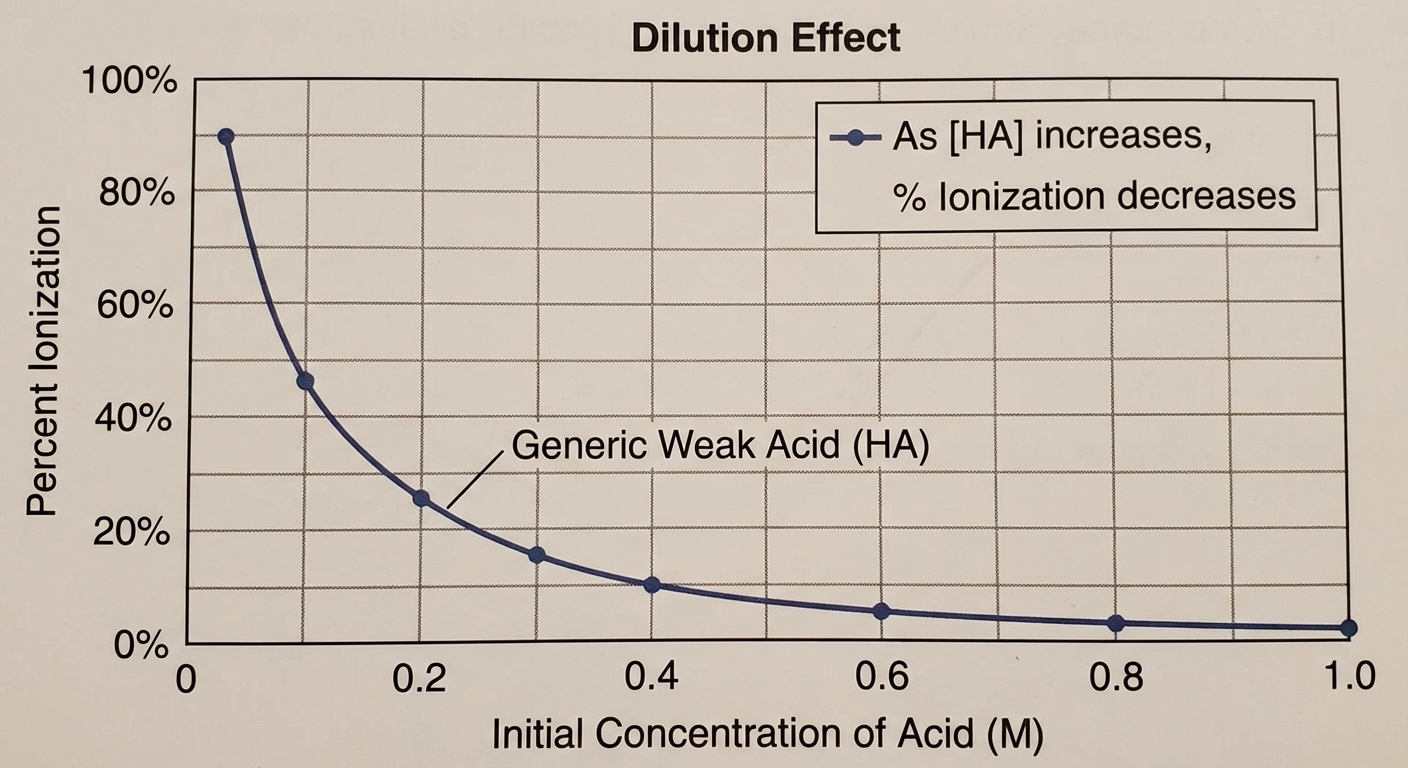
Percent Ionization
Note that as the initial concentration of a weak acid decreases (becomes more dilute), the percent ionization increases (Le Chatelier's Principle regarding dilution), even though the total $[H^+]$ decreases.
Molecular Structure of Acids and Bases
Why is $HF$ a weak acid while $HCl$ is strong? Why is $HClO_4$ stronger than $HClO$? The answer lies in molecular structure and bond polarity.
Binary Acids ($H-X$)
Acid strength depends on how easily the $H-X$ bond is broken.
- Bond Polarity (Across a Period): As electronegativity increases (left to right), the bond becomes more polar, making the $H$ more positive and easier to remove. Therefore, acidity increases.
- $CH4 < NH3 < H_2O < HF$
- Bond Strength (Down a Group): As the size of atom $X$ increases (top to bottom), the bond length increases and bond strength decreases. Weaker bonds break easier. This is the dominant factor.
- $HF < HCl < HBr < HI$ (Acidity increases going down)
Oxyacids ($H-O-Z$)
These are acids containing hydrogen, oxygen, and a central nonmetal $Z$.
- Electronegativity of Z: If $Z$ is highly electronegative, it pulls electron density away from the $O-H$ bond, weakening it and making the $H^+$ easier to remove (Inductive Effect).
- $HOCl > HOBr > HOI$
- Number of Oxygen Atoms: Adding Oxygens to the central atom increases the electron withdrawal, stabilizing the negative charge on the conjugate base.
- $HClO < HClO2 < HClO3 < HClO_4$
- Explanation: $HClO4$ has three non-protonated oxygens pulling electron density, making the $O-H$ bond extremely weak and the resulting $ClO4^-$ anion very stable due to resonance.
Common Mistakes & Pitfalls
- Sig Figs in Logs: The number of significant figures in the concentration determines the number of decimal places in the pH/pOH.
- $[H^+] = 1.0 \times 10^{-3}$ (2 sig figs) $\rightarrow pH = 3.00$ (2 decimal places).
- Confusing Weak with Dilute: A strong acid can be dilute ($0.0001 M$ $HCl$), and a weak acid can be concentrated ($15 M$ Acetic Acid). Strength refers to dissociation ($K$), not molarity.
- Neglecting Stoichiometry for Group 2 Bases: Students often forget that $Ca(OH)_2$ produces two hydroxide ions. $[OH^-] = 2 \times [Base]$.
- Misusing $K$ values: You cannot use $Ka$ to solve for the pH of a base solution. You must convert $Ka$ to $Kb$ first using $Kw$.
- Temperature Trap: Assuming $pH = 7$ is always neutral. At higher temperatures, $Kw > 1.0 \times 10^{-14}$, so neutral pH < 7.